Documente Academic
Documente Profesional
Documente Cultură
External Calibration Method
Încărcat de
vanessa olgaDescriere originală:
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
External Calibration Method
Încărcat de
vanessa olgaDrepturi de autor:
Formate disponibile
Chem126 Lab Instrumental Analysis
EXPERIMENT 1
Spectrophotometric Determination of Fe2+ Ions using 1,
10-Phenanthroline
(External Calibration Method)
Vanessa Olga J. Dagondon and Ken M. Menez
Department of Chemistry, College of Arts and Sciences,
University of the Philippines Visayas, Miag-ao Iloilo
ABSTRACT
The determination of the Fe 2+ concentration in a branded iron
supplement tablet was developed using the external calibration
method. This method uses a series of standardized solutions to
obtain data through the UV VIS spectrophotometer to construct a
graphs that relate the wavelength to absorbance and, ultimately, to
the concentration of a solution. The standard used was a hydrated
derivative of ferrous sulphate, which is called ferrous ammonium
sulphate hexahydrate. Because of ferrous ions typically exist as
having a sulphate base in compounds; it was assumed that the iron
tablet contained mainly a high percentage of this ferrous sulphate.
This experiment also tackled to explain the principle as to how a
solution absorbs light, and how this could be related to the
concentration of ions in the solution. This experiment had also
provided first-hand experience to operating a UV VIS
spectrophotometer and practice to calculating necessary data
through the least squares method. This least squares method was
considered important at the event that an automatic calculating
device was not on hand. The principles followed by each property of
absorbance follow the Beers Law, which is reintroduced and
expanded in definition and use in this experiment. Lastly, since this
was a class experiment, the Fe 2+ concentration obtained by each
group was correlated and compared with each other in the whole
class. This was performed to verify the reliability of the method for
determining the said Fe2+ concentration.
INTRODUCTION
Spectrophotometric methods are based on the interaction of a
sample and of the light passing through it. These methods measure the
amount of light absorbed by the sample and thereby identifying the
amount or concentration of a part of the sample or of the sample which
interacts with it.
Page 1 of 10
Chem126 Lab Instrumental Analysis
The principle governing spectrophotometry is the Beer-Lamberts
Law or simply Beers law. According to this law, the absorbance of light is
directly proportional to the concentration and the pathlength or the
thickness of the sample.
A=bc
[1]
Equation [1] gives the mathematical relation of Beers law where A
is the proportionally constant or the molar
is the absorbance,
absorptivity, c is the concentration, and b is the pathlength of the
absorbing sample or species.
Spectrophotometer is the instrument used to measure the
absorbance of the sample at different concentrations. The basic
components of a spectrophotometer include the light sources, a
monochromator, sample holder, light detector and a read-out meter. An
analytical instrument such as a spectrophotometer must undergo
calibration so as its measured signal (for a spectrophotometer,
absorbance) will be free from determinate errors. Calibration is done
against a standard. Standardization is defined as the process of
determining the measured signal and the amount of analyte. A method is
standardized if the proportionality constant of the signal and amount of
analyte is known.
Many approaches in standardization are being used and improved to
lessen possible determinate errors that might occur in analytical
measurements. One of these standardization methods is the multi-point
external standard calibration method. This standardization method uses
two or more standards of known analyte prepared separately from the
samples containing the analyte. This is accomplished by constructing a
calibration curve. A calibration curve is the plot of the signal against the
concentration of the standards prepared. If the calibration curve is linear,
the slope of the line is the proportionality constant.
This experiment aims to use the multi-point external standard
calibration method in the spectrophotometric determination of Fe 2+ ions in
Ferrous sulfate tablets using 1, 10- Phenanthroline.
METHODOLOGY
A standard ferrous solution was formulated to be approximately 10
parts per million (milligrams per liter) by using a weighed amount of
standardized ferrous ammonium sulfate hexahydrate, or Fe (NH4)2 (SO4)2
6H2O. The solid was dissolved part-by-part before transferred into a one
liter volumetric flask, and dissolved with concentrated sulfuric acid before
being diluted to mark.
