Documente Academic
Documente Profesional
Documente Cultură
JCPR 2012 4 9 4323 4333
Încărcat de
Eduardo Damian Verde LeonDescriere originală:
Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
JCPR 2012 4 9 4323 4333
Încărcat de
Eduardo Damian Verde LeonDrepturi de autor:
Formate disponibile
Available online www.jocpr.
com
Journal of Chemical and Pharmaceutical Research, 2012, 4(9):4323-4333
Research Article
ISSN : 0975-7384 CODEN(USA) : JCPRC5
Rational Synthesis Design of a Potent Anti-Diabetic Therapeutic 'Pioglitazone' Using Retrosynthetic Analysis
Chittaranjan Bhanja and *Satyaban Jena Department of Chemistry, Utkal University, Bhubaneswar-751004, Odisha, INDIA.
_____________________________________________________________________________________________ ABSTRACT Within the realm of medicinal chemistry, synthesis plays a pivotal role in any drug research and development endeavor. The ability to design elegant and economical synthetic routes is often a major factor for making a drug a commercial winner. Retrosynthetic analysis/ Synthon disconnection approach, introduced and advanced by Prof. E. J. Corey of Harvard University has served as a powerful driving force on design elegant and economical synthetic routes to any useful molecule of scientific or industrial interest. Exploiting the advantages of this approach, we have proposed a good number of synthesis schemes for a potent anti-diabetic drug Pioglitazone.from the results of its retrosynthetic analysis. The proposed synthesis planning being a theoretical explorations, the actual laboratory execution requires a cross examination of so many factors such as reactions, reagents and order of events. The routes that utilize readily available starting materials, convergent, economical, safe and produce maximum yield in short reaction time are most feasible. Keywords: Antidiabetic agent, drug synthesis, pioglitazone, retrosynthetic analysis, synthon disconnection approach. _____________________________________________________________________________________________ INTRODUCTION Chemical synthesis is one of the key technologies in modern drug discovery and development process. Development of novel synthetic routes for convergent and efficient synthesis of pharmaceuticals/drugs to make them suited for therapeutic use is very fundamental to synthetic organic/medicinal chemistry and gives material benefits to man kind. Before the execution, every synthesis is planned carefully with respect to sequence of steps from starting materials to final product. Although the sequence is quite logical but the details as to how the chemist first formulated the sequence of steps is not always published. A systematic approach in planning of synthesis is promulgated as a result of Prof.E.J.Coreys development of Retrosynthetic analysis/ Synthon disconnection approach in design organic synthesis. Retrosynthetic analysis is a problem solving technique in the planning of organic syntheses. This is achieved by transforming a target molecule into simpler precursor structures by the disconnection of strategy bonds and functional group transformations which leads to simple or commercially available starting materials [1-3].The retrosynthetic analysis of any target structure may lead to a number of possible synthetic routes. The routes which are convergent, utilise readily available starting materials, produce maximum yield in short reaction time, safe, eco-friendly are the most feasible. Diabetes mellitus is one of the worlds most prevalent, non-communicable, debilitating and progressive disease and becomes the 4th to 5th leading cause of death in developed countries [4].According to recently compiled data from Global Prevalence of Diabetes (GPD) and American Diabetes Association (ADA), diabetes currently affects more
4323
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
then 285 million people world wide, a figure that is expected to rise to 435 millions by 2030 due to population growth, ageing, unhealthy diets, obesity and sedentary lifestyles.[5,6] Diabetes mellitus is a group of metabolic disease characterized by alternations in the metabolism of glucose(carbohydrate), fat and protein which are caused by a relative or absolute deficiency of insulin secretion or insulin resistance or both.