Documente Academic
Documente Profesional
Documente Cultură
Gel
Încărcat de
Shaezarah MohamudallyDrepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Gel
Încărcat de
Shaezarah MohamudallyDrepturi de autor:
Formate disponibile
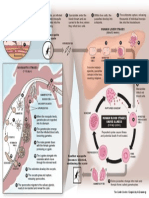
Gel Electrophoresis of DNA
The explosion of m olecular biology techniques that began in the mid-1970s (and
continues today) has provided tools to exam ine the physical structure of DNA, its nucleotide
sequence and how genes are read and regulated. One key tool is the ability to visualize DNA
m olecules and determ ine their length by using a technique called gel electrophoresis.
Introduction to gel electrophoresis
In gel electrophoresis, DNA fragm ents move through a porous m atrix m ade of agarose,
a gelatin-like substance purified from seaweed. The agarose is m elted like Jell-O and then
poured into a plastic tray to harden into a slab called a gel. A plastic comb inserted at one end
while the gel is hardening forms wells where DNA sam ples can be placed. The DNA is mixed
with a loading buffer that contains glycerol this makes it heavier than water, so it will sink
to the bottom of the well. The gel is then covered with a buffer solution that can carry electric
current, and electrodes are placed at each end of the gel and connected to a power supply.
Because DNA is negatively charged (each nucleotide has a negatively charged phosphate
attached to it), it will m ove toward the positive electrode. Larger m olecules m ove through the
agarose m ore slowly, while sm aller ones
can slip through the pores faster. So, the
fragments wind up arranged in order
according to size, with the sm aller ones
having m oved farther toward the positive
pole. Figure 47 shows an exam ple.
Because the DNA is invisible, the loading
buffer
also
con tain s
tw o
dyes:
brom ophenol blue (a sm all dye m olecule
that behaves like a DNA fragm ent about
600 bases long) and xylene cyanol (a
larger dye that acts like a DNA fragm ent of
about 4000 bases). These dyes form lines
that give you an idea of how far your DNA
has moved. Som e loading buffers also have
a third dye, behaving like a very sm all DNA
Figure 47. Gel electrophoresis separates DNA fragments by size.
m olecule (50 bases or so).
As the DNA m igrates, the different fragments will form bands; each band is composed
of many identical copies of a particular-size piece of DNA (you cant do gel electrophoresis with
one DNA molecule: you need m illions or billions of identical m olecules). The last step is to make
the DNA bands visible, using a fluorescent m olecule that inserts between the bases in the DNA
helix. W e use a com m ercial loading buffer called EZ-Vision which includes the fluorescent
m olecule, so the gel is already stained when its done running. Another m ethod is to soak the
gel in ethidium brom ide after running it. Either way, the bands can be seen using ultraviolet
light and photographed to m ake a perm anent record.
Sample preparation
Of course, gel electrophoresis requires som e kind of DNA sam ple a plasm id, a PCR
product, a segm ent of a chrom osom e, etc. If the m olecule is circular, enzymes are used to cut
the DNA (see the section on restriction digestion, page 87), because circular m olecules can be
either tightly or loosely coiled and dont wind up at the sam e place on a gel as a linear molecule
of the sam e size. W hatever your sam ple is, it must be mixed with loading buffer (containing
glycerol and dyes, as described above) before electrophoresis. Add a volum e of loading buffer
equal to 1/5 the volum e of your sample and m ix it well before loading your sam ple on the gel.
89
90
How to pour and run a gel
Once your DNA has been m ixed w ith loading buffer, the basic procedure for gel
electrophoresis is the sam e no m atter what kind of DNA it is. Follow the directions below.
1. For one gel, you will need a total of 30 ml of a 1 % agarose solution.
3 The concentration of agarose can be varied for use with DNA fragments of
different sizes. The table below gives some useful concentrations. For most typical-sized
DNA fragments, 1% is a good starting point.