Page 2 of 10
Chem126 Lab Instrumental Analysis
A solution of 1, 10 phenanthroline was formulated from dissolving
a known weight of its solid form into a small beaker. This solution was
transferred into a plastic reagent bottle for storage.
Next, a solution of hydroxylamine hydrochloride was prepared
similarly to the phenanthroline solution, but was transferred into a brown
glass reagent bottle.
A buffer solution was needed in the experiment to allow a
permanent color change. A solution of 0.1 molar sodium acetate and 0.1
molar acetic acid were prepared, and a specified ratio was used to mix the
two solutions and form a buffer solution which was placed in a small
beaker.
A solution for the sample of ferrous sulfate, which naturally
appeared as ferrous ammonium sulfate or Fe (NH 4)2 (SO4)2, was prepared
by using a 325 milligram RiteMed iron tablet. This tablet was dissolved in
water and 6 molar hydrochloric acid. Additional heating was required to
dissolve the tablet completely. The solution was taken care as to not turn
brittle solid as this will make the sample invalid. However, due to
malpractice and inexperience with the experiment, the beaker containing
the sample solution was heated too high and produced an invalid product.
The group had to utilize a correctly prepared sample from another group.
Seven solutions of different volumes from the standard ferrous
solution were prepared and labeled. Another solution was taken from the
unknown sample solution, and a blank solution was prepared from distilled
water.
Each flask was added with a measured amount of each prepared
solution of hydroxylamine hydrochloride, 1, 10 phenanthroline, and the
acetic acid sodium acetate buffer solution. The solutions were allowed to
settle form a permanent color change.
The solution with the highest ppm concentration was used as the
basis for the maximum wavelength of all other solutions. The
spectrophotometer was used to measure this wavelength and the varying
absorbances obtained from each different solution contained in its
cuvette.
The molar absorption coefficient, denoted by , was determined
using the relation of A =
bc. The absorbance data taken from the non-
blank solutions were tabulated and graphed against the corresponding
concentrations, with absorbance as the y-value, to form the calibration
curve. This required for the sample concentration to be solved before
graphing.
The standard deviation was solved by using the x value taken from
the samples calibration curve and the calculated mean of the whole class.
Page 3 of 10
Chem126 Lab Instrumental Analysis
Then, the weight of iron present in the tablet was calculated using the
concentration of the sample multiplied by a number of dilution factors.
RESULTS
AND
DISCUSSION
The maximum wavelength was determined from the absorption
spectrum in Figure 1. The absorption spectrum was made by plotting the
absorbance against the wavelength. The maximum wavelength, or the
max, was determined to be 510 nanometers. The standard solution with
the highest concentration was chosen as it can absorb the most light out
of all the solutions. The wavelength component of the highest absorbance
is considered the maximum wavelength. As concentration is directly
proportional to absorbance, this maximum wavelength was used as the
basis for all the other solutions with lower concentrations.
0.45
0.4
0.35
0.3
0.25
Absorbane
0.2
0.15
0.1
0.05
0
400
-0.05
450
500
550
Wavelength (nm)
Page 4 of 10
600
650
Chem126 Lab Instrumental Analysis
Fig. 1. Absorption Spectrum
0.45
0.4
0.35
0.3
0.25
Absorbance
0.2
0.15
0.1
0.05
0
0
0.5
1.5
Concentration (ppm)
Fig. 2. Calibration Curve from the Standard Ferrous Ion Solution
Beers Law states that the absorbance transmitted by a substance is
directly proportional to its concentration in solution and path length of the
cuvette. This relationship is shown mathematically in Equation 1. Using
this relationship the proportionality constant or molar absorptivity,
denoted by , was calculated to be 0.1980 L/mg cm. The path length
Page 5 of 10
2.5
Chem126 Lab Instrumental Analysis
for the cuvette used has a constant value of 1.00 centimeter, which
effectively cancels its value from the Beers Law equation.
