Documente Academic
Documente Profesional
Documente Cultură
Huntington PDF
Încărcat de
Madalina Ionela StanciuTitlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Huntington PDF
Încărcat de
Madalina Ionela StanciuDrepturi de autor:
Formate disponibile
Personal View
Beyond the brain: widespread pathology in
Huntingtons disease
Jorien MM van der Burg, Maria Bjrkqvist, Patrik Brundin
Huntingtons disease (HD) is an inherited neurodegenerative disorder caused by a polyglutamine stretch in the
huntingtin protein. Today, more than 15 years after the genetic defect underlying HD was discovered, the pathogenesis
is still not well understood and there is no adequate treatment. Research into this disorder has conventionally focused
on neurological symptoms and brain pathology, particularly neurodegeneration in the basal ganglia and cerebral
cortex. Mutant huntingtin is, however, ubiquitously expressed throughout the body. Indeed, contrary to earlier
thinking, HD is associated with abnormalities in peripheral tissues. These abnormal changes are not all secondary to
brain dysfunction, but most seem to be directly caused by expression of mutant huntingtin in peripheral tissues. In
this article, we highlight this emerging eld of research and how it might aect our understanding of the pathogenesis
of this disease, the development of novel biomarkers of disease progression, and the identication of new potential
treatments.
Introduction
Huntingtons disease (HD) research has conventionally
focused on the brain for obvious reasons: HDs core
symptoms include motor abnormalities such as
hyperkinesia and hypokinesia, as well as psychiatric and
cognitive problems (the panel provides an overview of
HD). These symptoms have all been linked to
neurodegeneration in the basal ganglia and cerebral
cortex.
In addition to the classic symptoms, HD is complicated
by other features, such as weight loss and skeletal-muscle
wasting, which are not necessarily directly associated
with changes in brain functions (gure 1). These
features sometimes appear early in the disease course
and can eventually contribute substantially to both
morbidity and mortality. These symptoms have been
suggested to be secondary to general sickness or to be
the consequences of neurological dysfunction. However,
a growing body of evidence indicates that these changes
might be due to the direct eect of mutant huntingtin
(the toxic protein that causes HD) on peripheral tissues.
This thinking is largely derived from recent studies in
animal models of HD, but the fact that similar
abnormalities also occur in peripheral tissues of
patients is gradually becoming evident. In this article,
we propose that an improved understanding of changes
in peripheral tissues in HD could give insight into the
pathogenesis of the disease, lead to discovery of novel
biomarkers of disease progression, and could open new
avenues for treatment.
HD is not only a brain disorder
Several facts indicate that peripheral features of HD,
such as weight loss and skeletal-muscle wasting, might
have little or nothing to do with neurological dysfunction
or general sickness. First, huntingtin is expressed in
many tissues and organs in human beings and other
mammals.24 The precise function of huntingtin is
unclear, but this protein seems to be involved in several
cellular processes, including transcriptional events,
www.thelancet.com/neurology Vol 8 August 2009
protein tracking, and vesicle transport.5 Studies in
knock-out mice have revealed that absence of huntingtin
leads to defects in all three germ layers and is
embryonically lethal,6 indicating that huntingtin is
essential for many tissues. Indeed, knocking down
huntingtin expression in zebrash results in peripheral
defects, such as impaired haemoglobin production.7
Second, mutant huntingtin seems to aect organelles
and functional systems that are essential to all cells (eg,
mitochondria, the ubiquitinproteasome system,
caspases, and chaperones). As a result, cells from
peripheral tissues from patients with HD and from
animal models have, for example, mitochondrial
dysfunction817 and transcriptional defects.1822 Moreover,
intracellular huntingtin aggregatesthe hallmark of HD
neuropathologyare present in peripheral organs of
transgenic mice with HD.2325 Third, dysfunction of
peripheral cells also occurs when these cells are
isolated,1416,26,27 indicating that this eect is a direct
consequence of endogenously expressed mutant
huntingtin and is not secondary to brain dysfunction. For
example, monocytes isolated from HD gene carriers,
which express mutant huntingtin, are pathologically
hyperactive in response to stimulation from
lipopolysaccharide.26 Fourth, when mutant huntingtin is
expressed only in cardiomyocytes of wild-type mice, the
animals develop heart failure.28,29
The above observations all support the hypothesis that
peripheral abnormalities in HD could be caused directly
by local expression of mutant huntingtin and could occur
independently from neurological defects or general
malaise.
