Documente Academic
Documente Profesional
Documente Cultură
NewCh 10-2 Denitrification 2012-2 (호환 모드) PDF
Încărcat de
Huy ĐoànDescriere originală:
Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
NewCh 10-2 Denitrification 2012-2 (호환 모드) PDF
Încărcat de
Huy ĐoànDrepturi de autor:
Formate disponibile
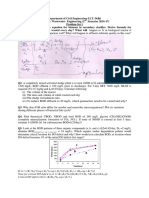
Example 10.
1 Stoichiometry of denitrification reactions
Denitrification scheme
H t t hi with
Heterotrophic
ith methanol
th
l
Heterotrophic with acetate
Autotrophic with H2
Parameters
f0s values : 0.36, 0.52, 0.21
SRT : 15 days
Compute the overall Stoichiometric reactions for each system
Example 10.1 Stoichiometry of denitrification reactions
1.
Calculate the fs, fe
2.
Cell synthesis (Rc) with NO3
( Equation C-2, Table 2.4, same for all)
3.
Acceptor equation (Ra)
(Equation I-7,
I 7, Table 2.2, same for all )
4.
Donor equation (Rd)
5.
Compute the stoichiometric equation
1 + ( 1 f d )b x
fs = f
1 + b x
0
s
R = f e Ra + f s Rc Rd
( methanol, acetate, H2, Table 2.3)
[3.33]
[2.37]
Example 10.1 Stoichiometry of denitrification reactions
i) Heterotrophic with Methanol
Assumption : b = 0.05/day(Table 10.1) , fd=0.8 for all system
1. Calculate fs, fe
fs
fe
0.267
0.733
2. Cell synthesis equation
1
5
29 +
1
11
NO 3- + CO 2 +
H + e- C5H 7 O 2 N + H 2O
28
28
28
28
28
3. Acceptor equation
4. Donor equation
1
6
1
3
NO 3- + H + + e - N 2 + H 2 O
5
5
10
5
1
1
1
H 2O
CO 2 + H + + e - CH 3OH +
6
6
26
Example 10.1 Stoichiometry of denitrification reactions
f s Rc
f e Ra
Rd
1
5
29 +
1
11
NO 3- + CO 2 +
H + e - C 5 H 7 O 2 N + H 2 O)
28
28
28
28
28
1
6
1
3
0.733 ( NO 3- + H + + e - N 2 + H 2 O)
5
5
10
5
1
1
1
CH 3OH +
H 2 O CO 2 + H + + e 6
26
6
0 267 (
0.267
0.1667CH 3OH + 0.1561NO 3- + 0.1561H +
0.00954 C 5 H 7 O 2 N + 0.0733 N 2 + 0.3781H 2 O + 0.119CO 2
Example 10.1 Stoichiometry of denitrification reactions
i) Heterotrophic with methanol
0.1667CH 3OH + 0.1561NO 3- + 0.1561H +
0.00954 C 5 H 7 O 2 N + 0.0733 N 2 + 0.3781H 2 O + 0.119CO 2
ii) Heterotrophic with acetate
0.125CH 3COO - + 0.1438 NO 3- + 0.1438H +
0.0122C 5 H 7 O 2 N + 0.0658 N 2 + 0.125HCO 3- + 0.0639CO 2 + 0.1542 H 2 O
iii) Autotrophic
A t t
hi with
ith H2
0.5H 2 + 0.1773 NO 3- + 0.0246CO 2 + 0.1773H +
0.00493C 5 H 7 O 2 N + 0.00862 N 2 + 0.5714H
0 5714H 2 O
Table 10.2.
Summary of Example 10.1
Summary of Example 10.1
1.
The observed yield of denitrifiers (fs ) declines significantly from acetate to
methanol to H2 as the donor.
2
2.
4 3 ~ 13 % of e- eq of NO3- -N
4.3
N consumed is used as the N source in synthesis
synthesis.
The great % (13 %) is associated with the greatest fs (0.342).
For acetate, e- eq as N-source =0.342 x (8/28) = 0.098 (eqns. at p.502)
% of e- eq = (0.098/0.756) x 100 = 13 %.
3. 28.6 % of the oxygen demand of the biomass is invested in reducing the Nsource (NO3-) to the -3 oxidation state.