[7-9].Type -2 diabetes is the commonest form of diabetes and is characterized by disorders of insulin secretion and insulin resistance. It adversely affects the functioning of the kidneys, eyes, nervous and vascular systems. Therefore, once diagnosed, it is essential to control blood glucose levels during the early stages of the disease. The drugs/medicines used to treat diabetes mellitus are known as antidiabetic agents or oral hypoglycemic agents. The most attractive and direct approach to treat diabetes however will be administration of orally active hypoglycemic drugs. Members of the thiazolidinedione (TZD) drug class [10] are well known as anti-hyperglycemic drugs, used for the treatment of diabetes mellitus type2 [11] Pioglitazone (Fig 1), marketed as trademarks Actos is one of the potent drugs in this class. It selectively stimulates the nuclear receptor peroxisome proliferator-activated receptor gamma (PPAR-) in order to modulate the transcription of the insulin sensitive genes that are involved in glucose and lipid metabolism.[12,13]More recently, Pioglitazone and other active TZDs have been shown to bind to the outer mitochondrial membrane protein mitoNEET with affinity comparable to that of Pioglitazone for PPAR.[13,14] Pioglitazone has also been used to treat non-alcoholic steatohepatitis (fatty liver), but this use is presently considered experimental.[15] Pioglitazone has also been found to reduce the risk of conversion from prediabetes to diabetes mellitus type-2 by 72%.[16] O Et NH S N O O
Figure: I
Despite the availability of few synthetic approaches to Pioglitazone in literature, some alternative synthetic routs are still required for its commercial success. Keeping an overview on the published works both in journals [17-19] and patent literatures [20-21] herein, we wish to focus our research attentiveness by proposing a good number of synthesis schemes for a potent anti-diabetic drug Pioglitazone based on retrosynthetic analysis/ synthon disconnection approach. To our current knowledge, this type of work has not been reported elsewhwre.The choice of this molecule for synthesis planning is obvious as Pioglitazone is one of the most commonly prescribed medications as anti-diabetic agent. Again the pharmaceutical industries are unquestionably vibrant today in search of alternative, cost-effective, scale-up synthesis for potent anti-diabetic drugs for their commercial success. EXPERIMENTAL SECTION The structure and information about Pioglitazone as drug candid has been collected from different books [22-24]. The proposed synthesis planning are then exploited in a novel way from the result of the retrosynthetic analysis of the drug structure using the basic principle outlined in the pioneering works of Prof. E.J. Corey [25]. The symbols and abbreviations are synonymous to that represented in different books [25-30]. The analysissynthesis schemes being theoretical propositions; obviously the syntheses have not been executed in the laboratory. The actual laboratory execution requires the cross examination of a considerable number of factors such as reagents, reactions, order of events, economical viability, environmental benign, saftyness, short time and scalable synthesis. RESULTS AND DISCUSSION Condensation of ethyl chloroacetate 7 with thiourea 6 produces an intermediate 2-iminothiazolidone 4 which on hydrolysis gives 2,4-thiazolidinedione 3 (Hantzsch method).Reaction of 5-ethyl-2-methyl pyridine 10 with formaldehyde 11 at high temperature gives 2-(5-ethyl-2-pyridyl)ethanol 9.Alcohol 9 through its methane sulfonate derivative then condenses with 4-hydroxybenzaldehyde 8 in basic medium and affords 4-[2-(5-ethyl-pyridin-2yl)-ethoxy]-benzaldehyde 2. Reacting 4-[2-(5-ethyl-pyridin-2-yl)-ethoxy]-benzaldehyde 2 with 2, 4thiozolidinedione 3 provides 5-[4-[2-(5-ethyl-2-pyridyl)ethoxy]benzilidene]-2,4-thiazolidinedione 1.Reduction of 1 with Mg/MeOH forms the target molecule (T M) (Scheme-1).
4324
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
Retrosynthetic Analysis-1
O Et NH N TM Et + N 9 C-C Et N 10 + H2C=O 11 Cl 7 O OEt + H2 N S 6 NH2 O C-N Cl NH HS NH 5 C-S NH S NH 4 OH HO 8 N 2 O O S O H O C-O Et N O 1 C=C S O H O + S 3 O FGI O O NH FGA Et NH O
Synthesis-1
O O Cl 7 OEt + H2N S 6 NH2 NH S 4 NH
i. MeSO2Cl Et N 10 H2C=O 11 Heat Et N ii. HO 9 OH H O Et 8 NaOH CH2Cl2 N 2 O H
O Hydrolysis S 3 O
O NH S O 3 O Piperidine/AcOH
NH
O Et NH N O 1 S O
Scheme: 1
O Mg/MeOH Reduction Et NH N O S O (TM)
Retrosynthetic Analysis-2
O Et NH N O TM S O N O 1 S O FGA Et NH S 3 O O C-C NH + O
4325
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
H Et N O 2 O FGI Et N O 12 Br C-O HO 13 Br + N 9 OH Et
Synthesis-2
i.MeSO2Cl Br ii. Et HO 13 base OH O H Br i.CO,H 2 N O 12 Et N O 2 NH S O 3 O Piperidine
Et N 9
ii.(Cy3P)PdCl2
O Et NH N O 1 S O
Scheme: 2
O i NaBH4 Et NH (TM) N O S O
ii.CoCl2-DMG