Fig. 2 shows the plot of the absorbance against the concentration of
the prepared standard ferrous solutions. This calibration curve describes
the linear relationship between the absorbance and concentration. The
slope of the line is the proportionality constant between the absorbance
and concentration of the standard solutions. The resulting slope is equal
to the molar absorptivity calculated using Beers Law. This also denotes
how the absorbance of a solution is directly proportional to its
concentration as a suggested in Equation 1. It was observed that as the
concentration of the analyte increases in each of the standard solution,
the color intensity also increases. This indicated a direct proportionality
between the two components.
The standardization technique used in the experiment was a multipoint external standard calibration method. It is widely used to analyze a
series of samples using a single calibration curve. In this technique the
samples matrix and that of the standard solutions matrix is assumed to
be the same and that would not affect the value of the proportionality
constant. However, this is not always the case and proportional
determinate error is introduced into the analysis. This is a limitation in this
kind of technique.
The equation of the line, obtained by least squares method, in the
calibration curve contained the value for the concentration of Fe 2+ in the
sample solution. This equation is in the form y = mx + b, where y
corresponds to the absorbance of the sample solution, m is the slope or
proportionality constant, b is the y intercept gained from the linear
regression process, and x is the concentration of the sample. The
concentration of the sample solution was then multiplied to a series of
dilution factors to obtain 61.7624 mg Fe2+.
The obtained concentration of ferrous ion in the sample is analyzed
statistically. Originally, triplicates of the sample should have been
analyzed however due to time and budget constraints only a single
sample was analyzed. The standard deviation from the results of the
calibration curve (sc) was calculated to measure the reliability of the result
of the external standard calibration. The standard deviation in the
regression line (sr), the relative standard deviation (RSD) was also
calculated. To measure the linearity of the calibration curve, R 2 was
calculated. Table 1 summarizes the statistics done in the groups sample.
The class standard deviation of the obtained concentration of the
ferrous ion in the sample was calculated. Also, the relative standard
deviation and coefficient of variance were determined to measure the
precision of measurements. Using Grubbs test on the concentrations of
Page 6 of 10
Chem126 Lab Instrumental Analysis
the five class sample solutions, there were no outliers detected in the
class data. Table 2 summarizes the statistics done on the class data.
CONCLUSION
The method used to standardize the sample was the external
standard calibration method.
The concentration of Fe2+ ion in the sample of 325 milligrams of
ferrous sulfate was calculated to be 61.7624 mg/tablet. The s c was equal
to 0.02235, representing reliability for the external standard calibration
method to produce data points with precision. The mean class data for the
concentration of the sample solution had an average of 61.7722
mg/tablet. The standard deviation for the whole class was calculated to be
1.9666. This suggests that there is a slight variation in the results relative
to the mean of the class data.
LITERATURE CITED
Harvey, D. May 2002. External Standards or Standard Additions? Selecting
and Validating a Method of Standardization. Journal of Chemical
Education Vol. 79: 613-615.
Harvey, D. Modern Analytical Chemistry. United State of America: The
McGraw-Hill Companies, Inc.; 2000 [cited 2016 February]. Available
from: http://elibrary.bsu.az/
Skoog D. A., West D. M., Holler F. J., Crouch S. R. 2014. Fundamentals of
Analytical Chemistry Ninth Edition. Canada: Nelson Education, Ltd.
1026p.