4. For heterotrophic nitrification with acetate requires an input of
~ 4g BODL / g NO3- -N and produces ~ 0.69 g VSS of biomass (=sludge
wasting rate) and 3.6 g as CaCO3 of alkalinity (= buffer requirement).
10.3 One-Sludge Denitrification
Tertiary denitrification : the water does not contain the necessary electron
donor and thus an exogenous electron donor must be provided.
One-sludge denitrification : the water contains an electron donor that can
drive denitrification.
- One-sludge denitrification (= single-sludge = combined denitrification)
uses the BOD in the influent of a wastewater to drive denitrification
- Two g
goals in one-sludge
g denitrification :
1) Providing aerobic conditions that allow full nitrification
2) Providing anoxic conditions and Reserving BOD (organic electron donor)
for denitrification.
*1) and 2) seems to conflict with each other, but they should be done simultaneously .
Consequently, the task is to how to reconcile them.
10.3 One-Sludge Denitrification
Benefits :
- No exogenous electron donor needs to be added.
chemical costs are reduced over tertiary denitrification.
- Some of the influent BOD is oxidized with nitrate as the electron
acceptor (not O2).
)
aeration costs are reduced compared to alternative systems that
oxidize all BOD and nitrify the reduced-nitrogen forms in the influent
with O2 .
- Full
F ll or nearly
l ffullll N removall iis achieved
hi
d protecting
i receiving
i i waters
at risk from cultural eutrophication.
10.3.1 Basic One-Sludge Strategies
Influent wastewater contains TKN. In one sludge denitrification, the TKN must
be oxidized to NO3- - N without oxidizing all the BOD before denitrification
takes place.
Total Kjeldahl nitrogen (TKN) = organic BOD and reduced nitrogen (NH4+)
Despite a wide range of engineering configurations
configurations,
all one-sludge processes rely on one or more of three basic strategies.
Three basic strategies for reserving organic electron donor while nitrification
takes place :
1) Biomass storage and decay
2)) Classical predenitrification
p
3) Simultaneous nitrification with denitrification
10.3.1
1) Biomass storage and decay
- The synthesis of biomass stores electron equivalents that originally came
from the BOD and can be released through endogenous respiration to
drive denitrification
denitrification.
-
TKN NO3 -N
BOD: partly oxidized and
partly stored (synthesis)
NO3--N N2
Biomass as electron donor
((endogenous
g
respiration)
p
)
Fig.10.1 (a) biomass storage and decay
10.3.1
1) Biomass storage and decay
- It is called Wuhrmann biomass decayer (Swiss engineer K. Wuhrmann, 1964)
- Biomass storage and decay has limited applicability by itself and is not often
employed as a stand-alone process due to two shortcomings:
1) endogenous respirations has slow kinetics (b = 0.05/d)
0 05/d)
a high conc. of MLVSS (operating problems with settler and recycle)
and a long HRT in a anoxic tank
high capital costs are necessary
2) the decay of biomass always releases
+
NH4 -N from the anoxic step although at
concentrations lower than in the influent.
10.3.1 Classical predenitrification
- The first tank (anoxic) ; the influent BOD (electron donor) is directly utilized
for denitrification.
- The second tank ((aerobic);
); the influent TKN is nitrified to NO3 and
any remained BOD is oxidized.
- The nitrate formed in the aerobic tank is recycled to a anoxic tank (Qr2 ; large)
Directly utilizes the influent BOD
(electron donor) for denitrification
TKN NO3--N
Remained BOD: aerobically consumed
Fig.10.1
Fig
10 1
(b) classical predenitrification
10.3.1 Classical predenitrification
Qr2
-Fractional removal of N = ~
Q + Qr2
Q : plant flow rate
Qr2 : mixed-liquor
recycle flow rate
- The large recycle flow of NO3from the second to the first tank
is necessary, because NO3- not recycled
leaves in the effluent.
- Recycle ratios of 400 percent or more
are employed to bring enough NO3
back to the anoxic tank so that total N
removals are substantial.