2. Weigh 0.3 g of agarose and pour it into a 125-ml flask (0.3 g in 30 ml = 1 g in 100 ml = 1% w/v).
3. Prepare 300 ml of 10 TBE buffer by measuring 30 ml (directly from the carboy) into a 500-ml
graduated cylinder, then filling the cylinder to the 300-ml mark with dH 2O (from the tap in the media
room, not from a wash bottle, etc.).
3 TBE stands for Tris, boric acid and EDTA. The Tris and boric acid provide a buffer
which allows electric current to flow through the gel and maintains an appropriate pH.
EDTA binds to metal ions needed by enzymes that can break down DNA, so it helps
protect your DNA while it is in the gel. The TBE stock solution is made up at 10 times the
needed concentration (we call this 10 TBE), so we dilute it 1:10 for use at 1.
4. Cover the cylinder with a piece of parafilm (you only need a small piece stretch it to get a good
seal) and mix by inverting several times (never try to use a stir bar in a cylinder).
5. Pour 30 ml of the 1 TBE into the flask with your agarose. The remaining TBE will be your running
buffer, which carries the current from one electrode to the other.
6. Heat the agarose in the microwave on high for 15 seconds. Remove it (caution: hot!) and swirl the
flask gently. See if the agarose has completely melted (no powder, no floating transparent flakes).
If not, heat again for 10 seconds at a time until it all melts. Dont let it boil.
3 Do not overheat the solution! Agarose can become superheated and boil over
very rapidly, ruining your experiment and potentially causing burns.
7. Allow the melted agarose to cool on the
bench for two minutes while you set up the
casting tray.
8. Insert a gel plate into a casting tray as
shown in Figure 48. The gel plate has to be
open on the ends to allow the current to flow
through; the foam pads on the ends of the
casting tray allow the gel to harden without
leaking.
Figure 48. Inserting the gel plate into the casting tray.
91
9. The comb height should be set to leave just a little space between the bottom of the teeth and the
bottom of the gel tray. You can place a piece of thin cardboard under the comb to check its height.
10.Use a bubble level to ensure that your gel plate is
level. Adjust its position in the casting tray if its not.
11.Pour the entire 30 ml of melted agarose into the gel
casting tray. Insert a clean comb (which will form the
wells) near one end, and slide it up against the
handles on the gel plate, as shown in Figure 49.
12.Let the agarose harden for at least 15 minutes. You
should notice that it becomes a little more opaque.
13.When the agarose has fully hardened, carefully
remove the comb. Pull it straight up without tearing
the wells.
14.Remove the gel plate, with the gel on it, from the
casting tray and place it in the gel box.
Figure 49. Inserting the comb. (Two combs are
shown here, but you will generally only need one).
3 The plastic gel box is filled with polyethylene
glycol as a coolant and can be kept in the freezer prior to use for extra cooling if needed.
This keeps the gel from melting from the heat of the electric current, particularly on a long
run.
15.Add running buffer until the gel is completely submerged: if you look from the side, it should be
covered by a few millimeters of liquid. The gel is now ready for use.
16.Move your gel close to the power supply so you wont have to move it once samples are loaded.
17.Use a micropipettor to transfer your DNA size standards to the first well. If you are using the 1-kb
ladder (see below) as your size standard, you will need 5 :l.
3 You want the tip of your pipette just inside the well; the DNA will then fall to the
bottom of the well as you release it slowly. You should be able to see the well clearly if
you look straight down on it with the black benchtop underneath. Be careful not to poke
your pipette through the bottom of the well, and also avoid dripping DNA anywhere but
in the desired well. Don't release the plunger of the pipettor until you pull it out of the
buffer, or it'll suck your DNA back up!
18.Use a m icropipettor and a clean tip to transfer each of your DNA samples to a well. Keep track of
which sample is in which well.
3 Maximum volume in the wells is 20-25 :l. Typically, you should load 5-15 :l.
19.Place the lid on the gel box and fit the power cords over the two electrodes.
