Documente Academic
Documente Profesional
Documente Cultură
Cem 3005 Tutorial August 2013 Natural Products and Organoanalytical Chemistry
Încărcat de
Zama MakhathiniDescriere originală:
Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Cem 3005 Tutorial August 2013 Natural Products and Organoanalytical Chemistry
Încărcat de
Zama MakhathiniDrepturi de autor:
Formate disponibile
1
CEM 3005 Tutorial Natural Products and Organoanalytical Chemistry
August 2013
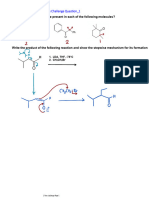
1) Present steps (with mechanisms) for each of the dotted arrows in the following natural product biosynthesis. Which class does each one belong to? a)
b)
c) For Boldine, a benzylisoquinoline alkaloid: i) Use a phenol oxidative coupling (following some selective methylations) to cyclize to the key intermediate shown, and ii) a 1,2-rearrangement involving a carbocation (Wheland-like) to transform to the natural product.
2 2) Assign a class of natural product (fatty acid, polyketide, alkaloid, terpene or steroid) to each of the following natural products, explaining the biosynthetic source:
3) Present a protocol for chemical separation of each of the following: a)
b)
4) Present structures for the following: a) Hint: Note resonance at ~ 205 ppm in 13C NMR spectrum + IR
b) Hint: Take note of IR
RH
S-ar putea să vă placă și
- New Microsoft Word DocumentDocument2 paginiNew Microsoft Word DocumentJithesh ParambathÎncă nu există evaluări
- Por Jorge L: Uis Breña OréDocument32 paginiPor Jorge L: Uis Breña OréAlexa TorresÎncă nu există evaluări
- Chemistry Worksheet on Alcohols, Phenols and Carboxylic AcidsDocument8 paginiChemistry Worksheet on Alcohols, Phenols and Carboxylic AcidsmanasvÎncă nu există evaluări
- Class: XII Marks: 60 Time: One Period: Bangladesh International Tutorial Class Test (I) Session: 2021 - 22Document13 paginiClass: XII Marks: 60 Time: One Period: Bangladesh International Tutorial Class Test (I) Session: 2021 - 22JerryÎncă nu există evaluări
- Worksheet of Carbonyl CompoundsDocument11 paginiWorksheet of Carbonyl CompoundsAyush ChaurasiaÎncă nu există evaluări
- From Biosynthesis to Total Synthesis: Strategies and Tactics for Natural ProductsDe la EverandFrom Biosynthesis to Total Synthesis: Strategies and Tactics for Natural ProductsÎncă nu există evaluări
- DPP803Document7 paginiDPP803anikephantomÎncă nu există evaluări
- Alcohol, Phenol and EthersDocument7 paginiAlcohol, Phenol and EthersgreekyÎncă nu există evaluări
- QOI0809 AlkenesDocument30 paginiQOI0809 Alkenesmtucker17Încă nu există evaluări
- Gaikwad Institute NEET & JEE Chemistry Class on Alcohols, Phenols & EthersDocument1 paginăGaikwad Institute NEET & JEE Chemistry Class on Alcohols, Phenols & EthersAKASH NAMADEÎncă nu există evaluări
- Guia de Ejercicios de Sintesis OrganicaDocument5 paginiGuia de Ejercicios de Sintesis Organicajnacho099Încă nu există evaluări
- CHM556 TUTORIAL 1 ALDEHYDES AND KETONESDocument5 paginiCHM556 TUTORIAL 1 ALDEHYDES AND KETONESNik Muhammad Afiq AzlanÎncă nu există evaluări
- ARTC BIOC 022 - UnlockedDocument6 paginiARTC BIOC 022 - UnlockedAna Martha Remigio RamirezÎncă nu există evaluări
- Ach 2234 Organic Chemistry PDFDocument4 paginiAch 2234 Organic Chemistry PDFlucas omondiÎncă nu există evaluări