10.3.1 Classical predenitrification
Widespread use worldwide;
- Advantages:
i) direct use of influent BOD for denitrification
reduces aeration costs for the removal of BOD
ii) faster
f t kinetics
ki ti than
th with
ith biomass
bi
storage
t
and
dd
decay
+
iii) no release of NH4 -N in the effluent
- Disadvantage:
i) large mixed-liquor recycle rate
increases
i
costs
t off piping
i i and
d pumping
i
10.3.1 Simultaneous nitrification with denitrification
Three factors allow all reactions to occur simultaneously.
1) Various nitrogen reductases using N as e- acceptor are repressed
only when the D.O. conc. is well above 1 mg/L.
2) However, inhibition of the nitrogen reductase is not severe when the D.O.
conc. is less than 1 mg/L.
NO3 NO2 NO N 2 O N 2
NO3 + 2e + 2 H + = NO2 + H 2 O
Nitrate Re ductase
NO2 + e + 2 H + = NO + H 2 O
Nitrite Re ductase
2 NO + 2e + 2 H + = N 2 O + H 2 O
Nitric Oxide Re ductase
N 2 O + 2e + 2 H + = N 2 + H 2 O
Nitrous Oxide Re ductase
10.3.1 Simultaneous nitrification with denitrification
Three factors allow all reactions to occur simultaneously.
3) D.O.
D O conc.
conc is depressed inside the aggregates that normally form in
treatment systems; thus, denitrification can occur inside the floc (or biofilm),
as long as the electron donor (BOD) penetrates inside.
- When D.O. concentration is poised at a suitably low level (< ~ 1 mg/L),
anoxic denitrification can occur in p
parallel to the aerobic reactions of
nitrification and aerobic BOD oxidation.
Fig.10.1 (c) simultaneous nitrification with denitrification
10.3.1 Simultaneous nitrification with denitrification
- 100 percent N removal by simultaneous nitrification with denitrification
has been documented (Rittmann and Langeland
Langeland, 1985)
1985), and small
amounts of denitrification probably occur in most activated sludge
systems that nitrify and have D.O. conc. below saturation (deSilva, 1997)
- Simultaneous nitrification with denitrification offers all the advantages
of predenitrification, but overcomes the main disadvantage, the high
recycle rate.
- Maintaining
g a low D.O. conc. throughout
g
one reactor creates an infinitely
y
high recycle ratio and allows essentially 100 percent N removal.
- Drawback
: We do not yet know the combinations of SRT, HRT, and D.O. conc. that
guarantee reliability.
reliability
10.3.1 Epilog for One-Sludge Processes
Common features of all one-sludge processes :
- one community (or one sludge) of microorganisms carries out all the
reactions.
- The heterotrophs switch back and forth between aerobic and
anoxic respiration, or they do both simultaneously.
- Because the nitrifiers are slow growing autotrophs,
their growth rate controls the SRT needed. SRTs greater than 15 days
are req
required
ired in most cases
cases, sometimes m
much
ch longer SRTs are used.
sed
- The longer SRTs provide an added safety factor for the nitrifiers,
who experience periods of low or zero DO.
- The long SRTs mean that accumulation of inert suspended solids is important.
10.3.1 Epilog for One-Sludge Processes
Common features of all one-sludge processes :
-
A practical outcome of a long SRT is that the HRT (in settler) needs to
increase in order to keep the MLSS conc. within reasonable limits dictated by
settler performance.
Thats why settler parameters for one-sludge denitrification is similar to
those used for extended aeration activated sludge.
- HRTs for predinitrification and simultaneous nitrification with denitrification
are at least 10 h for typical sewage, and 24 h or greater are used in some
instances.
instances
- To overcome the limitations of one-sludge denitrification, the Barnard
process, sequencing batch reactor (SBR), and biofilm systems are
developed.
