Documente Academic
Documente Profesional
Documente Cultură
Acne Vulgaris Pathogenesis and Diagnosis
Încărcat de
Fitrianto Anwar0 evaluări0% au considerat acest document util (0 voturi)
131 vizualizări5 paginiThe many expressi ons of acne rarel y present a di agnostic chal l enge. Correct cl assi fi cati on of acne I s crucial al I n choosi ng the appropriate therapies. Acne is a complex disease with multifactorial pathogenesis and variation in severity.
Descriere originală:
Drepturi de autor
© © All Rights Reserved
Formate disponibile
PDF, TXT sau citiți online pe Scribd
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentThe many expressi ons of acne rarel y present a di agnostic chal l enge. Correct cl assi fi cati on of acne I s crucial al I n choosi ng the appropriate therapies. Acne is a complex disease with multifactorial pathogenesis and variation in severity.
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PDF, TXT sau citiți online pe Scribd
0 evaluări0% au considerat acest document util (0 voturi)
131 vizualizări5 paginiAcne Vulgaris Pathogenesis and Diagnosis
Încărcat de
Fitrianto AnwarThe many expressi ons of acne rarel y present a di agnostic chal l enge. Correct cl assi fi cati on of acne I s crucial al I n choosi ng the appropriate therapies. Acne is a complex disease with multifactorial pathogenesis and variation in severity.
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PDF, TXT sau citiți online pe Scribd
Sunteți pe pagina 1din 5
VOLUME 70, AUGUST 2002 101
Continuing Medical Education
Acne vul gari s i s a chroni c i nfl ammatory di sease
of the pi l osebaceous uni ts. It i s a pl eomorphi c
di sorder wi th mul ti factori al pathogenesi s. The
many expressi ons of acne rarel y present a di ag-
nosti c chal l enge, but correct cl assi fi cati on of
acne i s cruci al i n choosi ng the appropri ate ther-
api es. Al though previ ous research has provi ded
a better understandi ng of the pathogeni c fac-
tors, there i s sti l l a great deal to be l earned.
Perspective
Acne vulgaris, the most common skin disorder, affects
virtually all individuals at least once. Incidence peaks
in 18-year-olds, but substantial numbers of 20- to
40-year-olds also develop the disease. The effects of
acne should not be underestimated. It can persist for
years; produce disfigurement and permanent scarring;
and have significant psychosocial consequences,
including diminished self-esteem, embarrassment,
social withdrawal, depression, and unemployment.
1
The extent and severity of these effects underline the
importance of providing adequate therapy, which
produces satisfactory results in most cases.
Pathogenesis
Acne is a complex disease with multifactorial patho-
genesis and considerable variation in severity. Thus,
Acne Vulgaris, I:
Pathogenesis and Diagnosis
Steve S. Oberemok, MD; Alan R. Shalita, MD
From the Department of Dermatology, State University of
New York Downstate Medical Center, Brooklyn. Dr. Oberemok is a
Clinical Assistant Instructor. Dr. Shalita is a Distinguished Teaching
Professor and Chairman.
Reprints: Alan R. Shalita, MD, Department of Dermatology, SUNY
Downstate Medical Center, 450 Clarkson Ave, Brooklyn, NY 11203
(e-mail: ashalita@downstate.edu).
GOAL
To describe the pathogenic mechanisms associated with acne and
the variable clinical presentation
OBJECTIVES
Upon completion of this activity, dermatologists and general practitioners should be able to:
1. Discuss the morphologic changes that give rise to acne lesions.
2. Outline the role of sebum and Propionibacterium acnes in the production of acne.
3. Describe the clinical presentation, range of lesion severity, and differential diagnosis for acne.
CME Test on page 97.
This article has been peer reviewed and
approved by Michael Fisher, MD, Professor of
Medicine, Albert Einstein College of Medicine.
Review date: July 2002.
This activity has been planned and implemented
in accordance with the Essential Areas and Policies
of the Accreditation Council for Continuing Medical
Education through the joint sponsorship of Albert
Einstein College of Medicine and Quadrant
HealthCom, Inc. The Albert Einstein College of
Medicine is accredited by the ACCME to provide
continuing medical education for physicians.