APPENDICES
x
5.6616
Sxx
x
7.8090
Syy
TABLES
y
1.1590
Sxy
3.2299
sr
0.1267
Xunknown
0.6394
sc
Page 7 of 10
y2
0.3186
Slope (m)
0.1980
RSD
xy
1.5769
Y
intercept
(b)
0.005448
R2
Chem126 Lab Instrumental Analysis
0.004041
61.7624
0.02235
0.0003618
Table 1. External Standard Calibration Method Group Data
xsample
ssample
sample
0.9994
RSDsample
308.8608
61.7722
1.9666
0.0318
Gmax
Gmin
Gexp
CV
1.0147
1.4148
1.5712
3.1836
Table 2. External Standard Calibration Method Class Data
CALCULATIONS
x= x i=0.1011+0.2022+0.3033+ 0.5055+ 1.011+1.5165+2.0220=5.6616
x 2= xi2 =0.0102+ 0.0408+0.0920+0.2555+1.0221+2.2998+ 4.0885=7.8090
y=y i=0.029+0.042+0.068+ 0.103+0.202+0.311+0.404=1.1590
2
y =y i = 0.000841 + 0.001764 + 0.004624 + 0.010609 + 0.040804
+ 0.096721 + 0.163216 = 0.318579
xy= 0.002932 + 0.008492 + 0.020624 + 0.05207 +
0.471632 + 0.816888 = 1.576857
S xx = x 2
( x )2
( 5.6616 )2
=7.8090
=3.2299
n
7
( y )2
( 1.1590 )2
S yy = y
=0.318579
=0.1267
n
7
2
( xy )2
[(5.6616)(1.1590)]
S xy =xy
=1.5769
=0.6394
n
7
m=
S xy 0.6394
=
=0.1980
S xx 3.2299
Page 8 of 10
0.204222 +
Chem126 Lab Instrumental Analysis
b=
y
x 1.1590
5.6616
m
=
( 0.1980 )
=0.005448
n
n
7
7
( )
y b
m
sr =
0.250.005448
=61.7624
0.1980
x unknown=
2
S yy m2 S xx
0.1267( 0.1980 ) (3.2299 )
=
=0.004041
n2
72
( m
S xx |=
RSD=
x unknown
R =1
0.25
1.1590
7
0.004041 1 1
+ +
=0.02235
0.1980 1 7 ( 0.19802 ) ( 3.2299 )
1 1
+ +
M N
sr
m
s c =
sc
y
n
0.02235
=0.0003618
61.7624
[ y i( b +m x i ) ]
S yy
=0.9994
x sample =58.9899+63.7677+63.5354 +61.0000+61.5678=308.8608
x sample =
x sample 308.8608
=
=61.7722
n
5
Page 9 of 10
Chem126 Lab Instrumental Analysis
( x ix )
( 58.989961.7722 )2+ ( 63.767761.7722 )2 + ( 63.535461.7722 )2+ ( 6161.7722 )2+ ( 6
s sample =
=
n
5
1.9666
RSD sample =
ssample 1.9666
=
=0.0318
x sample 61.7722
Gmax =
x max x sample 63.767761.7722
=
=1.0147
s sample
1.9666
Gmin =
x sample x min 61.772261.0000
=
=1.4148
s sample
1.9666
CV =RSD sample 100 =0.0318 100 =3.180
Gexp=
( )(
n1
n
) ( )(
CV 2
51
=
2
n2+CV
5
3.1802
=1.5712
52+3.1802
Page 10 of 10
S-ar putea să vă placă și
- Spectrometric Determination of The Acid Dissociation Constant of Methyl RedDocument11 paginiSpectrometric Determination of The Acid Dissociation Constant of Methyl Redエンパーラナー カイザー ニロÎncă nu există evaluări
- Experiment 7 - Classification Tests For HydrocarbonsDocument10 paginiExperiment 7 - Classification Tests For HydrocarbonsDanielle De GuzmanÎncă nu există evaluări
- Experiment 8 31.1Document28 paginiExperiment 8 31.1Jessa Libo-onÎncă nu există evaluări
- Lab chm301 Carboxylic AcidDocument7 paginiLab chm301 Carboxylic AcidbbbbÎncă nu există evaluări
- Gravimetric Determination of Moisture and Phosphorus in Fertilizer SamplesDocument6 paginiGravimetric Determination of Moisture and Phosphorus in Fertilizer SamplesDaphne MercadoÎncă nu există evaluări
- Structural Effects of ReactivityDocument4 paginiStructural Effects of ReactivityMarivic BarandaÎncă nu există evaluări
- PH and Buffer LabDocument13 paginiPH and Buffer LabAdellaine Lois GreyÎncă nu există evaluări
- Acid Base TitrationDocument12 paginiAcid Base TitrationMsfaeza HanafiÎncă nu există evaluări
- Complexometric Determination of Water Hardness Lab ReportDocument5 paginiComplexometric Determination of Water Hardness Lab ReportMichelle50% (2)
- Volumetric AnalysisDocument66 paginiVolumetric AnalysisAvan100% (1)