10.3.2
Variations on the Basic One-Sludge processes
Barnard process (1975) by Dr. J. Barnard of South Africa
50 mg/L
g TKN
Influent TKN : 50 mg/L
recycle ratio (Qr2 ) from reactor 2 to 1
: 400 %
Reactor
R
t 1:
1 d
denitrification
it ifi ti rate
t is
i 100% off
recycled input and 80 % of the influent TKN
(Qr2=400%)
Reactor 2
NO3--N/L leaving : 10 mg/L (20%)
10 mg/L (20%)
+
3 mg NH4 -N /L
Reactor 3 :endogenous decay off cells
+
- 0.3 mg NH4 -N is released per mg NO3-N
due to endogenous cell decay
-Because 10 mg/L of NO3-N is converted to
N2 gas, Reactor 3 releases 3mg NH3-N/L
Reactor 4 releases 3 mg NO3-N/L
Final effluent TKN : 3 mg NO3-N/L
Total removal rate : 94 %
3 mg NO3 -N/L
(6 %)
Fig.10.2 Schematic of the Barnard process
10.3.2
Variations on the Basic One-Sludge processes
50 mg/L
g TKN
Barnard process
Well established worldwide one-sludge
denitrification process (> 90% N removal)
10 mg/L
Drawbacks
- need many tanks
- long
l
HRT
- significant mixed-liquor recycle
(400 %)between reactors 2 and 1
3 mg NH4 -N /L
3 mg NO3 -N/L
Fig.10.2 Schematic of the Barnard process
S-ar putea să vă placă și
- Biological DenitrificationDocument40 paginiBiological DenitrificationNordine MerahÎncă nu există evaluări
- N Removal: CIE4485 Laboratory ExperimentDocument13 paginiN Removal: CIE4485 Laboratory ExperimentAbass MarrakchiÎncă nu există evaluări
- 1 KineticsDocument5 pagini1 KineticsBon BonÎncă nu există evaluări
- Biodegradable Polymers As Solid Substrate and Biofilm Carrier For Denitrification in Recirculated Aquaculture SystemsDocument11 paginiBiodegradable Polymers As Solid Substrate and Biofilm Carrier For Denitrification in Recirculated Aquaculture SystemsNam NguyenÎncă nu există evaluări
- Houas 2001Document13 paginiHouas 2001chem19111117Încă nu există evaluări
- Completely Autotrophic Nitrogen Removal Over Nitrite in One Single Reactor - 2002 - Water ResearchDocument8 paginiCompletely Autotrophic Nitrogen Removal Over Nitrite in One Single Reactor - 2002 - Water ResearchAlvaro HueteÎncă nu există evaluări
- CT4485 Exam 31-01-2012 With AnswersDocument7 paginiCT4485 Exam 31-01-2012 With AnswersVali100% (1)
- Che 211 Referred Eos 2022-23-2Document7 paginiChe 211 Referred Eos 2022-23-2Clevas MseluleÎncă nu există evaluări
- CO2 Absorption in Aqueous MDEA With CA - Preprint As AcceptedDocument18 paginiCO2 Absorption in Aqueous MDEA With CA - Preprint As AcceptedGaurav GuptaÎncă nu există evaluări
- Chemistry Worksheet 2Document3 paginiChemistry Worksheet 2LemontÎncă nu există evaluări
- Essential Chemical Concepts Session IIDocument26 paginiEssential Chemical Concepts Session IIHamza QureshiÎncă nu există evaluări
- CEL795 - HW8 Solution and Additional ProblemsDocument11 paginiCEL795 - HW8 Solution and Additional ProblemsSaddy KhanÎncă nu există evaluări
- 111-1 環化第1次段考詳解 V2Document5 pagini111-1 環化第1次段考詳解 V2Kyle LuÎncă nu există evaluări
- Chapter 9 - Biological Waste Water TreatmentDocument17 paginiChapter 9 - Biological Waste Water Treatmenttonytelegraph0% (1)
- SAMPLE PAPER-04 (Solved) Class - XII Answers: NO Fe H NO Fe HO Feho NO Feho NO HODocument8 paginiSAMPLE PAPER-04 (Solved) Class - XII Answers: NO Fe H NO Fe HO Feho NO Feho NO HOShivam KumarÎncă nu există evaluări
- PDF 170897 93786Document9 paginiPDF 170897 93786ali abdulrahman al-ezziÎncă nu există evaluări
- 1 s2.0 S138589471200558X MainDocument8 pagini1 s2.0 S138589471200558X Main罗代兵Încă nu există evaluări