Lancet Neurol 2009; 8: 76574
Neuronal Survival Unit,
Department of Experimental
Medical Science, Wallenberg
Neuroscience Center,
Lund University BMC A10,
Lund, Sweden
(J M M van der Burg PhD,
M Bjrkqvist PhD, P Brundin MD)
Correspondence to:
Jorien MM van der Burg,
Neuronal Survival Unit,
Department of Experimental
Medical Science, Wallenberg
Neuroscience Center, Lund
University BMC A10,
221 84 Lund, Sweden
jorienvanderburg@gmail.com
Non-neuronal abnormalities might contribute
to symptoms
In this section we discuss in more detail how
abnormalities in peripheral tissues could underlie various
signs and symptoms of HD. Patients with this disorder
have several non-neurological features, including weight
loss and skeletal-muscle wasting. Although less
765
Personal View
Genetic disorder
Autosomal dominant neurodegenerative disorder
thoroughly investigated, glucose intolerance, osteoporosis, cardiac failure, gastrointestinal abnormalities,
and testicular atrophy might also belong to the HD
phenotype in aected patients (gure 1). These features
of the disease are clinically important as they reduce
quality of life and, in some cases, correlate with disease
progression and contribute to early death.
Epidemiology
48 per 100 000
Weight loss
Panel: Overview of Huntingtons disease*
First accurate description
In 1872 by George Huntington
Onset
First symptoms can start at any age, but mean age at onset
is between 35 and 45 years
Clinical presentation
Classic neurological symptoms
Motor symptoms
Cognitive deterioration
Psychiatric and behavioural problems
Other clinical presentation features
Weight loss
Atrophy of skeletal muscle
Sleep disturbance
Autonomic disturbances
Cause
A mutation in HTT causes the formation of the mutant
protein huntingtin. Normal individuals have less than 36 CAG
repeats in HTT. Individuals with >39 CAG repeats will develop
Huntingtons disease. Reduced penetrance occurs with
3639 CAG repeats.
Pathogenesis
The role of normal huntingtin is not completely clear, but
when mutated it causes, for example, the formation of
intracellular inclusion bodies, impaired intracellular transport,
defected gene transcription, and apoptosis.
Genetic animal models
Some of the most widely studied mouse models
R6/2: expresses exon 1 of human mutant HTT with about
150 CAG repeats
YAC 72: expresses full-length human mutant HTT with
72 CAG repeats, created using a yeast artical
chromosome as vector
YAC 128: expresses full-length human mutant HTT with
128 CAG repeats, created using a yeast artical
chromosome as vector
N17182Q: expresses the 171 amino acids at the
N-terminal of mutant huntingtin with 82 CAG repeats
HdhQ92: model with 92 CAG repeats knocked in into the
mouse Htt
Other genetic animal models
Caenorhabditis elegans models
Drosophila melanogaster models
Transgenic rhesus macaques
*A comprehensive overview of HD is provided by Walker and colleagues.1
766
One of the most common peripheral abnormalities that
aects nearly all individuals with HD, and which is not
necessarily associated with pathology of the nervous
system, is unintended weight loss. This weight loss is
progressive, beginning as minor loss in presymptomatic
gene carriers and ending with profound cachexia in
advanced-stage patients,3038 and correlates with CAG
repeat number.39 Why patients with HD lose weight is
not known. Many studies have indicated that this feature
is not secondary to hyperactivity30,34,38 or anorexia,30,35,38,40
but results from an increased metabolic rate.38,41,42 Patients
with HD with a higher body mass index at onset of
symptoms tend to have a slower rate of disease
progression,43 suggesting that weight loss could be a
valuable target for therapeutic intervention.