Albert Einstein College of Medicine designates
this educational activity for a maximum of 1.0 hour
in category 1 credit toward the AMA Physicians
Recognition Award. Each physician should claim
only those hours of credit that he/she actually spent
in the educational activity.
This activity has been planned and produced in
accordance with ACCME Essentials.
Acne Vulgaris, I: Pathogenesis and Diagnosis
102 CUTIS
therapy can be directed at multiple factors and mod-
ified for individual patients.
Acne is a disorder of the sebaceous follicles,
which are special pilosebaceous units located on the
face, neck, chest, upper back, and upper arms. These
units consist of relatively large sebaceous glands
associated with small hair follicles. Acne arises from
the interaction of 4 factors:
1. Comedogenesissebaceous follicle obstruc-
tion arising from increased cohesiveness of
follicular epithelial cells, hyperproliferation
of ductal keratinocytes, or both.
2
2. Excessive sebum production caused by andro-
genic stimulation of sebaceous glands at or
around adrenarche or later.
3. Proliferation of Propionibacterium acnes, an
anaerobic diphtheroid that populates seba-
ceous follicles and is a normal constituent of
cutaneous flora. P acnes produces chemotac-
tic factors and proinflammatory mediators
that may lead to inflammation.
3
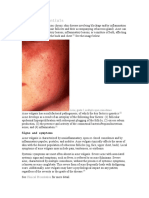
4. Inflammation is a direct or indirect result of
P acnes proliferation. Follicular rupture and
extension of inflammation into the dermis
result in formation of the inflammatory
lesions of acne vulgarispapules, pustules,
and nodules (Figure).
The earliest morphologic change or primary
pathologic event is sebaceous follicle obstruction
giving rise to microcomedones, precursors of all acne
lesions. The mechanism of microcomedo formation
is unclear, but reasonable evidence supports hyper-
proliferation of ductal keratinocytes.
4
This mecha-
nism was identified immunohistochemically using a
monoclonal antibody to Ki-67, a nuclear marker
expressed by actively cycling cells. In subjects with
acne but not in normal control subjects, Ki-67
showed increased labeling by basal keratinocytes of
the follicular epithelium of both comedones and
microcomedones. In addition, suprabasal expression
of keratin K16, a marker of hyperproliferation and
abnormal differentiation, occurs in ductal keratino-
cytes of acne lesions.
Some normal follicles of acne-prone skin also
may show overexpression of Ki-67 and K16
5
which underscores the need for topical treatment of
all acne-prone skin that shows early clear evidence
of microcomedo formation. Increased keratinocyte
cohesion as a primary event has been questioned as
a factor in comedogenesis, as there is no clear
evidence of abnormality of ductal desmosomes.
Some evidence, however, shows that the com-
position of sebaceous lipids becomes altered and
that such alteration may cause follicular cells to
adhere to one another more. Increases in free fatty
Abnormal follicular epithelial desquamation
Microcomedo formation
Excessive sebum production
Mixture of cells and sebum provides environment
for Propionibacterium acnes
Noninflammatory
Open and closed comedones
(blackheads and
whiteheads)
Chemotaxis
Cytokines
Inflammatory lesions
papules, pustules, nodules
Propionibacterium
acnes
Pathogenesis of acne.
Acne Vulgaris, I: Pathogenesis and Diagnosis
VOLUME 70, AUGUST 2002 103
acids,
6
squalene, and squalene oxide
7
and a
decrease in sebaceous linoleic acid could all trigger
abnormal cohesion of cells in sebaceous follicles.
According to another hypothesis, hyperkeratosis in
sebaceous follicles is the result of a local deficiency
of vitamin A.
8
Androgens may have an important
role in controlling ductal hyperproliferation either
directly or indirectly through stimulation of seba-
ceous glands. More recently, cytokines proved to
have an important controlling effect.