Williamsons ether synthesis between methane sulfonate derivative of 2-(5-ethylpyridine-2-yl) ethanol 9 and 4bromophenol 13 in presence of a base produces 1-bromo-4-(2-(5-ethylpyridine-2-yl) ethoxy)benzene 12. Transition metal catalyzed hydroformylation of 12 produces 4-[2-(5-ethylpyridine-2-yl) ethoxy] benzaldehyde 2. Knoevenagel condensation of aldehyde 2 and thiazolidine 2,4-dione 3 produced as in scheme-1,affords 1.Reduction of the double bond of the condensed product 1 using sodium borohydride, cobalt(II) chloride-DMG affords the target molecule (TM) (Scheme-2). Retrosynthetic analysis-3
O Et NH N O TM
H Et N O 2 O FGI Et N 12 O Br FGI Et N 14 O NH2
O FGA Et NH N O 1 S O S C=C
O NH + 3 O
S O
FGI
Et N O 15
NO2
C-O F 16
NO2 +
Et N 9 OH
4326
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
Synthesis-3
NO2 Et N 9 OH F 16 NaH Et N O 15 NO2 H2,Pd-C Et N 14 O NH2
O H NaNO2/HCl CuBr, Et N O 12 Br i.CO,H2 ii.[Pd] Et N O 2 O NH S 3 O Piperidine
O Et NH N O 1 S O
Scheme: 3
O i. NaBH4 ii.CoCl2-DGM Et NH (TM) N O S O
Nucleophilic aromatic substitution reaction between 2-(5-ethylpyridine-2-yl)ethanol 9 and 4-fluoronitrobenzene 16 using NaH as base provides 1-nitro-4-[2-(5-ethylpyridine-2-yl)ethoxy] benzene 15.Redution of nitrogroup of 15 with H2/Pd-C affords corresponding amine 14.The amine through its diazonium salt forms corresponding bromoproduct 1-bromo-4-[2-(5-ethylpyridine-2-yl)ethoxy]benzene 12 via Sendmeyer reaction.The bromocompound then undergoes transition metal catalysed hydroformylation reaction to afford aldehyde 2. Condensation of aldehyde 2 with thiazolidine-2,4-dione 3 prepared as in scheme-1, in basic medium affords 5-[-4-[2-(5-ethylpyridine-2yl)ethoxy] benzylidene] thiazolidine-2,4-dione 1.Hydrogenation of dione with sodium borohydride in presence of a cobalt ion and dimethyl glyoxime affords the target molecule (TM )(Scheme-3). Retrosynthetic Analysis-4
O Et NH N O TM
O NH HO 18 S NH C-S C-N H2N
O C-O Et N + OH NH HO 17 S O FGI
S O
S 6 NH2 + HO 19
S
COOH Br
FGI HO 20
COOH NH2
Synthesis-4
O NH HO S 18 NH H/H2O COOH i.NaNO /HCl 2 HO 20 NH2 ii.CuBr, HO 19 Br COOH i. H2N 6 NH2 ii. NaOAc/EtOH
4327
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
O NH HO 17 S O Et i. 9 ArSO2Cl ii.NaOH/DCM N OH Et N
Scheme: 4
O NH (TM) O S O
Sandmayers reaction of tyrosine 20 through its diazonium salt forms the bromide derivative 19. Reflux of the bromide 19 with thiourea 6 and sod. acetate in ethanol produces 5-(4-hydroxybenzyl)-2-iminothiazolidine-4-one 18. The imine on hydrolysis forms 5-(4-hydroxybenzyl) thiazolidine -2,4-dione 17. The thiazolidine dione then undergoes nucleophilic substitution with p-toluene sulfonate intermediate formed in situ from 2-(5-ethyl-2-pyridyl) ethanol 9 and aryl sulfonyl chloride in presence of NaOH /DCM provides the target molecule (TM) (Scheme- 4). Retrosynthetic Analysis-5
O Et NH N TM
H E t N 2 FGI E t N 23 O CN C-O Et + N 9 OH F 24 CN O O +
O FGA Et NH N O 1
O C-N NCS 21 NH 4 C-S OEt Cl
C=C
S O
O
S O
O OEt 7 + NaSCN 22
FGI
NH S 3 O NH S
Synthesis-5
O Cl 7 OEt
NaSCN 22
O NCS 21 OEt cyclisation S 4
CN
O NH NH H+/H2O S 3
O NH O
O H NH S O N O 2 3 O Piperidine
Et N 9
F 24 NaH OH S Ar N
Et N O 23
CN
Ra-Ni H-COOH
Et
O Et NH N O 1 S O
Scheme: 5
O H2 /Pd-C dioxane Et N O S O NH (TM)
4328
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
Condensation of ethyl chloroacetate 7 with sodium thiocyanate 22 forms an intermediate 21 which on cyclisation and subsequent acid hydrolysis produces 2, 4-thiazolidinedione 3 (Techerniac process). Nucleophilic aromatic substitution reaction between 2-(5-ethylpyridine-2-yl) ethanol 9 and 4-fluorobenzonitrile 24 using NaH as base provides 4-[2-(5-ethylpyridine-2-yl)ethoxy] benzonitrile 23.Reduction of 23 with Raney-nickel and formic acid affords aldehyde 4-[2-(5-ethylpyridine-2-yl) ethoxy] benzaldehyde 2. Knoevenagel condensation of aldehyde 2 with thiazolidine-2, 4-dione 3 in basic medium affords 5-[-4-[2-(5-ethylpyridine-2-yl)ethoxy] benzylidene] thiazolidine2,4-dione 1.Catalytic hydrogenation of dione 1 provides target molecule(TM)(Scheme-5). Retrosynthetic Analysis-6
O E t N
S H2 N 6 NH2 + N O 26
O FGI Et NH N O 25
COOMe Br C-C O 27 OMe Et + N O 14 NH2
C-S C-N
NH O TM
Et
S O
S NH
FGI
Et N 15 O
NO2
C-O
Et N 9 + OH F 16
NO2
Synthesis-6
NO2 Et F 16 N 9 OH NaH N O 15 Et NO2 H2,Pd-C N O 14 Et NH2
S i.NaNO2,HBr Me2CO/MeOH OMe ii. 27 O Et N O 26 Br COOMe Et H2N 6 NH2 NaOAc N O 25 S
O NH NH
O HCl H2O Et N O S O
Scheme: 6
NH (TM)
The condensation of 2-(5-ethyl-2-pyridyl) ethanol 9 with 4-fluoronitrobenzene 16 in presence of NaH / DMF gives 4-[2-(5-ethyl-2-pyridyl)ethoxy]nitrobenzene 15. Reduction of 15 with H2 over Pd/C in methanol yields the corresponding aniline 14. The reaction of 14 with NaNO2/HBr and then with methyl acrylate 27 in acetone/methanol affords the 2-bromopropionate derivative 26. Cyclistion of 26 with thiourea 6 by means of NaOAc in refluxing ethanol provides the 2-imino-4-thiazolidinone 25.Acid hydrolysis of 25 produces the target molecule (TM) (Scheme-6).
