Documente Academic
Documente Profesional
Documente Cultură
F321 Module 3 Practice 2
Încărcat de
coughsyrup123Descriere originală:
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
F321 Module 3 Practice 2
Încărcat de
coughsyrup123Drepturi de autor:
Formate disponibile
F321 module 3 Practice 2:
1.
Radium reacts vigorously when added to water.
Ra(s) + 2H2O(l) Ra(OH)2(aq) + H2(g)
(i)
Use the equation to predict two observations that you would see during this
reaction.
.........................................................................................................................
.........................................................................................................................
[2]
(ii)
Predict a pH value for this solution.
.........................................................................................................................
[1]
[Total 3 marks]
2.
Reactions of the Group 2 metals involve removal of electrons. The electrons are
removed more easily as the group is descended and this helps to explain the
increasing trend in reactivity.
(i)
The removal of one electron from each atom in 1 mole of gaseous radium atoms
is called the .....................................................................................................
[2]
The equation for this process in radium is:
.........................................................................................................................
[2]
The King's CE School
(ii)
Atoms of radium have a greater nuclear charge than atoms of calcium.
Explain why, despite this, less energy is needed to remove an electron from a
radium atom than from a calcium atom.
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
[3]
[Total 7 marks]
3.
Chewing chalk has been used for many years to combat excess stomach acid and
indigestion tablets often contain calcium carbonate, CaCO3. Suggest, with the aid of an
equation, how these tablets work.
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
[Total 2 marks]
The King's CE School
4.
Chlorine is used in the preparation of many commercially important materials such as
bleach and iodine.
Bleach is a solution of sodium chlorate(l), NaOCl, made by dissolving chlorine in
aqueous sodium hydroxide.
Cl2(g) + 2NaOH(aq) NaOCl(aq) + NaCl(aq) + H2O(l)
Determine the changes in oxidation number of chlorine during the preparation of bleach
and comment on your results.
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
[Total 3 marks]
5.
Iodine is extracted commercially from seawater with chlorine gas. Seawater contains
very small quantities of dissolved iodide ions, which are oxidised to iodine by the
chlorine gas.
(i)
Write an ionic equation for the reaction that has taken place.
.........................................................................................................................
[2]
(ii)
Use your understanding of electronic structure to explain why chlorine is a
stronger oxidising agent than iodine.
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
[2]
[Total 4 marks]
The King's CE School
6.
The atomic radii of nitrogen and oxygen are shown below.
element
nitrogen
oxygen
atomic radius/nm
0.075
0.073
Explain why a nitrogen atom is larger than an oxygen atom.
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
[Total 4 marks]
The King's CE School
7.
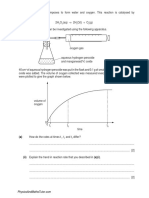
The first ionisation energies of the elements H to K are shown below. Use this diagram
to help with your answers to this question.
2500
1 s t io n is a tio n e n e r g y
/ k J m o l 1
He
N e
2000
1000
Be
M g
Si
500
0
Ar
1500
Li
0
Na
4
Al
10 11 12 13 14 15 16 17 18 19 20
a to m ic n u m b e r
(a)
Define the term first ionisation energy.
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
[3]
(b)
Explain why the first ionisation energies show a general increase across Period 2
(Li to Ne).
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
[2]
[Total 5 marks]
The King's CE School
8.
State and explain the trend in first ionisation energies shown by the elements with the
atomic numbers 2, 10 and 18.
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
[Total 4 marks]
9.
The Group 2 metal strontium, Sr, is very reactive.
Strontium metal is stored under oil and, when exposed to air, the shiny surface of the
strontium becomes dull.
Predict, with an equation, what reaction takes place when strontium is exposed to air.
..................................................................................................................................
..................................................................................................................................
..................................................................................................................................
[Total 2 marks]
10.
The reaction of strontium with water is a redox reaction. A student reacted 0.438 g of
strontium with 200 cm3 of water.