According to numerous studies, most of which in
transgenic mouse models, several tissues and organs
that are important in weight regulation might be aected
in HD (gures 2 and 3). For example, the digestive
system, which is important for nutrient absorption, is
dysfunctional. Patients suer from xerostomia,44 which
could aect taste, mastication, and swallowing, all of
which might contribute to weight loss. In the R6/2 mouse,
the most widely studied transgenic animal model of HD,
the number of ghrelin-producing neurons is reduced in
the stomach.41 Ghrelin normally stimulates food intake
and inhibits energy expenditure; therefore, a loss of this
hormone might contribute to weight loss. The pancreas
is also aected and both patients with HD and mice
models of the disease tend to develop impaired glucose
tolerance.4551 In a recent study by Lalic and colleagues,52
an impairment in insulin-secretion capacity alongside a
decrease in insulin sensitivity was seen in patients with
HD compared with control individuals. In mice,
pancreatic islet cells are atrophic and exhibit intranuclear
inclusions,50,53 resulting in defects in insulin, somatostatin,
and glucagon production.54 In patients, although islets
are of normal size, transcriptional dysregulation in islet
cells might underlie the increased tendency to develop
impaired glucose tolerance.55 One study indicated that
mutant huntingtin might also aect the liver.
Transcription of urea-cycle enzymes is impaired in the
liver of mice with HD, leading to high circulating
concentrations of ammonia with concomitant brain
damage and locomotor dysfunction.56 A low protein diet
reduced ammonia concentrations in the plasma, and
ameliorated brain damage and locomotor function in
these mice.56
www.thelancet.com/neurology Vol 8 August 2009
Personal View
In addition to the digestive system, adipocytes might
be aected in HD. Endocrine signals from white adipose
tissue, which normally reect energy levels within the
body, are aected in two HD mouse models.57,58 Expression
of fat-storage genes and concentrations of the adipokine
hormones leptin and adiponectin are impaired in these
mice, which might contribute to weight loss.58 These
transcriptional defects are a direct consequence of mutant
huntingtin expression in the mouse adipocytes and can
be replicated in an adipocyte cell line.58
Skeletal-muscle atrophy
Skeletal-muscle wasting is another hallmark of HD.31,32,35,59
The mechanisms underlying muscular atrophy in this
disorder are unclear and, of note, the skeletal muscles
undergo substantial wasting, despite the muscles being
highly active as a result of hyperkinesia.
The muscle wasting is possibly due to defects caused by
the presence of mutated protein in myocytes. Mutant
huntingtin forms inclusion bodies in muscle cells of
R6/2 mice25 and disrupts gene expression similarly to its
eect in the brain.18,19,22 This nding suggests that similar
pathogenetic mechanisms are operative in neurons and
muscle cells. Apart from aecting transcriptional
regulation, mutant huntingtin is known to aect mitochondrial function.60 Studies on cultures of skeletal-muscle
cells from patients with HD revealed several mitochondrial
abnormalities, including respiratory-chain dysfunction,
morphologically abnormal cristae, cytochrome c release,
and apoptosis.6163 In vivo studies also revealed altered
muscle energy metabolism in this disorder.10
Hence, myocytes are aected in HD, possibly as a
direct eect of mutant huntingtin, and muscles undergo
atrophy as a result. This nding opens up new possible
treatment strategies and novel ways of monitoring
disease progression.
Cardiac failure
Cardiac failure occurs in about 30% of patients with HD
(compared with only 2% in age-matched controls) and is a
leading cause of death in these patients.64 Nothing is
known about the pathophysiological mechanism
underlying cardiac failure in this disorder, but failure of
the autonomic nervous system might play a part.65
Alternatively, cardiac failure might be caused by the eects
of mutant huntingtin expression in cardiomyocytes. In
R6/2 mice, the myocardium is atrophic, mitochondria in
cardiomyocytes have abnormal shapes, and cardiac output
is reduced by 50%.66 Cardiomyocyte-specic expression of
polyglutamine fragments with 83 CAG repeats in wild-type
mice induces aggregate formation, autophagy, and
necrotic death of cardiomyocytes, eventually leading to
heart failure.28,29 Taken together, these experimental data
indicate that heart failure in HD could be a direct
consequence of mutant huntingtin expression in
cardiomyocytes, and clinical studies on cardiac tissue of
patients are needed to resolve this matter.