4329
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
Retrosynthetic Analysis-7
O Et NH N O TM S O FGI Et NH N O 25 S NH O C-N C-S H2N S 6 NH2
Et + N O 28
CO2Et OMs
FGI
Et N O 29
CO2Et OH
FGI
Et O N O 30
Darzene CO2Et glycidicester Cl disconnection 7 O
H OEt Et + N O 2 O C-O
H Et + N 9
Synthesis-7
i. MsCl H Et N 9 ii. HO 8 OH Et3N O Et N O 2 H Cl O 7 O N O 30 OEt Et O CO2Et
O HO 8
OH
S H2,Pd-C Et N O 29 CO2Et OH MsCl Et3N Et N O 28 CO2Et OMs H2N 6 NH2 NaOAc,EtOH
O Et NH N O 25 S NH
Scheme-7
O HCl H2O Et N O S O NH (TM)
Williamsons ether synthesis between methanesulfonate derivative of 2-(5-ethylpyridine-2-yl) ethanol 9 and 4hydroxybenzaldehyde 8 in presence of a base produces 4-[2-(5-ethylpyridine-2-yl)ethoxy] benzaldehyde 2. Darzene glycidicester condensation of aldehyde 2 with ethyl chloroacetate 7 and NaOEt/EtOH at room temperature yields cis-trans glycidic ester 30. Hydrogenolysis of the mixture using H2 in Pd-C/MeOH 28 and subsequent methylsulfonylation of the resulting alcohol 29 affords -methanesulfonyloxy ester 28.Reaction of ester with
4330
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
thiourea 6 and NaOEt (Hantzschs synthesis) generates 2-imino-4- thiazolidinone 25, which on hydrolysis with dil.HCl forms target molecule( TM) (Scheme-7). Retrosynthetic Analysis-8
O Et NH N O TM
O Et NH N OH O 32
H Et N OH O 34 O C-O HO 8 H O Et + N 35 O N 36 HO B r Et
O FGA Et NH N O Cl
Et NH N O OH 33 S O 3
FGI S O 31
O O C=C S O NH
S O
FGA S O
Et N 37
Synthesis-8
H i. Et N 37 NBS in aq.BuOH Et N 36 HO K2CO3 Br Et N 35 HO 8 ii. K2CO3/DMF O O Et N O OH 34 H O
O i. S 3 O ii. Py/MeOH NH Et N OH O 33 S O i. NaBH4 NH ii. CoCl2-DMG O Et N OH O 32 S O NH SOCl2 DCM O
O Et NH N Cl 31 O S O Zn-EtOH CH3COOH reduction Et N O S
O NH (TM) O
Scheme-8
4331
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
Reaction of 5-Ethyl-2-vinyl pyridine 37 with NBS in aq.t-BuOH affords bromohydrine 36.The bromohydrine on reaction with K2CO3 forms the oxirane 35.The oxirane then involves in displacement reaction with the pot.salt of 4hydroxy benzaldehyde 8 in a Williamson ether synthesis process to afford 4-[2(-5-ethylpyridin-2-yl)2hydroxyethoxy] benzaldehyde 34. Knovenagel condensation of aldehyde 34 with thiaolidine-2,4-dione 3 using pyrollidine/ethanol produces 5-(4-(2-(-5-ethylpyridine-2-yl)-2-hydroxyethoxy benzylidine) thiazolidine-2,4-dione 33.Reduction of double bond of thiazolidinone35 using NaBH4/CoCl2-DMG affords 32.Tratment of 32 with SOCl2 /DMF affords corresponding chloride 31.Reduction of 31 with Zn in MeOH-AOH affords target molecule( TM) (Scheme-8). CONCLUSION Retrosynthetic analysis is one of the most critical tools within the toolbox needed to solve synthesis problems. The goal of retrosynthetic analysis is the structural simplification. It not only requires logical approach for disconnecting a complex target molecule but also a through knowledge of an enormous set of organic reactions to imagine the experimental conditions necessary to produce a desired product from simple or readily available starting materials. It is a paper exercise; a full exploration of this type will provide many routes for synthesizing the target molecule. As a consequence of this approach, we have proposed a good number of synthesis schemes for a potent anti-diabetic drug Pioglitazone. Scalable synthetic routes for newly discovered natural products, pharmaceuticals and useful compounds not available in adequate quantities from natural resources can be best provide by this approach. Strategic application of this approach can also afford different routes to synthesize the target molecule that has never been synthesized earlier. With