Sr(s) + 2H2O(l) Sr(OH)2(aq) + H2(g)
(i)
Use oxidation numbers to show that strontium has been oxidised in this reaction.
.........................................................................................................................
.........................................................................................................................
.........................................................................................................................
[2]
The King's CE School
(ii)
Calculate how many moles of Sr were reacted.
Ar: Sr, 87.6
answer ............... mol
[1]
(iii)
Calculate the volume, in dm3, of H2(g) produced. You can assume that, under the
experimental conditions, 1.00 mol of H2(g) has a volume of 24.0 dm3.
answer .............. dm3
[1]
(iv)
Calculate the concentration, in mol dm3, of the Sr(OH)2 produced.
answer ............... mol dm3
[1]
[Total 5 marks]
The King's CE School
S-ar putea să vă placă și
- Ib PhysicsDocument10 paginiIb PhysicsAndres LopezÎncă nu există evaluări
- Further Mechanics Exam Pack MSDocument45 paginiFurther Mechanics Exam Pack MScoughsyrup123Încă nu există evaluări
- Topic 4 - Group 7Document9 paginiTopic 4 - Group 7Abirame SivakaranÎncă nu există evaluări
- Nitrogen & Fertilisers 4 QPDocument8 paginiNitrogen & Fertilisers 4 QPUsha PerumalÎncă nu există evaluări
- Chemical Energetics 2 QPDocument11 paginiChemical Energetics 2 QPWilliam TsuiÎncă nu există evaluări
- Balanced Equations & Associated Calc's 05 QPDocument8 paginiBalanced Equations & Associated Calc's 05 QPlmao lmaoÎncă nu există evaluări
- Chlorine and Its Compounds Chemistry Form 3 Topical Questions and AnswersDocument18 paginiChlorine and Its Compounds Chemistry Form 3 Topical Questions and Answersideal writersÎncă nu există evaluări
- Kinetics QuestionsDocument9 paginiKinetics QuestionsridithaÎncă nu există evaluări
- Periodic Table, Group 2 and The Halogens 2 QPDocument14 paginiPeriodic Table, Group 2 and The Halogens 2 QPmalakÎncă nu există evaluări
- t2 Chem Revision Ex 22Document19 paginit2 Chem Revision Ex 22Nicholas OwÎncă nu există evaluări
- P6 Chemical Equilibirua Le Chatelier's Principle and KCDocument73 paginiP6 Chemical Equilibirua Le Chatelier's Principle and KC/ “Nu” /Încă nu există evaluări
- Electricity & Chemistry 1 QPDocument11 paginiElectricity & Chemistry 1 QPValerine VictoriaÎncă nu există evaluări
- As Level Chemistry: Topic 2 - Amount of Substance TestDocument10 paginiAs Level Chemistry: Topic 2 - Amount of Substance Testkarima akterÎncă nu există evaluări
- Redox Group 2 and Group 7 HWDocument19 paginiRedox Group 2 and Group 7 HWsemirah anthonyÎncă nu există evaluări
- 10 Homework PackDocument10 pagini10 Homework PackatasayÎncă nu există evaluări
- I6 Reactions of Ions in Aqueous SolutionDocument16 paginiI6 Reactions of Ions in Aqueous Solution/ “Nu” /Încă nu există evaluări
- Mixed Topic Revision 1 DiffusionDocument23 paginiMixed Topic Revision 1 DiffusionYaakkwÎncă nu există evaluări
- Born-Haber Cycles 1 QP PDFDocument9 paginiBorn-Haber Cycles 1 QP PDFTalha KhanÎncă nu există evaluări
- A-Level Chemistry: Paper 3 Practice Paper 3Document20 paginiA-Level Chemistry: Paper 3 Practice Paper 3Jesus ChristÎncă nu există evaluări
- Particle QuestionsDocument14 paginiParticle QuestionsSaginskyÎncă nu există evaluări