www.thelancet.com/neurology Vol 8 August 2009
Muscle atrophy
Impaired glucose
tolerance
Cardiac failure
Weight loss
Osteoporosis
Testicular atrophy
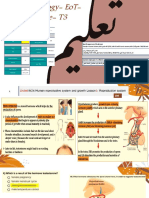
Figure 1: Non-neurological features of Huntingtons disease
Testicular atrophy
The highest levels of huntingtin expression are found in
the brain and testes.3,67 Although fertility is unaected in
patients with HD,68 men have reduced testosterone
concentrations69 as well as testicular pathology with
reduced numbers of germ cells and abnormal seminiferous
tubule morphology.70 Reduced plasma concentrations of
testosterone correlate with worsening of the disease.69,71
Similarly, both the R6/2 and YAC128 mouse models of HD
have testicular atrophy, albeit to a greater extent than
patients.70,7275 In R6/2 mice, concentrations of gonadotropinreleasing hormone are reduced, which suggests that the
eects might be secondary to hypothalamic dysfunction.73
In YAC128 mice, however, testicular degeneration develops
before any evidence of decreased testosterone
concentrations or loss of gonadotropin-releasing hormone
neurons in the hypothalamus, suggesting that testicular
pathology results from a direct toxic eect of mutant
huntingtin in the testes.70
Osteoporosis
Data from two preliminary reports suggest that
osteoporosis might also be part of the HD phenotype76,77
and that its severity correlates with the number of CAG
767
Personal View
Skeletal muscle
Cardiovascular system
Blood
Morphological
changes
Heart rate
variability
Altered gene
transcription
Mitochondrial
dysfunction
Altered cerebral
blood ow
Mitochondrial
dysfunction
ATP production
Caspase activity
Altered gene
transcription
Inammatory
markers
(eg, interleukin 6)
Skeleton
Digestive tract
Insulin
production
Osteoporosis
Altered gene
transcription in
islet cells
Genital system
Thyroid
Testosterone
(men only)
Levels of T3
Number of
spermatids
Diameter of
seminiferous
tubules
Figure 2: Peripheral pathology in patients with Huntingtons disease
repeats.77 The reason for decreased bone mineral density
in this disorder is not known; it might be secondary to
neuroleptic treatment, might result from immobility, but
could also be a direct eect of the disorder itself. For
example, decreased bone mineral density can result from
disrupted endocrine signalling, such as increased cortisol
concentrations. R6/2 mice have decreased bone density,
along with increased concentrations of cortisol, wasting
of skeletal muscle, accumulation of abdominal fat, and
insulin resistance, similar to a Cushing-like syndrome.78
These ndings led to the observation of increased urinary
concentrations of cortisol in patients with HD.78 Recent
studies have conrmed that there are increased cortisol
concentrations in these patients,71,79 but alterations are
probably too small to cause a Cushing-like syndrome and
osteoporosis. Instead, osteoporosis in HD might be
caused by a direct eect of mutant huntingtin on
osteoblasts or osteoclasts in bone tissue. However, this
hypothesis is highly speculative and further investigations
are needed to clarify the underlying cause of decreased
bone mineral density. Studies comparing very active and
768
immobile patients could, for example, clarify what role
immobility has.
Dysfunction of blood-derived cells
Blood cells are aected in many ways in HD.
Haematocytes have altered gene transcription,20,21 caspase
activity,80 and mitochondrial function.8,9,11,13 Additionally,
adenosine A2A receptor function81,82 and monoamine
oxidase activity83 are disrupted.