- Homework 1 (Ethanol) PDFDocument3 paginiHomework 1 (Ethanol) PDFBrittany MonteverdeÎncă nu există evaluări
- First Periodical Test, March, 2021Document2 paginiFirst Periodical Test, March, 2021Vivek PatelÎncă nu există evaluări
- CH110 Tutorial SHEET 8 - Organic ChemistryDocument10 paginiCH110 Tutorial SHEET 8 - Organic ChemistryJames MukopaÎncă nu există evaluări
- Revision Organic Tutorial 2Document3 paginiRevision Organic Tutorial 2Danish HamizanÎncă nu există evaluări
- 2016 Worksheet For Medicine StudentsDocument2 pagini2016 Worksheet For Medicine StudentsbedadadenbalprosperityÎncă nu există evaluări
- Nada Albuainain - Work Sheet On Alcohols and Aldehyde and Ketones. (SL)Document3 paginiNada Albuainain - Work Sheet On Alcohols and Aldehyde and Ketones. (SL)Nada AlbuainainÎncă nu există evaluări
- Class 12 Chemistry PYQs Shobhit NirwanDocument68 paginiClass 12 Chemistry PYQs Shobhit NirwanDron Pande83% (12)
- CHE-06 ENG-J18 CompressedDocument4 paginiCHE-06 ENG-J18 CompressedManish KumarÎncă nu există evaluări
- Alcohols 2 QPDocument6 paginiAlcohols 2 QPHailey WongÎncă nu există evaluări
- Assignment 3 BLC F20Document2 paginiAssignment 3 BLC F20Rémi MartineauÎncă nu există evaluări
- Carbonyl-Based Nucleophiles Challenge Question - 1Document3 paginiCarbonyl-Based Nucleophiles Challenge Question - 1yavaxoh935Încă nu există evaluări
- Hsslive-Xii-Chem-Qb-Ans-10. HDocument9 paginiHsslive-Xii-Chem-Qb-Ans-10. HJanet RoyÎncă nu există evaluări
- Assignment 1 - Aldehyde and Ketone Mac-Jul 2013Document2 paginiAssignment 1 - Aldehyde and Ketone Mac-Jul 2013anessismanisÎncă nu există evaluări
- Alcohols Past PapersDocument5 paginiAlcohols Past PapersDhairya KumarÎncă nu există evaluări
- Chapter Sk027 OverviewDocument12 paginiChapter Sk027 OverviewNor AfidahÎncă nu există evaluări
- HW2 2013Document3 paginiHW2 2013kitty2911Încă nu există evaluări
- TOPIC 8 EXERCISE 4 - Alcohols Reactions and IsomersDocument1 paginăTOPIC 8 EXERCISE 4 - Alcohols Reactions and IsomersemiliaÎncă nu există evaluări
- Alcohol Phenol Ether OldDocument1 paginăAlcohol Phenol Ether OldAdvanced AcademyÎncă nu există evaluări
- Hydrocarbons JeeDocument9 paginiHydrocarbons JeeDanish AlamÎncă nu există evaluări
- Biosurfactants: A Sustainable Replacement For Chemical Surfactants?Document9 paginiBiosurfactants: A Sustainable Replacement For Chemical Surfactants?Jamal Ud-DinÎncă nu există evaluări
- Revision Organic Tutorial 2 - MSDocument5 paginiRevision Organic Tutorial 2 - MSDanish HamizanÎncă nu există evaluări
- Tutorial 5.1Document3 paginiTutorial 5.1Ijal JaminÎncă nu există evaluări
- Working With Hazardous Chemicals: A Publication of Reliable Methods For The Preparation of Organic CompoundsDocument3 paginiWorking With Hazardous Chemicals: A Publication of Reliable Methods For The Preparation of Organic CompoundsSilambarasan SivalingamÎncă nu există evaluări
- Grade Xii (Chemistry) : Aldehydes, Ketones and Carboxylic Acids (Term - 2) : Most Expecting QuestionsDocument5 paginiGrade Xii (Chemistry) : Aldehydes, Ketones and Carboxylic Acids (Term - 2) : Most Expecting QuestionsSupreeta KhatiwadaÎncă nu există evaluări
- 4 Phenylbut 3 en 2 One 6Document5 pagini4 Phenylbut 3 en 2 One 6Hóa Học Thạc SỹÎncă nu există evaluări