the advancement of novel methods and transformations developed within the academic community, the synthesis of best selling pharmaceuticals can be rethinking through this approach in the later stages of drug research and process development. Acknowledgements The author CB acknowledges UGC, ERO, Kolkata, India for financial support as Teacher Fellow Grants. The author also thanks the authorities of IIT Bhubaneswar, IMMT Bhubaneswar and NISER Bhubaneswar for permission to collect information from books and journals from their library. REFERENCES [1] E.J. Corey. Pure. Appl. Chem. 1967, 14 (1) 19-38. [2] E.J. Corey. W.T.Wipke. Science. 1969,166, 178-192. [3] E. J. Corey. Q. Rev .Chem. Soc. 1971, 25, 455-482. [4] A. Amos. D. McCarty.; P. Zimmet. Diabetic Med. 1987, 14, S1S85. [5] S. Wild.; G.Roglic.; A Green. Diabetes Care. 2004, 27(5), 1047-1053. [6] American Diabetes Association. Diagnosis and classification of diabetes Mellitus. Diabetes Care. 2005,28 (Suppl 1),S37-S42. [7] P.J. Kumar.; M. Clark. Textbook of Clinical Medicine. Saunders (London), pp 1099-1121, 2002. [8] Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of Diabetes Mellitus.Diabetes Care. 1997, 20, 1183-1197. [9] B. Beverley.; E. Eschwge. Textbook of Diabetes. (1/e) John C Pickup and Gareth Williams ,(3/e) Chapter 2, pp 2.1-2.11, 2003. [10]B.C.Centello.; M.A.Cawthorne.; G.P. Cottam.; P.T. Duff.; D. Haigh.; R.M. Hindley.;C.A. Lister.; S.A Smith.; P.L Thurlby. J. Med. Chem. 1994, 37,3977-3985. [11] B.C.Centello.; M.A.Cawthorne.; D. Haigh.; R.M. Hindley.; C.A. Lister.; S.A Smith.;P.L Thurlby.Bioorg. Med. Chem .Lett. 1994, 4, 1181-1184. [12] U. Smith. Int J Clin Pract Suppl . 2001,121, 138. PMID 11594239. [13] J.R. Colca.; W.G. McDonald.; D.J. Waldon.; J.W. Leone.; J.M. Lull.; C.A. Bannow.;E.T. Lund.; W.R. Mathews. Am. J. Physiol. Endocrinol.Metab. 2004,286 (2), E2,52- 60. [14] M.L. Paddock.; S.E. Wiley.; H.L. Axelrod.; A.E. Cohen.; M. Roy.; E.C. Abresch.;D. Capraro.; A.N. Murphy. Proc. Natl. Acad. Sci. U.S.A. 2007,104 (36) 1434214347. [15] R. Belfort.; S.A. Harrison.; K. Brown.; C. Darland.; J .Finch.; J. Hardies.; B. Balas.;Gastaldelli. N. Engl. J. Med. 2006,355 (22), 2297307. [16] DeFronzo., A. Ralph. N Eng J Med 2011, 364, 1104-1115. [17] J.Prous.;J.Castaner. Drugs Fut 1990, 15, 11, 1080.
4332
Chittaranjan Bhanja et al J. Chem. Pharm. Res., 2012, 4(9):4323-4333 ______________________________________________________________________________
[18] K.Meguro.; Y. Momose.; T.Sohda.; S.Oi.; H.Ikeda.; C. Hatanaka. Chem Pharm Bull .1991, 39( 6), 1440-1445. [20 ] K.Meguro.; T.Fujita.; C.Hatanaka; S. Oi.U.S.Patent 4,812,570,1989. [21 ] J.E. Huber,.U.S. Patent 5.585,495,1996. 1986, 1987. [22] Foys, W. O.; Lemke, T. L.; Williams, D. A. Foyes Principles of Medicinal Chemistry. 2007,pp-627. [23] Brittain, H. G. Analytical Profiles of Drug substances and Excipients. Academic Press, 1996,pp-443. [24] Ledincer, D.; Mitscher, L.A. The Organic Chemistry of Drug synthesis. Vol-4, 1990,pp-57. [25] Corey E.J.; Chang X.M. The Logic of Chemical Synthesis. Wiley, New York, 1989. [26] Warren S. Designing Organic Synthesis: A Programme Introduction to Synthon Approach. John Wiley and Sons, New York, 1978. [27] Warren S. Organic Synthesis-The Disconnection Approach. John Wiley and Sons,New York,1982. [28]Clayden, J.; Greeves, N.; Warren, S.; Wothers, P. Retrosynthetic Analysis In Organic Chemistry. Oxford University Press Inc., New York, 2001, pp. 773-778. [29] Fuhrhop, J-H., Li, G. Organic Synthesis: Concepts and Methods.Wiley-VC GmbH and Co.KGaA, 2003. [30] Starkey, L.S. Introduction to Strategies for Organic Synthesis. John Wiley and Sons Inc, New Jercy, 2012.