Blood plasma samples of patients with HD show signs
of immune activation, such as increased concentrations
of interleukins 8 and 6.26,84 Altered inammatory signalling
might contribute to several features of HD, including
weight loss and muscle wasting. Proinammatory
cytokines are associated with muscle atrophy, exerted
through inhibition of myogenic dierentiation and
enhanced apoptosis.85,86 Moreover, these cytokines can
aect energy metabolism and body weight.87
The immune system in HD might be activated in
reaction to cellular pathology, such as aggregate
formation, oxidative damage or necrotic cell death.
www.thelancet.com/neurology Vol 8 August 2009
Personal View
Skeletal muscle
Cardiovascular system
Blood
Morphological
changes
Cardiac output
Altered gene
transcription
Mitochondrial
dysfunction
Protein nitration
Mitochondrial
dysfunction
ATP production
Thickness of myocardium
Altered gene
transcription
Inammatory
markers
(eg, interleukin 6)
Caspase activity
Bone
Bone density
Digestive tract
Inclusion bodies
Loss of
ghrelin-producing
neurons
White adipose tissue
Altered
transcription of
fat-storage genes
Inclusion bodies
Leptin and
adiponectin
Urea cycle
deciency
Genital system
Testosterone
and GnRH
Testicular atrophy
Dysorganised
seminiferous
epithelium
Insulin, glucagon,
somatostatin
production
Inclusion bodies
Figure 3: Peripheral pathology in animal models of Huntingtons disease
However, concentrations of IL-6 are already increased as
early as 16 years before the predicted onset of symptoms.26
Moreover, as mentioned earlier, isolated monocytes from
patients with HD and asymptomatic mutant HTT gene
carriers express mutant huntingtin and are pathologically
hyperactive in response to stimulation.26 These
observations all suggest that inammatory changes in
this disease result from dysfunction of immune cells and
are not secondary to brain pathology or general sickness.
Are changes in peripheral tissue secondary to
brain dysfunction?
There are many peripheral abnormalities in HD that seem
to occur independently from neuronal dysfunction.
However, brain pathology, for example in the hypothalamus,
could lead to altered endocrine signalling and secondary
changes in peripheral tissues. Several neurodegenerative
disorders, including Alzheimers disease, can also be
complicated by peripheral abnormalities,88 such as weight
loss, which could suggest a role of the brain in causing
these features. The hypothalamus is aected in patients
www.thelancet.com/neurology Vol 8 August 2009
with HD and in transgenic mice,89,90 and its pivotal role in
endocrine regulation means that the pathology restricted
to this brain structure can aect the whole body. Changes
have been found in several of the hypothalamicpituitary
axes and include altered concentrations of luteinising
hormone,69 growth hormone,71 and cortisol.78,79 Peptides
that could possibly be linked to caloric intake and weight
loss are also aected in this disorder.91,92 In the hypothalamus
of HD mice, the expression of vasopressin, oxytocin,
cocaine, and amphetamine-regulated transcript peptide
(CART),93 gonadotropin-releasing hormone,73 and vasoactive intestinal polypeptide and its receptor94 are reduced.
Despite multiple changes in the hypothalamic or
pituitary control of endocrine functions, results from
various studies indicate that brain dysfunction might not
be involved in the genesis of many peripheral abnormalities
in HD. Decreased concentrations of testosterone in
patients with HD do not correlate with decreased
concentrations of luteinising hormone.69 On the contrary,
patients with lowest testosterone concentrations have
normal concentrations of luteinising hormone,69
769
Personal View
suggesting that altered testosterone production is not
caused by defects in hypothalamicpituitary signalling.
Mice with HD can also exhibit testicular pathology without
evidence of disrupted hypothalamicpituitary signalling.70
Additionally, changes in hypothalamic peptides regulating
energy metabolism and body weight were not clearly
linked to weight loss in HD mice;41 neither did decreased
vasopressin concentrations lead to increased water
consumption in these mice.44 A study in functioning of the
hypothalamicpituitary axes in patients with HD was also
unable to link several observed peripheral abnormalities to
disrupted neuroendocrine signalling.71 Finally, some
peripheral alterations are detectable many years before
neuronal pathology is evident and neurological symptoms
are present,26 suggesting that the central and peripheral
changes are not linked.