- Chemical Biotechnology For The Specific Oxyfunctionalization of Hydrocarbons On A Technical ScaleDocument10 paginiChemical Biotechnology For The Specific Oxyfunctionalization of Hydrocarbons On A Technical ScaleMehran IsgandarliÎncă nu există evaluări
- 2018 Hydroxy Cpds Lecture NotesDocument23 pagini2018 Hydroxy Cpds Lecture NotesAmelia WongÎncă nu există evaluări
- Organic Chemistry Practice TestDocument4 paginiOrganic Chemistry Practice TestGirishmaÎncă nu există evaluări
- Chapter 12 Aldehyde & KetonesDocument10 paginiChapter 12 Aldehyde & KetonesForzen flamesÎncă nu există evaluări
- Halides and Arenes Chemistry ProblemsDocument7 paginiHalides and Arenes Chemistry ProblemsVishnuÎncă nu există evaluări
- Regorosa LKA Worksheet 1&2Document5 paginiRegorosa LKA Worksheet 1&2Lenuel RegorosaÎncă nu există evaluări
- Physical Sciences p2 Last PushDocument54 paginiPhysical Sciences p2 Last Pushmonkbons15Încă nu există evaluări
- Working With Hazardous Chemicals: A Publication of Reliable Methods For The Preparation of Organic CompoundsDocument3 paginiWorking With Hazardous Chemicals: A Publication of Reliable Methods For The Preparation of Organic CompoundsMd. BadrulÎncă nu există evaluări
- Chemistry CT 1 22-23Document6 paginiChemistry CT 1 22-23Sancia SamÎncă nu există evaluări
- Che 303 Cat 1Document1 paginăChe 303 Cat 1KEVIN OMONDIÎncă nu există evaluări
- Hsslive-10. Haloalkanes and HaloarenesDocument7 paginiHsslive-10. Haloalkanes and Haloarenessindhumv631Încă nu există evaluări
- M.phil. Questions End Dec 2017Document3 paginiM.phil. Questions End Dec 2017Biswajit RoyÎncă nu există evaluări
- XII -Second Unit Test -CHEMISTRYDocument3 paginiXII -Second Unit Test -CHEMISTRYrshobana78Încă nu există evaluări
- QUESTION 1 (52 Marks) : Biochemistry 3 BCH 314Document4 paginiQUESTION 1 (52 Marks) : Biochemistry 3 BCH 314victorÎncă nu există evaluări
- Chemistry MCQDocument9 paginiChemistry MCQMOHD SARWAR AZAMÎncă nu există evaluări
- Tutorial Questions On Free Radical Reactions PDFDocument3 paginiTutorial Questions On Free Radical Reactions PDFHarry ZgamboÎncă nu există evaluări
- Introduction MCC Oxo ProcessDocument5 paginiIntroduction MCC Oxo ProcessDeep PatelÎncă nu există evaluări
- Tugas - 6 Senyawa AlkunaDocument3 paginiTugas - 6 Senyawa AlkunaBaiq ArinÎncă nu există evaluări
- Alcohols Ncert SolutionDocument35 paginiAlcohols Ncert SolutionJewelÎncă nu există evaluări
- Examen 1Document12 paginiExamen 1Sahimara de JesúsÎncă nu există evaluări
- Transition Metal-Catalyzed Benzofuran Synthesis: Transition Metal-Catalyzed Heterocycle Synthesis SeriesDe la EverandTransition Metal-Catalyzed Benzofuran Synthesis: Transition Metal-Catalyzed Heterocycle Synthesis SeriesÎncă nu există evaluări
- Cem 3005W Tutorial On Stereochemistry and Nmr/Conformation: Solutions April 2013Document6 paginiCem 3005W Tutorial On Stereochemistry and Nmr/Conformation: Solutions April 2013Zama MakhathiniÎncă nu există evaluări
- Aromatic Electrophilic Substitution Test Your Understanding Answers 2012Document1 paginăAromatic Electrophilic Substitution Test Your Understanding Answers 2012Zama MakhathiniÎncă nu există evaluări
- CEM3005W Formula Sheet For Physical Chemistry of LiquidsDocument2 paginiCEM3005W Formula Sheet For Physical Chemistry of LiquidsZama MakhathiniÎncă nu există evaluări