- Redox Titration Winkler Method For DissoDocument3 paginiRedox Titration Winkler Method For DissoJemimahÎncă nu există evaluări
- Laboratory Plan 1:: Standardization of Sodium Hydroxide (Naoh) With Potassium Hydrogen Phthalate (KHP)Document4 paginiLaboratory Plan 1:: Standardization of Sodium Hydroxide (Naoh) With Potassium Hydrogen Phthalate (KHP)Alliza Kaye CasullaÎncă nu există evaluări
- FULL REPORT Food Chemistry Food ColorantDocument10 paginiFULL REPORT Food Chemistry Food ColorantWong Su ZuanÎncă nu există evaluări
- Chem 31.1 Expt 2Document1 paginăChem 31.1 Expt 2Kelvin LabarezÎncă nu există evaluări
- EXPERIMENT 5 Common Ion EffectDocument4 paginiEXPERIMENT 5 Common Ion EffectNat DabuétÎncă nu există evaluări
- Exp 4Document9 paginiExp 4Nurul AqielaÎncă nu există evaluări
- PH and Buffers 7 NewDocument2 paginiPH and Buffers 7 NewVincent ManganaanÎncă nu există evaluări
- Expt 8 26.1Document7 paginiExpt 8 26.1Kyle CortezÎncă nu există evaluări
- Objective: The Objective of This Laboratory Is: - To Standardise of A Hydrochloric Acid (HCL) SolutionDocument12 paginiObjective: The Objective of This Laboratory Is: - To Standardise of A Hydrochloric Acid (HCL) SolutionShaker HusienÎncă nu există evaluări
- Chem 18.1 Experiment 6 Formal ReportDocument5 paginiChem 18.1 Experiment 6 Formal Reportlouize_1496Încă nu există evaluări
- Application of Statistical Concepts in The Determination of Weight Variation in Coin SamplesDocument3 paginiApplication of Statistical Concepts in The Determination of Weight Variation in Coin SamplesMicah PeraltaÎncă nu există evaluări
- Full Report Carbs On 161.1Document23 paginiFull Report Carbs On 161.1Kim Leonard BolandosÎncă nu există evaluări
- Quantitative Determination of Total Hardness in Drinking Water by Complexometric Edta TitrationDocument2 paginiQuantitative Determination of Total Hardness in Drinking Water by Complexometric Edta TitrationKeith SmithÎncă nu există evaluări
- Precipitation TitrationDocument3 paginiPrecipitation TitrationBanana SenpaiÎncă nu există evaluări
- Results and DiscussionsDocument4 paginiResults and DiscussionsAila RamosÎncă nu există evaluări
- RDR 6 Quantitative Determination of Oxalate by Permanganate TitrationDocument5 paginiRDR 6 Quantitative Determination of Oxalate by Permanganate TitrationAlyssa Bautista100% (2)
- CHM145L - Exp 1 - Melting Point and Boiling Point of Organic CompoundsDocument6 paginiCHM145L - Exp 1 - Melting Point and Boiling Point of Organic CompoundsBiancaTardecillaÎncă nu există evaluări
- Application of Statistical Concepts in The Determination of Weight Variation in SamplesDocument4 paginiApplication of Statistical Concepts in The Determination of Weight Variation in SamplesBulawan LaNeref Oremse100% (1)
- Exp 8 Data SheetDocument5 paginiExp 8 Data SheetBianca Bat-ogÎncă nu există evaluări
- ElectrophoresisDocument13 paginiElectrophoresisGIRMA SELALE GELETA100% (1)
- Lab Report 2 Alkane Alkene AlkyneDocument6 paginiLab Report 2 Alkane Alkene AlkyneNurAneesaÎncă nu există evaluări
- CHEM 334L - Conductance of Solutions - Estimating K For A Weak AcidDocument4 paginiCHEM 334L - Conductance of Solutions - Estimating K For A Weak Acidfdobonat613100% (1)
- Che21l - Experiment 2 - Complete Lab ReportDocument4 paginiChe21l - Experiment 2 - Complete Lab ReportMishel P. FerolinoÎncă nu există evaluări