- EDUC 3136 A TeST 1 Reaction Kinetics 2023 PDFDocument11 paginiEDUC 3136 A TeST 1 Reaction Kinetics 2023 PDFKgaugelo FenyaneÎncă nu există evaluări
- Partial Nitrification Under Limited Dissolved Oxygen ConditionsDocument7 paginiPartial Nitrification Under Limited Dissolved Oxygen ConditionsPHƯỢNG NGUYỄNÎncă nu există evaluări
- CVL723PS1Document5 paginiCVL723PS1Ronald Ewa100% (1)
- Assignment 4Document3 paginiAssignment 4Đạt Trương MinhÎncă nu există evaluări
- Seminários 7Document6 paginiSeminários 7Angeline MoraisÎncă nu există evaluări
- MCH 05 2020 April Final Exam Option OneDocument20 paginiMCH 05 2020 April Final Exam Option Onepaul kapambweÎncă nu există evaluări
- Ja5061388 Si 001Document38 paginiJa5061388 Si 001vasut.nakÎncă nu există evaluări
- Physiology and Kinetics of Autotrophic Denitrification by Thiobacillus DenitrificansDocument7 paginiPhysiology and Kinetics of Autotrophic Denitrification by Thiobacillus DenitrificansPatrick Antony Bent BowieÎncă nu există evaluări
- The Photo-Fenton Reaction and The TiO2/UV Process For Waste Water Treatment Novel DevelopmentsDocument14 paginiThe Photo-Fenton Reaction and The TiO2/UV Process For Waste Water Treatment Novel DevelopmentsJOSEÎncă nu există evaluări
- CIV4042F - Tutorial 5 - 2021 PDFDocument1 paginăCIV4042F - Tutorial 5 - 2021 PDFGOATÎncă nu există evaluări
- Sk0014 Physical & Iinorganic Chemistry Tutorial 4: Reaction KineticsDocument2 paginiSk0014 Physical & Iinorganic Chemistry Tutorial 4: Reaction KineticsNeil8353 GgÎncă nu există evaluări
- Assignment 4Document3 paginiAssignment 4Duy Do MinhÎncă nu există evaluări
- Numerical Modeling of Biodegradation: Analytical and Numerical Methods by Philip B. BedientDocument27 paginiNumerical Modeling of Biodegradation: Analytical and Numerical Methods by Philip B. BedientVikash JaiswalÎncă nu există evaluări
- Bor 3Document8 paginiBor 3petru apopeiÎncă nu există evaluări
- Application of The Anammox ProcessDocument28 paginiApplication of The Anammox ProcessEmiliano Rodriguez TellezÎncă nu există evaluări
- Styrene Production Plant Dwsim FlowsheetDocument11 paginiStyrene Production Plant Dwsim FlowsheetmurtadaÎncă nu există evaluări
- Classwork Alle Arbeitsblätter Und FINAL EXAMS - 28 - 02 - 2019-1Document77 paginiClasswork Alle Arbeitsblätter Und FINAL EXAMS - 28 - 02 - 2019-1aslamfathima.20Încă nu există evaluări
- Topic 1: Monitoring The Environment: Stage 2 Chemistry Must Know ListDocument40 paginiTopic 1: Monitoring The Environment: Stage 2 Chemistry Must Know ListThalia MaesÎncă nu există evaluări
- Homework 3Document3 paginiHomework 3Haonan ZhangÎncă nu există evaluări
- Water ChemistryDocument55 paginiWater ChemistrypanduÎncă nu există evaluări
- Environmental FateDocument41 paginiEnvironmental FatedhikaÎncă nu există evaluări
- 8 DenitrificationDocument50 pagini8 DenitrificationArjun RanabhatÎncă nu există evaluări
- BTC 450 - BiorremediacionDocument2 paginiBTC 450 - Biorremediacionpia_shupaÎncă nu există evaluări
- Sample WW QuestionsDocument10 paginiSample WW QuestionsKrishna Kumar MeenaÎncă nu există evaluări
- Lecture 11Document11 paginiLecture 11Qasim KhanÎncă nu există evaluări
- CBSE Class 12 Chemistry Sample Paper Solution Set 1Document9 paginiCBSE Class 12 Chemistry Sample Paper Solution Set 1Sidharth SabharwalÎncă nu există evaluări