3 Be sure that the positive (red) cord is connected to the electrode at the bottom
(non-well end) of the gel. Remember that DNA is negatively charged and will migrate to
the positive pole. Or, just remember begin at black, run to red.
20.Connect the other ends of the power cords to a power supply, again being sure that the polarity is
correct. Turn on the power and set the voltage to 150 V. It should take about 50-60 minutes to run
the gel at this voltage.
3 You should see a large num ber of tiny bubbles forming along the wires at the
negative pole. If not, something is wrong.
92
21.Normally, you would want to stop the gel when the bromophenol blue (dark blue) dye line is near
the bottom edge of the gel but has not yet started to run off. Stop it sooner if you need to see small
DNA fragments. Turn off the power supply and disconnect the leads.
22.Carefully remove the gel tray and gel from the gel box. Don't let the gel slide off!
23.Slide the gel off the tray onto the UV light box of the photodocumentation system.Use the
photodocumentation system (see instructions on page 95) to photograph your gel and to save an
image in electronic format (for your notebook, lab report, poster, etc.) if desired.
24.W hen you're sure you have a good photo, dispose of the gel by wrapping it in a paper towel and
discarding it in the trash. Give the UV light box a squirt of deionized water and wipe it dry with a
Kimwipe.
25.Pour the running buffer down the drain and rinse off the gel box, gel tray, comb and casting tray with
water before putting them away.
Determining DNA fragment sizes
W hen you see the DNA fragm ents on your gel, youll know that the ones that w ent
farthest are the sm allest, but how can you determ ine their actual sizes? W e can do this by
comparing their position on the gel to the positions of known fragm ents.
The length of a DNA m olecule is measured by the number of nucleotide pairs, or basepairs (abbreviated bp) that m ake it up. For a long molecule, we use kilobase-pairs (kb): 1 kb
= 1000 bp. The distance that the molecule m oves in the gel (we say it migrates) is proportional
to its length. So, we put DNA size m arkers (a m ixture of fragm ents of known sizes) in one well
of each gel, m easure how far each one m igrated and then m ake a graph (called a standard
curve) that allows us to determ ine the length of any other fragm ent on the gel.
There are m any different DNA size m arkers available; you can m ake your ow n or buy
a pre-made set from a biotech com pany. In our labs, we usually use a comm ercially prepared
set called the 1-kb Plus DNA Ladder, produced by LifeTechnologies, Inc.
3 Be sure you know what ladder you are using! Different companies make different
ladders, and several are called 1-kb ladders.
As its nam e indicates, the largest 11 fragm ents in the 1-kb Plus
mixture are 1 kb, or 1000 bp, apart: 12,000 bp through 2,000 bp (Figure 50).
Then, there are several sm aller fragm ents about 100 bp apart. Usually, youll
be using a gel designed to effectively separate either large or small fragm ents,
and its likely that either your large m arker fragm ents will get sm ooshed up so
that you cant tell them apart or that your sm all ones will run off the gel. So,
how can you be sure which marker band is which? Notice the pattern of the
bands in the picture: There are two bands close together, with the lower one
brighter than the upper: these are the 2,000-bp and 1,650-bp fragm ents.
Although your ladder m ay not look exactly like the one here, you should be able
to see this landm ark pattern. Then, count up or down to identify as m any
other bands as you can. Dont worry if you cant identify all 20 bands.
To determ ine the sizes of the fragm ents in your gel, first m easure the
distance migrated for each band that you can identify in the 1-kb ladder lane.
Use a ruler and simply m easure the distance from the well to the band, in m m .
Measure from the bottom of the well to the bottom of the band. This has to be
done separately for every gel, because no two gels will be absolutely identical
(comb may be placed a little differently, run a little longer, etc.). Measure to the
nearest tenth of a millim eter.
Put the distances migrated into one column of an Excel spreadsheet,
and the fragm ent sizes into the next colum n. Have Excel graph fragm ent size
(in bases, on the y-axis) vs. distance m igrated (in m m , on the x-axis), but dont
let it connect the dots. See the graphing section of this m anual (page 43) for
Figure 50. The
LifeTechnologies
1kb Plus DNA
Ladder.