4333
S-ar putea să vă placă și
- Peptide and Protein DeliveryDe la EverandPeptide and Protein DeliveryChris Van Der WalleEvaluare: 2 din 5 stele2/5 (1)
- Pharm Cvs ProdrugsDocument23 paginiPharm Cvs ProdrugsSindu SaiÎncă nu există evaluări
- Kumarsingh 2017Document15 paginiKumarsingh 2017Quoc HuyÎncă nu există evaluări
- Molecular Docking Studies of Quinoline3carbohydrazide As Novel Ptp1b Inhibitors As Potential Antihyperglycemic AgentsDocument11 paginiMolecular Docking Studies of Quinoline3carbohydrazide As Novel Ptp1b Inhibitors As Potential Antihyperglycemic Agentssunaina agarwalÎncă nu există evaluări
- Sambiloto PDFDocument7 paginiSambiloto PDFMarselina SattuÎncă nu există evaluări
- Lipid-Modifying Effects of NutraceuticalsDocument14 paginiLipid-Modifying Effects of NutraceuticalsBlanca MezaÎncă nu există evaluări
- Protein and Peptide Biopharmaceuticals: An OverviewDocument8 paginiProtein and Peptide Biopharmaceuticals: An OverviewSam SonÎncă nu există evaluări
- Molecular Docking Studies of Shc1 Protein: A Drug Target To Treat ObesityDocument6 paginiMolecular Docking Studies of Shc1 Protein: A Drug Target To Treat ObesityPrasanna BabuÎncă nu există evaluări
- Plant-Based Antidiabetic Nanoformulations: The Emerging Paradigm For EDocument44 paginiPlant-Based Antidiabetic Nanoformulations: The Emerging Paradigm For EAlyna AlynaÎncă nu există evaluări
- Bio-Flavonoids With Promising Anti-Diabetic Potentials: A Critical SurveyDocument26 paginiBio-Flavonoids With Promising Anti-Diabetic Potentials: A Critical SurveyKris Natalia ManihurukÎncă nu există evaluări
- Probiotics For The Management of Type 2 Diabetes MellitusDocument11 paginiProbiotics For The Management of Type 2 Diabetes MellitusKelly BarrosÎncă nu există evaluări
- Leucocephala and Trigonella Foenum-Graecum Chemical Constituents OnDocument11 paginiLeucocephala and Trigonella Foenum-Graecum Chemical Constituents OnPuspaÎncă nu există evaluări
- Biomedicine & Pharmacotherapy: SciencedirectDocument7 paginiBiomedicine & Pharmacotherapy: SciencedirectMirja AdiyaksaÎncă nu există evaluări
- Proposal 14.08.2023Document6 paginiProposal 14.08.2023gopiÎncă nu există evaluări
- Pharmaceutics 03 00538Document34 paginiPharmaceutics 03 00538Mia karolinaÎncă nu există evaluări
- Colloids and Surfaces A: Physicochemical and Engineering AspectsDocument12 paginiColloids and Surfaces A: Physicochemical and Engineering Aspectsjoyhar9Încă nu există evaluări
- Molecules 23 01519 With CoverDocument31 paginiMolecules 23 01519 With Coverroony rooÎncă nu există evaluări
- Caplan 2017Document9 paginiCaplan 2017Zuldan Karami100% (1)
- Don 2018Document16 paginiDon 2018Putri Pah Kumala DewiÎncă nu există evaluări
- Article Wjpps 1467683350 PDFDocument13 paginiArticle Wjpps 1467683350 PDFNur FitriÎncă nu există evaluări
- E3 FullDocument2 paginiE3 FullafandianddonkeyÎncă nu există evaluări
- Ni Hms 604442Document26 paginiNi Hms 604442Gaby ValenzuelaÎncă nu există evaluări
- A Review On Estimation of Metformin Hydrochloride and Vildagliptin in Pharmacutical Dosage FormDocument6 paginiA Review On Estimation of Metformin Hydrochloride and Vildagliptin in Pharmacutical Dosage FormEditor IJTSRDÎncă nu există evaluări
- PHD Thesis Drug Delivery SystemDocument4 paginiPHD Thesis Drug Delivery Systemgj9zvt51100% (2)
- Article (2006) - Isoniazid Biography (Subrayado)Document10 paginiArticle (2006) - Isoniazid Biography (Subrayado)pixoguiasÎncă nu există evaluări
- Pharmaceutics: Basic Principles and Application to Pharmacy PracticeDe la EverandPharmaceutics: Basic Principles and Application to Pharmacy PracticeAlekha DashÎncă nu există evaluări
- Commentary Biowaiver Monographs For Immediate Release Solid Oral Dosage Forms: PrednisoloneDocument20 paginiCommentary Biowaiver Monographs For Immediate Release Solid Oral Dosage Forms: PrednisolonePrisca WicitaÎncă nu există evaluări
- Handbook of Polymers for Pharmaceutical Technologies, Structure and ChemistryDe la EverandHandbook of Polymers for Pharmaceutical Technologies, Structure and ChemistryÎncă nu există evaluări
- Tan, 2022Document8 paginiTan, 2022Mirilláiny AnacletoÎncă nu există evaluări
- Translational Glycobiology in Human Health and DiseaseDe la EverandTranslational Glycobiology in Human Health and DiseaseMichelle KilcoyneÎncă nu există evaluări
- Metabolic Syndrome: Underlying Mechanisms and Drug TherapiesDe la EverandMetabolic Syndrome: Underlying Mechanisms and Drug TherapiesMinghan WangÎncă nu există evaluări