- Water and HydrogenDocument7 paginiWater and HydrogenOkumu KevinsÎncă nu există evaluări
- BondingDocument14 paginiBondingMuizzudin AzaliÎncă nu există evaluări
- Periodicity Past QuestionsDocument17 paginiPeriodicity Past QuestionsMohammad KhanÎncă nu există evaluări
- Reversible Reactions WorksheetDocument14 paginiReversible Reactions WorksheetShriyaÎncă nu există evaluări
- A-Level Paper 3 pp8Document19 paginiA-Level Paper 3 pp822S48 SUNDARAM RAMASUBBU RAKSHAÎncă nu există evaluări
- Module 806Document18 paginiModule 806Hema LataÎncă nu există evaluări
- Question Database For Amount of SubstancesDocument23 paginiQuestion Database For Amount of SubstancesKamrul Alam MasumÎncă nu există evaluări
- F321: Atoms, Bonds and Groups Redox: Plymstock School 1Document7 paginiF321: Atoms, Bonds and Groups Redox: Plymstock School 1Younes AlahmadÎncă nu există evaluări
- Rate of Reaction 2 QP (Tomek)Document9 paginiRate of Reaction 2 QP (Tomek)Tomasz OstrowskiÎncă nu există evaluări
- Structure of The Atom and The Periodic Table Chemistry Form 2 Topical Questions and AnswersDocument27 paginiStructure of The Atom and The Periodic Table Chemistry Form 2 Topical Questions and Answersdonaldsonvincent9Încă nu există evaluări
- Week 26 A2 Questions Name .. /49 1Document5 paginiWeek 26 A2 Questions Name .. /49 1Solace HusseinÎncă nu există evaluări
- Amount of Substance 1 QPDocument10 paginiAmount of Substance 1 QPHajhoj CellÎncă nu există evaluări
- Rate of Reaction PPsDocument23 paginiRate of Reaction PPstcs202231Încă nu există evaluări
- Exam 1Document9 paginiExam 1Alejandro DuarteÎncă nu există evaluări
- Redox 2 QPDocument7 paginiRedox 2 QPPramitaÎncă nu există evaluări
- Redox Questions Igcse ChemDocument7 paginiRedox Questions Igcse ChemCaylinÎncă nu există evaluări
- Periodicity QsDocument15 paginiPeriodicity QsJack SmitÎncă nu există evaluări
- Carbon and Some of Its CompoundsDocument17 paginiCarbon and Some of Its Compoundsbarus michealÎncă nu există evaluări
- Topical Questions For ElectrolysisDocument6 paginiTopical Questions For Electrolysisokguserfucker idontgiveashitÎncă nu există evaluări
- 2 HoursDocument17 pagini2 HoursOTTO OLIMAÎncă nu există evaluări
- Energy Changes, Rate, Reversible, NH3, H2SO4Document5 paginiEnergy Changes, Rate, Reversible, NH3, H2SO4HudaÎncă nu există evaluări
- 2.4 2.5 2.6 Assessed HomeworkDocument7 pagini2.4 2.5 2.6 Assessed HomeworkRabia Rafique100% (1)
- Clour Questions and MarkschemeDocument13 paginiClour Questions and Markschemesquidthekid2005Încă nu există evaluări
- PDF DocumentDocument125 paginiPDF Documentnabila OktavianiÎncă nu există evaluări
- 4.1 Reactivity of Metals 3 QPDocument16 pagini4.1 Reactivity of Metals 3 QPDumpsterFireGamingÎncă nu există evaluări
- Water 1Document10 paginiWater 12025svyasÎncă nu există evaluări
- Cambridge International AS & A Level: CHEMISTRY 9701/22Document12 paginiCambridge International AS & A Level: CHEMISTRY 9701/22Spider Gamer22Încă nu există evaluări
- t2 Chem Revision Ex 12Document16 paginit2 Chem Revision Ex 12Nicholas OwÎncă nu există evaluări
- Aqa A2 Biology LDRDocument21 paginiAqa A2 Biology LDRjames100% (4)
- Equilibria AsDocument39 paginiEquilibria AsyousafÎncă nu există evaluări
- Electrolysis RevisionDocument15 paginiElectrolysis RevisionPunitha PanchaÎncă nu există evaluări