93
help. Give the y-axis a logarithm ic scale. The points for your 1-kb m arkers should then form
almost a straight line (m aybe a little curved at the upper end). Figure 51 shows an example (but
it is just an example you will have to draw your own graph for your own gel!).
Once the points
are plotted, draw a bestfit line. If your points are
plotted well, you should
be able to get Excel to
draw a good line (hint: try
an exponential trendline,
which should com e out
linear
in
appeara n ce
because of the logarithm ic
scale). Now, the size of
any unknown fragm ent in
the gel can be calculated
by using the equation of
the best-fit line. But, be
wise in how you use this
equation: look carefully at Figure 51. Using known fragment sizes to produce a standard curve.
your
l in e
and
pay
attention to how well it fits the data. If your data curve away from the line in som e area, then
you know that the equation will lead you to m is-size bands in that area.
One final im portant point about using the 1-kb Plus ladder: it can also be used to
estim ate the amount of DNA you have in a particular band. The m anufacturer tells us that the
1650-bp band contains about 8% of the total m ass of DNA in your ladder lane. W hen we buy
the ladder, we dilute it to 100 ng/l in 1 loading buffer. So, if you use 5 l of ladder in your
m arker lane, you have 500 ng of DNA total, and the 1650-bp band should represent 500 0.08
= 40 ng of DNA. (Obviously, youll have to adjust this calculation if you load a different amount.)
Now, you can either eyeball a band and get a rough idea of how much DNA it contains, or you
could capture an im age of your stained gel (see the Gel Docum entation section) and use image
analysis software such as Im ageJ to quantitate the DNA in your band as compared to the 1650bp ladder band.
S-ar putea să vă placă și
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5794)
- Evidence For DNA As The Hereditary MoleculeDocument2 paginiEvidence For DNA As The Hereditary MoleculeShaezarah MohamudallyÎncă nu există evaluări
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (399)
- Ch10-1 Gen MaterialDocument36 paginiCh10-1 Gen MaterialarcheologistsÎncă nu există evaluări
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- MicropipetteDocument7 paginiMicropipetteMike Serge Razafi0% (1)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (894)
- Ascomycotina Class PPT 2014Document80 paginiAscomycotina Class PPT 2014Shaezarah Mohamudally67% (3)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
- Gram StainDocument1 paginăGram StainShaezarah MohamudallyÎncă nu există evaluări
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- Malaria PDFDocument36 paginiMalaria PDFYesi Novia AmbaraniÎncă nu există evaluări
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- Identifying Unknown Bacterium-GuideDocument13 paginiIdentifying Unknown Bacterium-GuideShaezarah MohamudallyÎncă nu există evaluări
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- Harvard System of ReferenceDocument7 paginiHarvard System of Referencejaya1816Încă nu există evaluări
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (587)
- Biochemical Composition of Fish - A PrimerDocument6 paginiBiochemical Composition of Fish - A PrimerShaezarah MohamudallyÎncă nu există evaluări
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (265)
- Turbidity ScienceDocument26 paginiTurbidity ScienceShaezarah MohamudallyÎncă nu există evaluări
- Similarities Between Prokaryotic and Eukaryotic CellsDocument2 paginiSimilarities Between Prokaryotic and Eukaryotic CellsShaezarah MohamudallyÎncă nu există evaluări