- Buffer Preparation and PH Measurement Using The Electrometric Method and Colorimetric MethodDocument2 paginiBuffer Preparation and PH Measurement Using The Electrometric Method and Colorimetric MethodArndrei CunananÎncă nu există evaluări
- Application of Statistical Concepts in The Determination of Weight Variation in SamplesDocument3 paginiApplication of Statistical Concepts in The Determination of Weight Variation in SamplesLope Santos IVÎncă nu există evaluări
- Experiment No. 3 Determination of Acetic Acid Content in VinegarDocument14 paginiExperiment No. 3 Determination of Acetic Acid Content in VinegarClandy CoÎncă nu există evaluări
- Experiment 2 PDFDocument2 paginiExperiment 2 PDFJoone Xyron Creencia100% (1)
- EXPERIMENT NO 2 Separation of An Organic Mixture, Re Crystallization and Melting Point DeterminationDocument7 paginiEXPERIMENT NO 2 Separation of An Organic Mixture, Re Crystallization and Melting Point DeterminationJanina NemisÎncă nu există evaluări
- Determination of Acid Content of Vinegars and Wines Lab ReportDocument3 paginiDetermination of Acid Content of Vinegars and Wines Lab ReportAlleia Mae Urbano MazoÎncă nu există evaluări
- Chem 31.1 Post Lab 3Document61 paginiChem 31.1 Post Lab 3Belle AnasarioÎncă nu există evaluări
- EXercise 2 (Recrystallization and Melting Point Determination)Document3 paginiEXercise 2 (Recrystallization and Melting Point Determination)fangirltonÎncă nu există evaluări
- Gravimetric Determination of Moisture CoDocument5 paginiGravimetric Determination of Moisture CoDEFIN BIMA REYNANDAÎncă nu există evaluări
- Formal Report For Synthesis of An Alkyl HalideDocument5 paginiFormal Report For Synthesis of An Alkyl HalideLovelyn Marie Morada Nievales80% (5)
- Quantitative Determination of Soda Ash Composition by Double Indicator Titration PDFDocument6 paginiQuantitative Determination of Soda Ash Composition by Double Indicator Titration PDFHanzLouelLazaroFlorendoÎncă nu există evaluări
- Quantitative Determination of Dissolved Oxygen Content by Winkler Redox TitrationDocument5 paginiQuantitative Determination of Dissolved Oxygen Content by Winkler Redox Titrationneesan222Încă nu există evaluări
- Experiment No. 8 HydrocarbonsDocument7 paginiExperiment No. 8 HydrocarbonsMa Jessa DuntingÎncă nu există evaluări
- Expt On Solubility of Organic CompoundsDocument4 paginiExpt On Solubility of Organic CompoundsJasmine Juit100% (1)
- Quantitative Determination of Total Hardness of WaterDocument11 paginiQuantitative Determination of Total Hardness of WaterJoshua Oliveros50% (2)
- Experiment 13 Results and Discussion Report: Determination of Total Ion Concentration Using Ion Exchange ChromatographyDocument3 paginiExperiment 13 Results and Discussion Report: Determination of Total Ion Concentration Using Ion Exchange ChromatographyNathalie Dagmang100% (3)
- Test For PhenolsDocument3 paginiTest For PhenolsdocuAddictÎncă nu există evaluări
- Titration Solutions PDFDocument3 paginiTitration Solutions PDFBirmej NatapgasÎncă nu există evaluări
- Chem 31.1 Experiment 1 Lab ReportDocument5 paginiChem 31.1 Experiment 1 Lab ReportMara Krista CooÎncă nu există evaluări
- Atq 4Document4 paginiAtq 4Martina BlasÎncă nu există evaluări
- PREPARATION and STANDARDIZATION of HCL and NaOHDocument3 paginiPREPARATION and STANDARDIZATION of HCL and NaOHJoshua Williams Paramore100% (3)