9
In the in vitro
model used in the study, comedones were produced
under the influence of interleukin-1 (IL-1), and
this process can be inhibited by adding IL-1
receptor antagonist to the system.
Sebum, the lipid-rich secretion of sebaceous
glands, has a central role in the pathogenesis of
acne and provides a growth medium for P acnes.
Enlargement of sebaceous glands and increased
production of sebum are stimulated by increased
production of adrenal and gonadal androgensa
change preceding clinical onset of puberty. Inter-
estingly, sebum production rates are inversely pro-
portional to the concentration of linoleic acid in
skin-surface lipids; subjects with acne and normal
control subjects are marked by a difference in
sebum composition. According to a current expla-
nation of acne pathogenesis, follicular epithelium
differentiation may be influenced by a lower con-
centration of linoleate in sebum. The relation
between acne and sebum overproduction has long
been acknowledged, as has been the correlation
between acne severity and sebum production rates.
Androgenic hormones control sebaceous gland
secretions. Testosterone is the main circulating
androgen. In women, however, the adrenal gland
and its main androgens dehydroepiandrosterone
(DHEA) and sulfate salt of DHEA (DHEAS) have
important roles in androgen control of sebaceous
glands. Testosterone is assumed to convert to
dihydrotestosterone, which then binds to a high-
affinity specific cytoplasmic receptor protein that is
transported to the cell nucleus where the DNA-
driven events occur.
Even though free testosterone levels may be ele-
vated in women with acne compared with women
without acne, actual values fall within the normal
range.
10
Given the efficacy of antiandrogen treat-
ments, however, the sebaceous glands of patients
with acne are likely to be hypersensitive to andro-
gens. In men, the association of acne and high
androgen concentrations is less consistent.
11
To be
sure, there are no reports of conclusive repro-
ducible studies supporting a consistent mechanism
involved in sebum overproduction as it relates to
androgen production.
P acnes is the predominant organism in seba-
ceous follicles, where it grows in a relatively anaer-
obic lipid-rich environment of microcomedones.
P acnes counts on the skin of teenage subjects with
acne and P acnes counts on the skin of age-matched
control subjects differ significantly. P acnes, how-
ever, is not essential for comedogenesis, as was con-
firmed by research involving children with early
acne. P acnes produces an extracellular lipase that
hydrolyzes sebum triglycerides to glycerol, used by
the bacteria as a growth substrate, and to free fatty
acids, which may possess comedogenic and/or
proinflammatory qualities.
12
Inhibition of bacterial
lipase alone, however, failed to improve inflamma-
tory acne.
13
The initial event in acne inflammation
may be the disruption of follicular epithelium,
which allows microcomedones to come into con-
tact with inflammatory systems. Early, apparently
intact comedones contain neutrophils, which sug-
gests that soluble inflammatory factors may diffuse
from comedones. Lee et al
14
found that P acnes also
produced high-molecular-weight chemotactic fac-
tors, one of which was the lipase itself. In subse-
quent studies, P acnes may have simultaneously
produced both high- and low-molecular-weight
chemoattractants, and the majority of neutrophil
chemotactic activity in P acnes culture supernatant
was less than 2 kd.
15
In another study, the role of
cellular immunity in initiation of acne lesions was
explored.
16
Not surprisingly, CD4
+
lymphocytes
were prominent in early acne lesions. This finding,
which is consistent with elevation of antiP acnes
cellular immunity in patients with severe acne, is
significant, as antibody titers to P acnes parallel
acne severity.
17
These antibodies are required to
activate the classic pathway of complement. On
the other hand, the alternative pathway is acti-
vated by P acnes wall carbohydrate.
The comedo also may contain other inflammatory
factors. Allaker and Greenman
18
showed that P acnes
produces compounds that engage in histaminelike
activity. Ingham et al
19
found significant levels of
IL-1like activity and tumor necrosis factorlike
molecules in a majority of open comedones. The ori-
gins and actual structures of these factors have yet to
be defined.