- Malaria Parasites and DiseaseDocument7 paginiMalaria Parasites and DiseaseBalaji Rao NÎncă nu există evaluări
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- Fungus Lec NotesDocument8 paginiFungus Lec NotesShaezarah MohamudallyÎncă nu există evaluări
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (73)
- Characteristics of Fungi N ProtistDocument8 paginiCharacteristics of Fungi N ProtistShaezarah MohamudallyÎncă nu există evaluări
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (344)
- Malaria Life Cycle ChartDocument1 paginăMalaria Life Cycle ChartShaezarah MohamudallyÎncă nu există evaluări
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- LieDocument1 paginăLieShaezarah MohamudallyÎncă nu există evaluări
- Mal LCDocument4 paginiMal LCreborn007Încă nu există evaluări
- Boltzmann DistributionDocument2 paginiBoltzmann DistributionShaezarah MohamudallyÎncă nu există evaluări
- Lecture of Organic ChemistryDocument13 paginiLecture of Organic ChemistryNutDen R X NuttapongÎncă nu există evaluări
- Growth Curves of Micro-OrganismsDocument31 paginiGrowth Curves of Micro-OrganismsShaezarah MohamudallyÎncă nu există evaluări
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- Topic3 BIOL1030NRDocument11 paginiTopic3 BIOL1030NRShaezarah MohamudallyÎncă nu există evaluări
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2219)
- PolarityDocument1 paginăPolarityDeepakÎncă nu există evaluări
- Fungus Lec NotesDocument8 paginiFungus Lec NotesShaezarah MohamudallyÎncă nu există evaluări
- Microsoft PowerPoint - Bio1009ch13Document48 paginiMicrosoft PowerPoint - Bio1009ch13Shaezarah MohamudallyÎncă nu există evaluări
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1090)
- Lecture of Organic ChemistryDocument13 paginiLecture of Organic ChemistryNutDen R X NuttapongÎncă nu există evaluări
- Diff Cent PDFDocument33 paginiDiff Cent PDFShaezarah MohamudallyÎncă nu există evaluări
- Cell Counting Using A HemacytometerDocument3 paginiCell Counting Using A HemacytometerKris Cahyo Mulyatno100% (1)
- RAlaminarflowcabinets PDFDocument4 paginiRAlaminarflowcabinets PDFShaezarah MohamudallyÎncă nu există evaluări
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (119)
- ChemistryDocument9 paginiChemistryShaezarah MohamudallyÎncă nu există evaluări
- CSI Polymerase Chain Reaction Lab ManualDocument6 paginiCSI Polymerase Chain Reaction Lab ManualDank MoviesÎncă nu există evaluări
- Analytical Method PDFDocument85 paginiAnalytical Method PDFiaderzÎncă nu există evaluări
- DNA From Caviar': NCBE, University of ReadingDocument4 paginiDNA From Caviar': NCBE, University of ReadingYosh MidhrasÎncă nu există evaluări
- Colloids and Surfaces A: Physicochemical and Engineering AspectsDocument7 paginiColloids and Surfaces A: Physicochemical and Engineering Aspectsandreea g.Încă nu există evaluări
- A Basic PCR ProtocolDocument4 paginiA Basic PCR ProtocolIrfan HussainÎncă nu există evaluări
- Microbial diversity of indigenous microorganism communities from Vietnam agro-ecosystemsDocument12 paginiMicrobial diversity of indigenous microorganism communities from Vietnam agro-ecosystemsrahmanÎncă nu există evaluări
- Developing Best-Practice Models For The Therapeutic Use of Extracellular VesiclesDocument10 paginiDeveloping Best-Practice Models For The Therapeutic Use of Extracellular VesicleschenÎncă nu există evaluări
- Protein Electrophoresis GEDocument82 paginiProtein Electrophoresis GEIsa Ribeiro100% (1)
- Ape Lex 2015Document106 paginiApe Lex 2015Bolawali KiÎncă nu există evaluări
- 7.biochemical and Structural Characterization of Sturgeon FishDocument10 pagini7.biochemical and Structural Characterization of Sturgeon FishJonathanCoralÎncă nu există evaluări