- FST556 Preparation and Standardisation of Base and Acid SolutionDocument6 paginiFST556 Preparation and Standardisation of Base and Acid SolutionNurizzatiainiÎncă nu există evaluări
- Experiment 1 (Simple and Fractional Distillation)Document6 paginiExperiment 1 (Simple and Fractional Distillation)Cheng BauzonÎncă nu există evaluări
- Determination of Copper (II) Concentration by Colorimetric MethodDocument5 paginiDetermination of Copper (II) Concentration by Colorimetric MethodBulawan LaNeref Oremse100% (2)
- Optical Method of AnalysisDocument9 paginiOptical Method of AnalysisJohn Kevin San JoseÎncă nu există evaluări
- Spectrophotometric Determination of Iron in Aqueous Solutions As A Complex of 1,10-PhenanthrolineDocument4 paginiSpectrophotometric Determination of Iron in Aqueous Solutions As A Complex of 1,10-PhenanthrolineJaimie LojaÎncă nu există evaluări
- Thermometric Titrimetry: International Series of Monographs in Analytical ChemistryDe la EverandThermometric Titrimetry: International Series of Monographs in Analytical ChemistryÎncă nu există evaluări
- Principle: Composition of TSI AgarDocument10 paginiPrinciple: Composition of TSI Agarvanessa olga100% (1)
- Experiment 6 Glycogen Intro and MethodsDocument6 paginiExperiment 6 Glycogen Intro and Methodsvanessa olgaÎncă nu există evaluări
- Experiment 6 Glycogen Intro and MethodsDocument6 paginiExperiment 6 Glycogen Intro and Methodsvanessa olgaÎncă nu există evaluări
- TLC Lipids Lab ReportDocument12 paginiTLC Lipids Lab Reportvanessa olga100% (1)
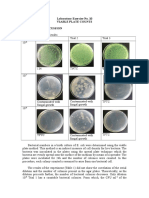
- Laboratory Exercise No. 10 Viable Plate Counts Results and DiscussionDocument3 paginiLaboratory Exercise No. 10 Viable Plate Counts Results and Discussionvanessa olga100% (1)
- Disinfectant 24h Incubation 48h Incubation 24h Incubation 48h Incubation BetadineDocument5 paginiDisinfectant 24h Incubation 48h Incubation 24h Incubation 48h Incubation Betadinevanessa olgaÎncă nu există evaluări
- Disinfectant 24h Incubation 48h Incubation 24h Incubation 48h Incubation BetadineDocument5 paginiDisinfectant 24h Incubation 48h Incubation 24h Incubation 48h Incubation Betadinevanessa olgaÎncă nu există evaluări
- 117 Report 3 Ternary DiagramDocument20 pagini117 Report 3 Ternary Diagramvanessa olga86% (7)
- Discovery Phase Hand OutDocument4 paginiDiscovery Phase Hand Outvanessa olgaÎncă nu există evaluări
- PVOH Chain LinkageDocument23 paginiPVOH Chain Linkagevanessa olga100% (2)
- Methyl RedDocument18 paginiMethyl Redvanessa olgaÎncă nu există evaluări
- Proximate AnalysisDocument16 paginiProximate Analysisvanessa olgaÎncă nu există evaluări
- Analysis of Permanganate - Dichromate MixtureDocument16 paginiAnalysis of Permanganate - Dichromate Mixturevanessa olga100% (4)
- Chemical Interferences in Atomic Absorption Spectrophotometric MeasurementsDocument15 paginiChemical Interferences in Atomic Absorption Spectrophotometric Measurementsvanessa olga100% (2)
- Integration Topical Test SolutionsDocument2 paginiIntegration Topical Test SolutionsLee Hue KeanÎncă nu există evaluări
- RepeatedeigsDocument7 paginiRepeatedeigsSaurabh YadavÎncă nu există evaluări
- Investigating Acid MixtureDocument5 paginiInvestigating Acid MixtureanisaedionÎncă nu există evaluări
- Finite Element Method: Beam AnalysisDocument10 paginiFinite Element Method: Beam AnalysisZaffira RahmanÎncă nu există evaluări