- Nama: Nuri Irianti NIM: 1009035009 Prodi: Teknik LingkunganDocument3 paginiNama: Nuri Irianti NIM: 1009035009 Prodi: Teknik LingkunganNuri IriantiÎncă nu există evaluări
- Hydrolysis 2016Document20 paginiHydrolysis 2016Kashish ParmarÎncă nu există evaluări
- Wastewater and BioremediationDocument7 paginiWastewater and BioremediationIshanSaneÎncă nu există evaluări
- Flue Gas Cleaning PDFDocument3 paginiFlue Gas Cleaning PDFwilderness_666Încă nu există evaluări
- 1998 - Garrido - Nitrification of Wastes Waters Form Fish Meal FactoriswaterDocument6 pagini1998 - Garrido - Nitrification of Wastes Waters Form Fish Meal FactoriswaterJulio ReyesÎncă nu există evaluări
- Photocatalytic Concrete Stones Containing Tio For Atmospheric No RemovalDocument6 paginiPhotocatalytic Concrete Stones Containing Tio For Atmospheric No RemovalAshish WaliaÎncă nu există evaluări
- Kinetika Kimia: Bambang WidionoDocument77 paginiKinetika Kimia: Bambang WidionoFardaawÎncă nu există evaluări
- ARTIGO 4 - NBSP Influence of C DownloadDocument6 paginiARTIGO 4 - NBSP Influence of C DownloadCamila Eduarda Fernandes PiresÎncă nu există evaluări
- Air Pollution Control Technologies: VOC IncineratorsDocument51 paginiAir Pollution Control Technologies: VOC IncineratorsRay CÎncă nu există evaluări
- Numerical Modeling of Biodegradation: Analytical and Numerical Methods by Philip B. BedientDocument27 paginiNumerical Modeling of Biodegradation: Analytical and Numerical Methods by Philip B. Bedientsirilak1726Încă nu există evaluări
- Biochemical Reaction Engineering CHE 505Document42 paginiBiochemical Reaction Engineering CHE 505Haru MasaÎncă nu există evaluări
- CHM 212 Assignment DR AbdulwahabDocument2 paginiCHM 212 Assignment DR Abdulwahabfortress generator servicesÎncă nu există evaluări
- MQP Ans 02Document13 paginiMQP Ans 02riddhirpatankarÎncă nu există evaluări
- De Pra - 2012Document7 paginiDe Pra - 2012Doulalas GiorgosÎncă nu există evaluări
- Treatment of Landfill Leachate by Reverse Osmosis: Water Research February 1999Document7 paginiTreatment of Landfill Leachate by Reverse Osmosis: Water Research February 1999My Bloom StyleÎncă nu există evaluări
- Nitrification and Denitrification in the Activated Sludge ProcessDe la EverandNitrification and Denitrification in the Activated Sludge ProcessÎncă nu există evaluări
- Selected Constants: Oxidation–Reduction Potentials of Inorganic Substances in Aqueous SolutionDe la EverandSelected Constants: Oxidation–Reduction Potentials of Inorganic Substances in Aqueous SolutionÎncă nu există evaluări
- Fulltext 10542 PDFDocument112 paginiFulltext 10542 PDFkshtriya04Încă nu există evaluări
- Suspended Growth Biological TreatmentDocument21 paginiSuspended Growth Biological Treatmentsaiful islamÎncă nu există evaluări
- 1 s2.0 S2589014X21001808 Main 1Document27 pagini1 s2.0 S2589014X21001808 Main 1Granja ChiniÎncă nu există evaluări
- Toefl Itp Reading Comprehension Main Idea and Organization of TextDocument18 paginiToefl Itp Reading Comprehension Main Idea and Organization of Textsuprianto sitompulÎncă nu există evaluări
- 14.2 - Carbon - Cycle Nitrogen Cycle Save My ExmasDocument18 pagini14.2 - Carbon - Cycle Nitrogen Cycle Save My ExmasFalahath JayranÎncă nu există evaluări
- Activated Sludge Model No 3Document11 paginiActivated Sludge Model No 3jcbobedaÎncă nu există evaluări