- Biochem PDFDocument53 paginiBiochem PDFMiriam JonesÎncă nu există evaluări
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)
- Heat Number in MilkDocument17 paginiHeat Number in MilkLaura MarcelaÎncă nu există evaluări
- Spife TouchDocument4 paginiSpife TouchRita LeongÎncă nu există evaluări
- A Short Review On Proteomics and Its ApplicationsDocument9 paginiA Short Review On Proteomics and Its ApplicationsMySCRIBDÎncă nu există evaluări
- Chapter 11 Biotechnology Principle and ProcessDocument50 paginiChapter 11 Biotechnology Principle and ProcessMkisneymar Meymar100% (1)
- Rapd Profile For Lab ReportDocument4 paginiRapd Profile For Lab Reportapi-341432127Încă nu există evaluări
- Table of ContentsDocument582 paginiTable of Contentsno contractÎncă nu există evaluări
- Western Blot ProtocolDocument6 paginiWestern Blot ProtocollinhÎncă nu există evaluări
- ElectrophoresisDocument38 paginiElectrophoresisHennah Usman67% (3)
- Go TaqDocument2 paginiGo TaqYogi ArismetÎncă nu există evaluări
- USP34-NF29 IndexDocument72 paginiUSP34-NF29 IndexAraya Rattanavichian100% (2)
- Good's Buffers (Biological Buffers) : Products Description / OverviewDocument9 paginiGood's Buffers (Biological Buffers) : Products Description / OverviewCHIRANJEEVIÎncă nu există evaluări
- Statistical Analysis For Plant Tissue Culture DataDocument68 paginiStatistical Analysis For Plant Tissue Culture Datadolt2111Încă nu există evaluări
- For The PA 800 Plus Pharmaceutical Analysis System: SDS-MW Analysis KitDocument42 paginiFor The PA 800 Plus Pharmaceutical Analysis System: SDS-MW Analysis KitThanh Tâm TrầnÎncă nu există evaluări
- Factors Affecting ElectrophoresisDocument20 paginiFactors Affecting ElectrophoresisMuhammad Ramzan0% (1)
- Gel Electrophorosis MpatDocument19 paginiGel Electrophorosis Mpatkavya nainitaÎncă nu există evaluări
- Vikash Kumar: Career ObjectiveDocument2 paginiVikash Kumar: Career ObjectiveAnikesh SinghÎncă nu există evaluări
- Exp 7-SDS-PAGEDocument18 paginiExp 7-SDS-PAGERadwan M SaadehÎncă nu există evaluări
- User Manual: Macherey-NagelDocument34 paginiUser Manual: Macherey-NagelBatuli MwaijandeÎncă nu există evaluări
- 2022 - Biochemistry - Esha D. Dalvie, Jordan C. Stacy, Keir C. Neuman - Recognition of DNA Supercoil Handedness During Catenation Catalyzed byDocument33 pagini2022 - Biochemistry - Esha D. Dalvie, Jordan C. Stacy, Keir C. Neuman - Recognition of DNA Supercoil Handedness During Catenation Catalyzed byjimgogreatÎncă nu există evaluări
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeDe la EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeEvaluare: 4.5 din 5 stele4.5/5 (3)
- Stuff Matters: Exploring the Marvelous Materials That Shape Our Man-Made WorldDe la EverandStuff Matters: Exploring the Marvelous Materials That Shape Our Man-Made WorldEvaluare: 4 din 5 stele4/5 (289)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDe la EverandChemistry for Breakfast: The Amazing Science of Everyday LifeEvaluare: 4.5 din 5 stele4.5/5 (14)
- Guidelines for Asset Integrity ManagementDe la EverandGuidelines for Asset Integrity ManagementEvaluare: 5 din 5 stele5/5 (1)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction in the Science of Everyday LifeDe la EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction in the Science of Everyday LifeEvaluare: 4 din 5 stele4/5 (9)
- The Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsDe la EverandThe Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsEvaluare: 4 din 5 stele4/5 (146)
- Science Goes Viral: Captivating Accounts of Science in Everyday LifeDe la EverandScience Goes Viral: Captivating Accounts of Science in Everyday LifeEvaluare: 5 din 5 stele5/5 (1)