When neutrophils arrive at the comedo, P acnes,
already opsonized by C3b or immunoglobulins, is
ingested. This interaction releases intracellular
hydrolytic enzymes but does not kill P acnes. These
lysosomal enzymes degrade the follicular epithelium,
which leads to rupture of the follicular wall and
extrusion of follicular epithelium, sebaceous lipids,
P acnes, and hair into the dermis, which in turn
causes inflammation. Several P acnesproduced
Acne Vulgaris, I: Pathogenesis and Diagnosis
104 CUTIS
extracellular enzymes (eg, hyaluronidase, protease)
also may be important in inflammation.
20
Clearly, the immune response in acne is not pro-
tective. Inappropriately activated immunity seems
likely to lead to propagation of inflammatory lesions
and subsequent scarring.
Diagnosis
Acne is generally limited to areas where sebaceous
glands are largest and most abundantthe face,
neck, chest, upper back, and upper arms. Individual
lesions are centered about sebaceous follicles.
Correct classification of lesion type (Table) is
essential for choosing the most effective therapy.
At the 1990 Consensus Conference on Acne
Classification,
21
a global evaluation of lesions and
their complications (eg, drainage, hemorrhage,
pain) was proposed. Psychosocial impact, failure
to respond to previous therapies, and occupa-
tional disability are 3 additional factors used in
grading acne.
Noninflammatory acne lesions are either closed
or open comedones. Acne that manifests as non-
inflammatory lesions is not classified severe unless
the number, size, and extent of such lesions are so
overwhelming as to warrant the designation.
Inflammatory acne lesions are papules, pustules,
and nodules. These lesions are classified as papulo-
pustular, nodular, or both. The severity grades
assigned (mild, moderate, severe) are based on
lesion count approximations (Table).
The term cystic acne should probably be aban-
doned, as there are no true cysts in acne (on rare
occasion, these cysts develop as a residual effect of
acne lesion healing). The term severe acne may be
applied if ongoing scarring or persistent lesion-
drainage is involved or if sinus tracts are present. In
addition, the most destructive forms of acneacne
conglobata, acne fulminans, follicular occlusion
triadare classified as very severe.
Differential Diagnosis
Although most acne diagnoses present no difficul-
ties, certain conditions or overlapping conditions
may be confusing. Differential diagnosis may be
stimulated by this noninclusive list: rosacea, ulery-
thema ophryogenes, gram-negative folliculitis,
steatocystoma multiplex, steroid acne, drug erup-
tions, perioral dermatitis, iododermas, verruca vul-
garis, verruca plana, bromodermas, syringomas,
sarcoidosis, trichoepitheliomas, follicular muci-
nosis, angiofibromas, infectious folliculitis, and
keratosis pilaris.
REFERENCES
1. Koo J. The psychological impact of acne: patients percep-
tions. J Am Acad Dermatol. 1995;32:S26-S30.
2. Plewig G, Fulton JE, Kligman AM. Cellular dynamics of
comedo formation in acne vulgaris. Arch Dermatol Forsch.
1971;242:12-29.
3. Vowels BR, Yang S, Leyden JJ. Induction of proinflam-
matory cytokines by a soluble factor of P acnes: implica-
tions for chronic inflammatory acne. Infect Immun.
1995;63:3158-3165.
4. Knaggs HE, Holland DB, Morris C, et al. Quantifica-
tion of cellular proliferation in acne using the mono-
clonal antibody Ki-67. J Soc Invest Dermatol.
1994;102:89-92.
5. Cunliffe WJ, Holland DB, Clark SM, et al. Comedogenesis:
some new aetiological, clinical and therapeutic strategies.
Br J Dermatol. 2000;142:1084-1091.
6. Kligman AM, Katz AC. Pathogenesis of acne vulgaris. Arch
Dermatol. 1968;98:53-57.
7. Motoyoshi K. Enhanced comedo formation in rabbit ear
skin by squalene and oleic acid peroxides. Br J Dermatol.
1983;109:191-198.
8. Kealey T. Hypovitaminosis A of follicular duct as a cause of
acne vulgaris [letter]. Lancet. 1988;2:449.