- Amount of Substance CombinedDocument23 paginiAmount of Substance CombinedJainam MehtaÎncă nu există evaluări
- DY Enthalpy TestDocument19 paginiDY Enthalpy TestStormzy 67Încă nu există evaluări
- 1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byDocument9 pagini1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byKelvin DenhereÎncă nu există evaluări
- Atomic and Nuclear PhysicsDocument254 paginiAtomic and Nuclear PhysicsAli AhmedÎncă nu există evaluări
- The Quantum in Chemistry: An Experimentalist's ViewDe la EverandThe Quantum in Chemistry: An Experimentalist's ViewÎncă nu există evaluări
- Advanced Synthesis of Gold and Zirconia Nanoparticles and their CharacterizationDe la EverandAdvanced Synthesis of Gold and Zirconia Nanoparticles and their CharacterizationÎncă nu există evaluări
- Metal Catalysed Carbon-Carbon Bond-Forming ReactionsDe la EverandMetal Catalysed Carbon-Carbon Bond-Forming ReactionsÎncă nu există evaluări
- Ligand Coupling Reactions with Heteroatomic CompoundsDe la EverandLigand Coupling Reactions with Heteroatomic CompoundsEvaluare: 4 din 5 stele4/5 (1)
- F321 Module 3 Practice 3 AnswersDocument4 paginiF321 Module 3 Practice 3 Answerscoughsyrup123Încă nu există evaluări
- F321 Module 3 Practice 5Document4 paginiF321 Module 3 Practice 5coughsyrup123Încă nu există evaluări
- F321 Module 3 Practice 5 AnswersDocument4 paginiF321 Module 3 Practice 5 Answerscoughsyrup123Încă nu există evaluări
- F321 Module 3 Practice 3Document10 paginiF321 Module 3 Practice 3coughsyrup123Încă nu există evaluări
- F321 Module 2 Practice 2 AnswersDocument5 paginiF321 Module 2 Practice 2 Answerscoughsyrup123Încă nu există evaluări
- F321 Module 2 Practice 6Document2 paginiF321 Module 2 Practice 6coughsyrup123Încă nu există evaluări
- F321 Module 3 Practice 1Document10 paginiF321 Module 3 Practice 1coughsyrup123Încă nu există evaluări
- F321 Module 1 Practice 5Document1 paginăF321 Module 1 Practice 5coughsyrup123Încă nu există evaluări
- F321 Module 1 Practice 4Document7 paginiF321 Module 1 Practice 4coughsyrup123Încă nu există evaluări
- Edexcel S1 NotesDocument52 paginiEdexcel S1 Notesmystreet123100% (1)
- F321 Module 1 Practice 1 AnswersDocument3 paginiF321 Module 1 Practice 1 Answerscoughsyrup123Încă nu există evaluări
- F321 01 Jun12 OCR Chemistry Question PaperDocument0 paginiF321 01 Jun12 OCR Chemistry Question PaperDaLilSPÎncă nu există evaluări
- Ib PPT 9 SL PDFDocument38 paginiIb PPT 9 SL PDFzarna nirmal rawalÎncă nu există evaluări
- Results For Kantipudi Steel Cemnt - Rajahmundry - Zonalinfo2Document4 paginiResults For Kantipudi Steel Cemnt - Rajahmundry - Zonalinfo2Manoj Digi LoansÎncă nu există evaluări
- The Periodic Table: TrendsDocument22 paginiThe Periodic Table: TrendsSpace MonkeyÎncă nu există evaluări
- University of Cambridge International Examinations General Certifi Cate of Education Advanced Subsidiary Level and Advanced LevelDocument12 paginiUniversity of Cambridge International Examinations General Certifi Cate of Education Advanced Subsidiary Level and Advanced LevelHubbak KhanÎncă nu există evaluări
- Cambridge International Examinations Cambridge International General Certificate of Secondary EducationDocument20 paginiCambridge International Examinations Cambridge International General Certificate of Secondary EducationAbdulBasitBilalSheikhÎncă nu există evaluări