- Nps 2 EE1Document16 paginiNps 2 EE1glreddy09Încă nu există evaluări
- Introduction to Biological and Small Molecule Drug Research and Development: Theory and Case StudiesDe la EverandIntroduction to Biological and Small Molecule Drug Research and Development: Theory and Case StudiesC. Robin GanellinEvaluare: 5 din 5 stele5/5 (1)
- (Group 3) Asparagus Officinalis - 141204Document23 pagini(Group 3) Asparagus Officinalis - 141204Tran Minh NhutÎncă nu există evaluări
- Pharmaceutical Application of Nanoparticles in Drug Delivery SystemDocument10 paginiPharmaceutical Application of Nanoparticles in Drug Delivery SystemKumar GalipellyÎncă nu există evaluări
- Current Treatments For Type 2 Diabetes, Their Side Effects and Possible Complementary TreatmentsDocument13 paginiCurrent Treatments For Type 2 Diabetes, Their Side Effects and Possible Complementary TreatmentsBianca GhibaÎncă nu există evaluări
- Pomegranate Juice HealingDocument23 paginiPomegranate Juice HealingKingÎncă nu există evaluări
- Cyclodextrin Drug Carrier SystemsDocument32 paginiCyclodextrin Drug Carrier SystemsVasudevan SubramaniyanÎncă nu există evaluări
- Mitochondria in Obesity and Type 2 Diabetes: Comprehensive Review on Mitochondrial Functioning and Involvement in Metabolic DiseasesDe la EverandMitochondria in Obesity and Type 2 Diabetes: Comprehensive Review on Mitochondrial Functioning and Involvement in Metabolic DiseasesBeatrice MorioÎncă nu există evaluări
- Biowaiver Monographs For Immediate Release Solid Oral DosageDocument10 paginiBiowaiver Monographs For Immediate Release Solid Oral DosageAgus StyawanÎncă nu există evaluări
- Synthesis and Evaluation of Some Amino Acid Conjugates of NSAIDSDocument9 paginiSynthesis and Evaluation of Some Amino Acid Conjugates of NSAIDSSACHIN BHASKAR NARKHEDEÎncă nu există evaluări
- Review Article: Pharmacogenomics of Drug Response in Type 2 Diabetes: Toward The Definition of Tailored Therapies?Document11 paginiReview Article: Pharmacogenomics of Drug Response in Type 2 Diabetes: Toward The Definition of Tailored Therapies?James CowellÎncă nu există evaluări
- The Human Gut-Liver-Axis in Health and DiseaseDe la EverandThe Human Gut-Liver-Axis in Health and DiseaseAleksander KragÎncă nu există evaluări
- A Nutraceutical Combination Improves Insulin Sensitivity in Patients With Metabolic SyndromeDocument7 paginiA Nutraceutical Combination Improves Insulin Sensitivity in Patients With Metabolic SyndromegiannidietÎncă nu există evaluări
- (216 235) V10N5CTDocument20 pagini(216 235) V10N5CTgritty gallantÎncă nu există evaluări
- Assessment of Antidiabetic Effect of 4-HIL in Type 2 Diabetic and Healthy Sprague Dawley RatsDocument11 paginiAssessment of Antidiabetic Effect of 4-HIL in Type 2 Diabetic and Healthy Sprague Dawley RatsMaxin BangsalÎncă nu există evaluări
- An Investigation of Antidiabetic ActivitiesDocument15 paginiAn Investigation of Antidiabetic Activitiesminhnhut_lxagÎncă nu există evaluări
- An Explicit Review On Quantitative Estimation of Candesartan Cilexetil Employing Various Analytical Techniques 2153 2435.1000254Document7 paginiAn Explicit Review On Quantitative Estimation of Candesartan Cilexetil Employing Various Analytical Techniques 2153 2435.1000254DrSrujan Kumar ReddyÎncă nu există evaluări
- Glycolysis: Tissue-Specific Metabolic Regulation in Physio-pathological ConditionsDe la EverandGlycolysis: Tissue-Specific Metabolic Regulation in Physio-pathological ConditionsRita FerreiraÎncă nu există evaluări
- Pharmacoepidemiology and Pharmacovigilance: Synergistic Tools to Better Investigate Drug SafetyDe la EverandPharmacoepidemiology and Pharmacovigilance: Synergistic Tools to Better Investigate Drug SafetyEvaluare: 4.5 din 5 stele4.5/5 (3)
- Molecules: Synthesis of 4-Methoxybenzoylhydrazones and Evaluation of Their Antiglycation ActivityDocument16 paginiMolecules: Synthesis of 4-Methoxybenzoylhydrazones and Evaluation of Their Antiglycation Activityzael18Încă nu există evaluări
- Final Report Amit NayakDocument48 paginiFinal Report Amit NayakAmit NayakÎncă nu există evaluări
- 2023 Nano Pharmaceuticals ArtigoDocument16 pagini2023 Nano Pharmaceuticals ArtigoLuís Rodrigues da SilvaÎncă nu există evaluări
- Journal Pre-Proof: E, Maria Kristina ParrDocument21 paginiJournal Pre-Proof: E, Maria Kristina ParrAlonso FernandezÎncă nu există evaluări
- Design and Development of Controlled Porosity Osmotic Pump Tablets of Zidovudine Using Sodium Chloride As Osmogen For The Treatment of Aids JPR 19Document8 paginiDesign and Development of Controlled Porosity Osmotic Pump Tablets of Zidovudine Using Sodium Chloride As Osmogen For The Treatment of Aids JPR 19gritty gallantÎncă nu există evaluări