9. Guy R, Greem MR, Kealey T. Modeling acne in vitro. J
Invest Dermatol. 1996;106:176-182.
Acne Classification by Severity of Inflammatory Lesions
Severity Papules/Pustules Nodules
Mild Few to several None
Moderate Several to many Few to several
Severe Numerous to extensive Many
10. Thiboutot D, Gilliland K, Light J, et al. Androgen
metabolism in sebaceous glands from subjects with and
without acne. Arch Dermatol. 1999;135:1041-1045.
11. Pochi PE, Strauss JS, Rao RS, et al. Plasma testosterone
excretion and sebum production in males with acne
vulgaris. J Clin Endocrinol Metab. 1965;51:287-291.
12. Shalita AR, Lee WL. Inflammatory acne. Dermatol
Clin. 1983;1:361-364.
13. Weeks JG, McCarty L, Black T, et al. The inability of a
bacterial lipase inhibitor to control acne vulgaris. J
Invest Dermatol. 1977;69:236-243.
14. Lee WL, Shalita AR, Suntharalingam K, et al. Neu-
trophil chemotaxis by Propionibacterium acnes lipase
and its inhibition. Infect Immun. 1982;35:71-78.
15. Webster GF, Leyden JJ. Characterization of serum-
independent polymorphonuclear leukocyte chemotactic
factors produced by Propionibacterium acnes. Inflammation.
1980;4:261-269.
16. Norris JFB, Cunliffe WJ. A histological and immunocy-
tochemical study of early acne lesions. Br J Dermatol.
1988;118:651-659.
17. Puhvel SM, Barfatani M, Warnick M. Study of anti-
body levels to Corynebacterium acnes. Arch Dermatol.
1964;90:421-427.
18. Allaker RP, Greenman S. The production of inflamma-
tory compounds by Propionibacterium acnes and other
skin organisms. Br J Dermatol. 1987;117:175-183.
19. Ingham E, Eady EA, Goodwin CE, et al. Proinflamma-
tory levels of IL-1-like bioactivity are present in the
majority of open comedones in acne vulgaris. J Invest
Dermatol. 1992;98:895-901.
20. Puhvel SM, Hoffman LK. The production of
hyaluronidase by Corynebacterium acnes. J Invest
Dermatol. 1972;58:66-70.
21. Report of the Consensus Conference on Acne Classification.
J Am Acad Dermatol. 1991;24:495-500.
Acne Vulgaris, I: Pathogenesis and Diagnosis
VOLUME 70, AUGUST 2002 105
DISCLAIMER
The opinions expressed herein are those of the authors and do not necessarily represent the views of the sponsor or its publisher. Please review complete prescribing
information of specific drugs or combination of drugs, including indications, contraindications, warnings, and adverse effects before administering pharmacologic
therapy to patients.
FACULTY DISCLOSURE
The Faculty Disclosure Policy of the College of Medicine requires that faculty participating in a CME activity disclose to the audience any relationship with a pharma-
ceutical or equipment company that might pose a potential, apparent, or real conflict of interest with regard to their contribution to the program. It is required by the
Accreditation Council for Continuing Medical Education that each author of a CME article disclose to the participants any discussion of an unlabeled use of a commer-
cial product or device or an investigational use not yet approved by the Food and Drug Administration.
Dr. Oberemok reports no conflict of interest. Dr. Shalita is a consultant for Allergan, Inc; Dermik Laboratories; Medicis Pharmaceutical Corporation; and Stiefel
Laboratories, Inc. He also has received research grants from and served on speakers bureaus for Allergan, Inc; Collagenx Pharmaceuticals, Inc; Connetics
Corporation; Dermik Laboratories; Galderma Laboratories, LP; Medicis Pharmaceutical Corporation; and Stiefel Laboratories, Inc. Dr. Fisher reports no conflict of interest.