- Groundwater Ecology: Developing A Method For Anammox Determination Using Natural Isotope Signature (Stephanye Zarama Alvarado)Document83 paginiGroundwater Ecology: Developing A Method For Anammox Determination Using Natural Isotope Signature (Stephanye Zarama Alvarado)S. ZaramaÎncă nu există evaluări
- Abstrat OrauxDocument174 paginiAbstrat OrauxSlim ToumiÎncă nu există evaluări
- Biological Nutrient RemovalDocument25 paginiBiological Nutrient RemovalŞeyda DağdevirenÎncă nu există evaluări
- Soil ScienceDocument12 paginiSoil ScienceSean HooeksÎncă nu există evaluări
- Denitrification: Denitrification Is A Microbially Facilitated Process of Nitrate Reduction That May Ultimately ProduceDocument4 paginiDenitrification: Denitrification Is A Microbially Facilitated Process of Nitrate Reduction That May Ultimately ProduceHanumant SuryawanshiÎncă nu există evaluări
- AquacultureDocument21 paginiAquaculturesunny0908Încă nu există evaluări
- Filtration and Reuse of Water in Fish Farming 1200dpiDocument39 paginiFiltration and Reuse of Water in Fish Farming 1200dpiJosé Antonio Retamal TosoÎncă nu există evaluări
- Wastewater Question Bank PDFDocument22 paginiWastewater Question Bank PDFLakshman IasÎncă nu există evaluări
- 8 Sutari Aeration Mixing SystemsDocument43 pagini8 Sutari Aeration Mixing SystemsTech ManagerÎncă nu există evaluări
- EVS CHAP 2 EcosystemDocument105 paginiEVS CHAP 2 EcosystemNida KhanÎncă nu există evaluări
- Biofilter Design PDFDocument119 paginiBiofilter Design PDFSumoharjo La MpagaÎncă nu există evaluări
- Experimental Methods in Wastewater TreatmentDocument362 paginiExperimental Methods in Wastewater Treatmentnacho100% (3)
- Integrated ScienceDocument32 paginiIntegrated ScienceGlenda Hernandez100% (2)
- Nitrogen: Nitrogen Measurements: Ammonium Nitrate NitriteDocument17 paginiNitrogen: Nitrogen Measurements: Ammonium Nitrate NitritealifbaharunÎncă nu există evaluări
- Science: Quarter 4 - Module 10: Roles of Organisms in The Cycling of MaterialsDocument20 paginiScience: Quarter 4 - Module 10: Roles of Organisms in The Cycling of MaterialsShiela Tecson GamayonÎncă nu există evaluări
- Nitrophoska Special BrochureDocument8 paginiNitrophoska Special Brochuresree778899Încă nu există evaluări
- Reduction of Nitrate by Zero Valent Iron (ZVI) - Based MaterialsDocument16 paginiReduction of Nitrate by Zero Valent Iron (ZVI) - Based MaterialsTatiana ArturiÎncă nu există evaluări
- Worksheet in Bio 102: Microbiology and Parasitology - Week No. 3Document4 paginiWorksheet in Bio 102: Microbiology and Parasitology - Week No. 3DELOS SANTOS JESSIECAHÎncă nu există evaluări
- Membrane Bioreactor For Mall Wastewater TreatmentDocument11 paginiMembrane Bioreactor For Mall Wastewater TreatmentJAVRÎncă nu există evaluări
- Design of Sewage Treatment Plant Based On SBR Technology: Tanveer Ali, Sapna Kumari, S.K AhmadDocument16 paginiDesign of Sewage Treatment Plant Based On SBR Technology: Tanveer Ali, Sapna Kumari, S.K Ahmadfoxtro_14Încă nu există evaluări
- Hardness of Water and FertilizersDocument10 paginiHardness of Water and FertilizersDennis NdoloÎncă nu există evaluări
- MBR Operators Workshop Draft PresentationsDocument79 paginiMBR Operators Workshop Draft PresentationsAbdallah GamalÎncă nu există evaluări
- Nutrient in Peaches PDFDocument15 paginiNutrient in Peaches PDFAnand Singh RawatÎncă nu există evaluări
- Biological Wastewater Treatment III - MBR Processes R1Document26 paginiBiological Wastewater Treatment III - MBR Processes R1NUR ASYIQIN BINTI AZAHARÎncă nu există evaluări