- CEM 3005W Typed Notes Reaction Classes and Pericyclic Reactions 2013Document12 paginiCEM 3005W Typed Notes Reaction Classes and Pericyclic Reactions 2013Zama MakhathiniÎncă nu există evaluări
- InvestmentsDocument2 paginiInvestmentsZama MakhathiniÎncă nu există evaluări
- Vasco - Sfundamentals 11-02-2011 PDFDocument155 paginiVasco - Sfundamentals 11-02-2011 PDFZama MakhathiniÎncă nu există evaluări
- Review Article Guidelines for Students on RotationDocument16 paginiReview Article Guidelines for Students on RotationZama MakhathiniÎncă nu există evaluări
- CEM 3005W Aromatic and Heteroaromatic Notes 2013Document26 paginiCEM 3005W Aromatic and Heteroaromatic Notes 2013Zama MakhathiniÎncă nu există evaluări
- CEM ReferncesDocument1 paginăCEM ReferncesZama MakhathiniÎncă nu există evaluări
- Cem2008s Tut 2 - 2012 AnswersDocument2 paginiCem2008s Tut 2 - 2012 AnswersZama MakhathiniÎncă nu există evaluări
- A Major Constituent of HumanDocument3 paginiA Major Constituent of HumanZama MakhathiniÎncă nu există evaluări
- The Acidity and Basicity of Organic Acids and Bases For VULADocument24 paginiThe Acidity and Basicity of Organic Acids and Bases For VULAZama MakhathiniÎncă nu există evaluări
- Apply for scholarships and bursaries from top South African fundersDocument1 paginăApply for scholarships and bursaries from top South African fundersZama MakhathiniÎncă nu există evaluări
- 3MS Metric Spaces: Department of Mathematics and Applied MathematicsDocument2 pagini3MS Metric Spaces: Department of Mathematics and Applied MathematicsZama MakhathiniÎncă nu există evaluări
- CEM 2008S Tutorial 10 2012Document2 paginiCEM 2008S Tutorial 10 2012Zama MakhathiniÎncă nu există evaluări
- Bonding in Transition Metal ComplexesDocument41 paginiBonding in Transition Metal ComplexesZama Makhathini100% (1)
- 11 Tut 6Document3 pagini11 Tut 6Zama MakhathiniÎncă nu există evaluări
- mgpg11 - A Beginner's Guide To Uncertainty of Measurement by Stephanie BellDocument41 paginimgpg11 - A Beginner's Guide To Uncertainty of Measurement by Stephanie Bellarylananyla100% (2)
- 11 Tut 7Document3 pagini11 Tut 7Zama MakhathiniÎncă nu există evaluări
- 11 Tut 5 SolDocument3 pagini11 Tut 5 SolZama MakhathiniÎncă nu există evaluări
- 2010 CEM2008S-2010 - Final ExaminationDocument12 pagini2010 CEM2008S-2010 - Final ExaminationZama MakhathiniÎncă nu există evaluări
- (Ebook - English) How To Improve Your MemoryDocument2 pagini(Ebook - English) How To Improve Your MemorytayyabemeÎncă nu există evaluări
- 11 Tut 5Document4 pagini11 Tut 5Zama MakhathiniÎncă nu există evaluări
- 11 Tut 4Document4 pagini11 Tut 4Zama MakhathiniÎncă nu există evaluări
- 11 Tut ADocument3 pagini11 Tut AZama MakhathiniÎncă nu există evaluări
- 11 Tut ADocument3 pagini11 Tut AZama MakhathiniÎncă nu există evaluări
- 11 Tut ADocument3 pagini11 Tut AZama MakhathiniÎncă nu există evaluări
- Jan98 MA1002 CalculusDocument6 paginiJan98 MA1002 CalculusZama MakhathiniÎncă nu există evaluări
- 18.1 Energy in The Microscopic World: 33-131 Matter and Interactions Fall 2010Document9 pagini18.1 Energy in The Microscopic World: 33-131 Matter and Interactions Fall 2010Zama MakhathiniÎncă nu există evaluări
- Properties of EnzymesDocument15 paginiProperties of EnzymesAreej AhmadÎncă nu există evaluări
- Food Chemistry - Activity 1Document2 paginiFood Chemistry - Activity 1Leda PeñaÎncă nu există evaluări