S-ar putea să vă placă și
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (895)
- D'CosmixDocument60 paginiD'Cosmixjones32Încă nu există evaluări
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- General Dermatology MCQsDocument197 paginiGeneral Dermatology MCQsDr.Tawheed95% (19)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
- Patient EducationDocument252 paginiPatient EducationAnn Peng100% (2)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5794)
- Galvanic FaqDocument9 paginiGalvanic FaqDang Quoc NguyenÎncă nu există evaluări
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- Zinc & Acne EfektifDocument3 paginiZinc & Acne EfektifAldi PurwandiÎncă nu există evaluări
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (266)
- VN Shegal - Textbook of Clinical Dermatology, 5th EditionDocument365 paginiVN Shegal - Textbook of Clinical Dermatology, 5th EditionAlbert Gheorghe93% (14)
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (400)
- Uses of Dalandan and CalamansiDocument8 paginiUses of Dalandan and CalamansiJhoel Banal67% (3)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- MoringaDocument3 paginiMoringachayle_fritz24Încă nu există evaluări
- "SKIN": Integument SystemDocument180 pagini"SKIN": Integument Systemraanja2Încă nu există evaluări
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- Homeopathy For Acne Treatment and PimplesDocument18 paginiHomeopathy For Acne Treatment and PimplesmiadelfiorÎncă nu există evaluări
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (588)
- Dr. Ujjwala Verma 2 Yr JRDocument60 paginiDr. Ujjwala Verma 2 Yr JRUjjwala Verma100% (1)
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- Osce QuestionsDocument128 paginiOsce Questionsdean100% (1)
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- Reti No IdesDocument15 paginiReti No IdesRobertoÎncă nu există evaluări
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (74)
- Air Force Waiver GuideDocument1.013 paginiAir Force Waiver GuideBrandon De La RosaÎncă nu există evaluări
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (345)
- A CASE of Acne VulgarisDocument4 paginiA CASE of Acne VulgarisHomoeopathic Pulse100% (1)
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- Well Cap Garcin e 2014Document23 paginiWell Cap Garcin e 2014herliaÎncă nu există evaluări
- Bielenda Katalog ENG PRODocument80 paginiBielenda Katalog ENG PROPiotr MimiGroupÎncă nu există evaluări
- A Randomised Controlled Study For The Treatment of Acne Vulgaris Using High Intensity 414nm Solid State Diode ArraysDocument29 paginiA Randomised Controlled Study For The Treatment of Acne Vulgaris Using High Intensity 414nm Solid State Diode ArraysCaerwyn AshÎncă nu există evaluări
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1090)
- Katalog Bielenda EN PDFDocument56 paginiKatalog Bielenda EN PDFBielendaÎncă nu există evaluări
- American Board of Family MedicineDocument72 paginiAmerican Board of Family MedicineMohamed Elmasry100% (6)
- What Is ImpetigoDocument4 paginiWhat Is ImpetigoKate Mylhen Espenido EnerlanÎncă nu există evaluări
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2259)
- Preview Cure YourselfDocument111 paginiPreview Cure YourselfShareef Ghouse50% (2)
- Practice Essentials AcneDocument6 paginiPractice Essentials AcneaigoooÎncă nu există evaluări
- Acne VC Comedogenesis and TreatmentDocument18 paginiAcne VC Comedogenesis and TreatmentYovie PrayektiÎncă nu există evaluări
- Medical Problems in AdolescentDocument65 paginiMedical Problems in Adolescentzarizijamil3918Încă nu există evaluări
- Acne Notes For MBBSDocument15 paginiAcne Notes For MBBSsobretilldawnÎncă nu există evaluări
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (121)
- SebumeterDocument3 paginiSebumeterwe sagara dewiÎncă nu există evaluări
- Dermatological DrugsDocument46 paginiDermatological DrugsJai MurugeshÎncă nu există evaluări
- Teenage Is A Fundamental Stage of Life That Each Human Being Passes ThroughDocument4 paginiTeenage Is A Fundamental Stage of Life That Each Human Being Passes ThroughDineish MurugaiahÎncă nu există evaluări
- Cosmetic Acupuncture: Clinical ExperienceDocument3 paginiCosmetic Acupuncture: Clinical ExperienceLuis Gomes100% (1)
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)