- 1 Decision ScienceDocument192 pagini1 Decision ScienceAbu MusaÎncă nu există evaluări
- CBSE Class 9 Mathematics Worksheet - PolynomialsDocument8 paginiCBSE Class 9 Mathematics Worksheet - PolynomialsSandya100% (4)
- Elementary Differential Equations With Boundary Value Problems - Pr. Trench PDFDocument806 paginiElementary Differential Equations With Boundary Value Problems - Pr. Trench PDFmnunez1995100% (1)
- Derivatives and Integrals FormulaDocument1 paginăDerivatives and Integrals FormulaPaul Daryl CutamoraÎncă nu există evaluări
- HKDSE Mathematics (Module 1) Topic 5: Indefinite IntegrationDocument6 paginiHKDSE Mathematics (Module 1) Topic 5: Indefinite IntegrationDexter Fung100% (1)
- ANOVA ExampleDocument6 paginiANOVA ExampleLuis ValensÎncă nu există evaluări
- Domain of Dependence and Range of InfluenceDocument16 paginiDomain of Dependence and Range of InfluenceMilind JainÎncă nu există evaluări
- Time HistoryDocument7 paginiTime HistoryshivamomshantiÎncă nu există evaluări
- Network DiagramDocument17 paginiNetwork DiagramJohn Robert GabrielÎncă nu există evaluări
- Decision-Making Based On Data AnalyticsDocument3 paginiDecision-Making Based On Data AnalyticsJMFMÎncă nu există evaluări
- Polytechnic Mathematics Vol - II: December 2011Document17 paginiPolytechnic Mathematics Vol - II: December 2011Chadaram Jagadish JagadishÎncă nu există evaluări
- NOVA 2.1.2 User Manual PDFDocument1.196 paginiNOVA 2.1.2 User Manual PDFMoab Souza0% (1)
- Binomial Expansion Practice Problems and MarkschemeDocument6 paginiBinomial Expansion Practice Problems and MarkschemeMohammed N TarmalÎncă nu există evaluări
- Dmaic QuizDocument6 paginiDmaic QuizARABELLA MALLARIÎncă nu există evaluări
- Table of ContentsDocument10 paginiTable of ContentsSanam RaiÎncă nu există evaluări
- STAT 112 20172S - y ExamWeek 10 - Quarterly Exam - QuarterlDocument4 paginiSTAT 112 20172S - y ExamWeek 10 - Quarterly Exam - QuarterlArmanÎncă nu există evaluări
- Quadratic Functions Assignment: Number 2Document3 paginiQuadratic Functions Assignment: Number 2EveÎncă nu există evaluări
- Optimization of Chemical Processes: Course Code: 13CH1136 L TPC 4 1 0 3Document2 paginiOptimization of Chemical Processes: Course Code: 13CH1136 L TPC 4 1 0 3victorÎncă nu există evaluări
- Chapter 05 W7 L1 Random Sample 2015 UTP C5Document8 paginiChapter 05 W7 L1 Random Sample 2015 UTP C5Jun Hao HengÎncă nu există evaluări
- Quantitative Methods MM ZG515 / QM ZG515: Handout and IntroductionDocument16 paginiQuantitative Methods MM ZG515 / QM ZG515: Handout and IntroductionviswanathÎncă nu există evaluări
- Solutions To Difference EquationsDocument22 paginiSolutions To Difference EquationsKenneth LandichoÎncă nu există evaluări
- Nptel: Mathematical Physics - 1 - Web CourseDocument2 paginiNptel: Mathematical Physics - 1 - Web CourseRahul SharmaÎncă nu există evaluări
- Stats1 Chapter 2::: Measures of Location & SpreadDocument53 paginiStats1 Chapter 2::: Measures of Location & SpreadLuna LedezmaÎncă nu există evaluări
- Socratic Seminar Template and SamplesDocument11 paginiSocratic Seminar Template and SamplesNazar RusliÎncă nu există evaluări
- PHD Course: Critical Analysis of Political DiscourseDocument4 paginiPHD Course: Critical Analysis of Political DiscourseRed RoseÎncă nu există evaluări
- Comandos Usados en MLDocument17 paginiComandos Usados en MLJohn DannyÎncă nu există evaluări