- Practice - DNA, RNA, Gene Expression (Solutions)Document4 paginiPractice - DNA, RNA, Gene Expression (Solutions)SophieÎncă nu există evaluări
- Translation in ProkaryotesDocument25 paginiTranslation in ProkaryotesNaomi Lamtiur0% (1)
- CXS - 033e Codex Aceite de OlivaDocument9 paginiCXS - 033e Codex Aceite de OlivaAnita Rosmery More LupúÎncă nu există evaluări
- Glycolysis and TCA Cycle - ShuttlesDocument1 paginăGlycolysis and TCA Cycle - ShuttlesDr. SHIVA AITHALÎncă nu există evaluări
- ECS129 Quiz 8 Secondary Structure PredictionDocument5 paginiECS129 Quiz 8 Secondary Structure PredictionSasikala RajendranÎncă nu există evaluări
- Rubric For DNA and RNA ModelDocument2 paginiRubric For DNA and RNA ModelDrew MarasiganÎncă nu există evaluări
- Every Vitamin PageDocument9 paginiEvery Vitamin Pageapi-3740719100% (1)
- Chapter 11 - 1Document12 paginiChapter 11 - 1JeanPaule JoumaaÎncă nu există evaluări
- Hematologi dan Kimia Bulan September-Oktober 2021Document46 paginiHematologi dan Kimia Bulan September-Oktober 2021Fernando PkpÎncă nu există evaluări
- Enzyme InhibitionDocument17 paginiEnzyme InhibitionazwelljohnsonÎncă nu există evaluări
- Inbound 2949823744268348325Document15 paginiInbound 2949823744268348325Tristan SiocoÎncă nu există evaluări
- Translation: Protein Biosynthesis Mrna Amino AcidsDocument4 paginiTranslation: Protein Biosynthesis Mrna Amino AcidsMalick AwanÎncă nu există evaluări
- CH4:Biological molecules overviewDocument34 paginiCH4:Biological molecules overviewToXicÎncă nu există evaluări
- Biological MoleculesDocument38 paginiBiological MoleculesmuzammalÎncă nu există evaluări
- Biochemistry Book 2Document119 paginiBiochemistry Book 2Jugnu JugnuÎncă nu există evaluări
- BI115 หลักชีววิทยา (Priciples of biology)Document315 paginiBI115 หลักชีววิทยา (Priciples of biology)สาธิต ปริ นทร์ทองÎncă nu există evaluări
- Bacterial Cell WallDocument20 paginiBacterial Cell WallNavnath HatvateÎncă nu există evaluări
- Pmbi130 FT 10 2022 1Document8 paginiPmbi130 FT 10 2022 1Kaoma MofyaÎncă nu există evaluări
- Analisis Kadar Vitamin C ManggaDocument5 paginiAnalisis Kadar Vitamin C ManggarilaÎncă nu există evaluări
- RNA StructureDocument52 paginiRNA StructureSoumyabrataÎncă nu există evaluări
- Cell Membranes and Energy Storage: The Role of Lipids in BiologyDocument2 paginiCell Membranes and Energy Storage: The Role of Lipids in Biologydanielle stephanieÎncă nu există evaluări
- Posttranslatonal Modfcatons of ProteinsDocument21 paginiPosttranslatonal Modfcatons of ProteinsRizka Ayu Atik SaputriÎncă nu există evaluări
- Protein Folding Virtual Activity 1Document5 paginiProtein Folding Virtual Activity 1api-521773978Încă nu există evaluări
- Introduction To Vitamins: With Dr. Georgina CornwallDocument11 paginiIntroduction To Vitamins: With Dr. Georgina CornwallimnasÎncă nu există evaluări
- DNA Replication and StructureDocument2 paginiDNA Replication and Structureloly62006Încă nu există evaluări
- TranscriptionTranslation ActivityDocument3 paginiTranscriptionTranslation ActivityDylan DiazÎncă nu există evaluări
- Metabolism of Other Amino Acids: GlycineDocument9 paginiMetabolism of Other Amino Acids: GlycineAsad AliÎncă nu există evaluări
- March 09, 2023 - DNA AND RNA STRUCTURESDocument4 paginiMarch 09, 2023 - DNA AND RNA STRUCTURESJunaisa OrabÎncă nu există evaluări