- Retrometabolic Drug DesignDocument7 paginiRetrometabolic Drug Designolivia523Încă nu există evaluări
- Bioorganic & Medicinal ChemistryDocument14 paginiBioorganic & Medicinal ChemistryAulia Fatmiyatun El-FalesyÎncă nu există evaluări
- Farmakologi Benincasa Hispida (Kundur)Document11 paginiFarmakologi Benincasa Hispida (Kundur)Syahrir ManaanÎncă nu există evaluări
- Nama: Yossi Tiara Pratiwi Kelas: X Mis 1 Mata Pelajaran: Bahasa InggrisDocument2 paginiNama: Yossi Tiara Pratiwi Kelas: X Mis 1 Mata Pelajaran: Bahasa InggrisOrionj jrÎncă nu există evaluări
- Fss Presentation Slide GoDocument13 paginiFss Presentation Slide GoReinoso GreiskaÎncă nu există evaluări
- 7400 IC SeriesDocument16 pagini7400 IC SeriesRaj ZalariaÎncă nu există evaluări
- Regions of Alaska PresentationDocument15 paginiRegions of Alaska Presentationapi-260890532Încă nu există evaluări
- Sam Media Recruitment QuestionnaireDocument17 paginiSam Media Recruitment Questionnairechek taiÎncă nu există evaluări
- TIMO Final 2020-2021 P3Document5 paginiTIMO Final 2020-2021 P3An Nguyen100% (2)
- IKEA SHANGHAI Case StudyDocument5 paginiIKEA SHANGHAI Case StudyXimo NetteÎncă nu există evaluări
- Carob-Tree As CO2 Sink in The Carbon MarketDocument5 paginiCarob-Tree As CO2 Sink in The Carbon MarketFayssal KartobiÎncă nu există evaluări
- Report Card Grade 1 2Document3 paginiReport Card Grade 1 2Mely DelacruzÎncă nu există evaluări
- Making Effective Powerpoint Presentations: October 2014Document18 paginiMaking Effective Powerpoint Presentations: October 2014Mariam TchkoidzeÎncă nu există evaluări
- Richardson Heidegger PDFDocument18 paginiRichardson Heidegger PDFweltfremdheitÎncă nu există evaluări
- Methods of Recording Retruded Contact Position in Dentate PatientsDocument15 paginiMethods of Recording Retruded Contact Position in Dentate PatientsYossr MokhtarÎncă nu există evaluări
- MGMT Audit Report WritingDocument28 paginiMGMT Audit Report WritingAndrei IulianÎncă nu există evaluări
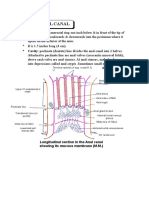
- Anatomy Anal CanalDocument14 paginiAnatomy Anal CanalBela Ronaldoe100% (1)
- Existential ThreatsDocument6 paginiExistential Threatslolab_4Încă nu există evaluări
- Human EpigenomicsDocument234 paginiHuman EpigenomicsHeron HilárioÎncă nu există evaluări
- Parliament of India: Rajya SabhaDocument64 paginiParliament of India: Rajya SabhaSivapothuraju KonathalaÎncă nu există evaluări
- Retailing in IndiaDocument11 paginiRetailing in IndiaVinod MalkarÎncă nu există evaluări
- Law of EvidenceDocument14 paginiLaw of EvidenceIsha ChavanÎncă nu există evaluări
- An Exploration of The Ethno-Medicinal Practices Among Traditional Healers in Southwest Cebu, PhilippinesDocument7 paginiAn Exploration of The Ethno-Medicinal Practices Among Traditional Healers in Southwest Cebu, PhilippinesleecubongÎncă nu există evaluări
- Impact of Advertising On Consumers' Buying Behavior Through Persuasiveness, Brand Image, and Celebrity EndorsementDocument10 paginiImpact of Advertising On Consumers' Buying Behavior Through Persuasiveness, Brand Image, and Celebrity Endorsementvikram singhÎncă nu există evaluări
- Companyprofil E: Erfanconstructionsolut IonDocument14 paginiCompanyprofil E: Erfanconstructionsolut IonNurin AleesyaÎncă nu există evaluări
- Hole CapacityDocument2 paginiHole CapacityAbdul Hameed OmarÎncă nu există evaluări
- Module 1: Overview of Applied Behaviour Analysis (ABA)Document37 paginiModule 1: Overview of Applied Behaviour Analysis (ABA)PriyaÎncă nu există evaluări
- Science7 - q1 - Mod3 - Distinguishing Mixtures From Substances - v5Document25 paginiScience7 - q1 - Mod3 - Distinguishing Mixtures From Substances - v5Bella BalendresÎncă nu există evaluări
- Nutrition and CKDDocument20 paginiNutrition and CKDElisa SalakayÎncă nu există evaluări
- Fusion Implementing Offerings Using Functional Setup Manager PDFDocument51 paginiFusion Implementing Offerings Using Functional Setup Manager PDFSrinivasa Rao Asuru0% (1)
- Presentation 11Document14 paginiPresentation 11stellabrown535Încă nu există evaluări
- Organization and Management Module 3: Quarter 1 - Week 3Document15 paginiOrganization and Management Module 3: Quarter 1 - Week 3juvelyn luegoÎncă nu există evaluări
- CH 1 - Democracy and American PoliticsDocument9 paginiCH 1 - Democracy and American PoliticsAndrew Philip ClarkÎncă nu există evaluări