- Group 4 CATHODIC INHIBITORDocument9 paginiGroup 4 CATHODIC INHIBITORfadelomar28_gmail_coÎncă nu există evaluări
- Special Metals-Corrosion-resistant Alloys For Oil and Gas Production PDFDocument44 paginiSpecial Metals-Corrosion-resistant Alloys For Oil and Gas Production PDFagnotts09Încă nu există evaluări
- Solids Liquids and Gases: Solid PropertiesDocument42 paginiSolids Liquids and Gases: Solid PropertiesAmin Sharif SharifÎncă nu există evaluări
- Experiment No. 10: Confirmatory Test of CODocument7 paginiExperiment No. 10: Confirmatory Test of COFact's FactoryÎncă nu există evaluări
- 1.1 Atomic StructureDocument24 pagini1.1 Atomic Structureabdulla idreesÎncă nu există evaluări
- BASF R0-20 Data SheetDocument1 paginăBASF R0-20 Data SheetAmir RahbariÎncă nu există evaluări
- To Prepare Ferric Hydroxide Sol - Chemistry Practical Class 12 - Learn CBSEDocument1 paginăTo Prepare Ferric Hydroxide Sol - Chemistry Practical Class 12 - Learn CBSESujal JhaÎncă nu există evaluări
- Scicent PPT 9 4 R eDocument101 paginiScicent PPT 9 4 R eApple LouÎncă nu există evaluări
- Equilibrium Constant AnswersDocument18 paginiEquilibrium Constant Answerselvisfan777Încă nu există evaluări
- Xi Salt Analysis em PDFDocument3 paginiXi Salt Analysis em PDFvvn natrajÎncă nu există evaluări
- Unit 7 Review Problem Set 3Document5 paginiUnit 7 Review Problem Set 3api-182809945Încă nu există evaluări
- Precision Steel Warehouse, IncDocument1 paginăPrecision Steel Warehouse, Incapi-28059540Încă nu există evaluări
- Density Galvalume N ZincalumeDocument2 paginiDensity Galvalume N ZincalumeAuranisa SyaikniÎncă nu există evaluări
- C Family Silicon, Silicates and Their TypesDocument6 paginiC Family Silicon, Silicates and Their TypesUsman GhaniÎncă nu există evaluări
- Cadmium vs. ZincDocument7 paginiCadmium vs. Zincvivek_recÎncă nu există evaluări
- Ar (1) .Engineering Chemistry Lab ManualDocument68 paginiAr (1) .Engineering Chemistry Lab ManualAl- Rahim KhanÎncă nu există evaluări
- New Product List Smart LabDocument5 paginiNew Product List Smart LabFadhlan Al AsyrafÎncă nu există evaluări
- The Life Cycle of Materials in Mobile PhonesDocument12 paginiThe Life Cycle of Materials in Mobile PhonesULdialogueÎncă nu există evaluări
- Material Technology IInd SemDocument262 paginiMaterial Technology IInd SemvinayakÎncă nu există evaluări
- Chemical EquilibriaDocument6 paginiChemical EquilibriaHajra NasirÎncă nu există evaluări
- Nonferrous Teaching MatDocument31 paginiNonferrous Teaching MatAsyiqotul UlyaÎncă nu există evaluări
- Extintor Tipo D WB-570 - BADGERDocument2 paginiExtintor Tipo D WB-570 - BADGERCoordinador TecnicoÎncă nu există evaluări
- CBSE Class 10 Science Revision Notes Chapter - 5 Periodic Classification of ElementsDocument4 paginiCBSE Class 10 Science Revision Notes Chapter - 5 Periodic Classification of ElementskunalÎncă nu există evaluări
- Limiting and Excess ReactantsDocument2 paginiLimiting and Excess Reactantsayvie1Încă nu există evaluări
- Standard Testing Method For Water Analysis For Reverse Osmosis and NanofiltrationDocument2 paginiStandard Testing Method For Water Analysis For Reverse Osmosis and NanofiltrationtalhaÎncă nu există evaluări