Taken together, these ndings suggest that peripheral
abnormalities do not necessarily arise from altered
hypothalamicpituitary signalling, but are due to primary
defects in peripheral organs. Contrary to previous
thinking, peripheral defects might even contribute to
brain pathology.56,95 Therefore, HD can be viewed as a
systemic disorder, in which many organs and tissues are
aected and changes in one organ trigger malfunction in
another. To unravel the precise role of each of these
organs and tissues in this disease, animal models that
selectively express mutant huntingtin in certain organs
or brain regions, such as the mouse model with
cardiomyocyte-specic expression of mutant huntingtin,29
might be helpful.
Understanding HD by looking into peripheral
tissues
Having described the many peripheral abnormalities
and their possible origins, we now discuss how
discoveries of these defects might help to resolve
outstanding problems in HD. Investigations of
peripheral abnormalities could lead to better understanding of the pathogenesis, development of novel
therapies, and the discovery of biomarkers of disease
progression. Peripheral tissues are typically easier to
study than the CNS. Sampling of peripheral tissues (eg,
blood cells) can be minimally invasive and inexpensive
compared with techniques to study brain function, such
as advanced brain imaging. Furthermore, peripheral
cells can be isolated from living patients, enabling the
study of pathological mechanisms in cells aected by
HD throughout the disease course.
Cellular mechanisms underlying peripheral pathology
Studies in peripheral tissues now substantially contribute
to our understanding of HD pathogenesis.11,15,19,20,81,82,96,97
Changes in peripheral cells, such as mitochondrial
dysfunction,11,15 cholesterol defects,9799 and transcriptional
alterations,18,19,22 often resemble abnormalities in the brain
and provide valuable insight for disease mechanisms.
For example, a recent study in lymphocytes from patients
770
with HD detected altered transcription of neuronal
RE1/NRSE (repressor element-1/neuron restrictive
silencer element), which had only been described in
post-mortem HD brains, but also occurs in vivo.100
Conversely, studies in peripheral tissues have led to
ndings that have resulted in the realisation of similar
changes in brain tissue.
Transcriptional and mitochondrial defects are
important pathogenetic mechanisms. These defects are
involved in both peripheral and CNS pathology in this
disease and could explain many symptoms. A
transcriptional defect that could be accountable for
several clinical features of HD is the altered expression of
PPAR coactivator 1 (PGC-1). PGC-1 regulates
mitochondrial biogenesis and respiration, and is
downregulated in several tissues of patients with HD,
including brain, muscle, and fat tissue.22,58,101,102 In the
brain of knock-in HD mice (with 140 CAG repeats
inserted into the murine huntingtin gene), expression of
PGC-1 is reduced in medium-sized spiny neurons, but
increased in cholinergic interneurons.101 Therefore,
changes in PGC-1 might be linked to the selective
vulnerability of medium-sized spiny neurons that is
characteristic of this disease. Decreased expression of
PGC-1 might underlie several other HD-associated
changes. For example, this change might be linked to
cardiomyopathy in HD mice, 66 as the myocardium largely
depends on mitochondrial energy and expresses high
concentrations of PGC-1. Other organs that highly
depend on ATP, such as the kidneys, might also be
aected in HD. However, to our knowledge, a study into
this possibility has not been done.
Taken together, peripheral abnormalities often
resemble brain pathology, and certain pathogenic
mechanisms, such as transcriptional dysregulation and
mitochondrial dysfunction, could underlie many
symptoms of HD.
Novel targets for therapeutic interventions
The study of peripheral abnormalities in HD has resulted
in the discovery of novel potential therapeutic targets.
The treatment of liver and pancreas defects ameliorated
brain pathology and neurological symptoms in mice
with HD.56,95 Furthermore, treatment of non-neurological
symptoms could improve quality of life and postpone
premature death. A starting point for a novel therapy
might be the observation that a higher body mass index
is associated with a slower progression of disease.43 A
possible cause-and-eect association between increased
body weight and slower progression of neurological
symptoms in HD is not yet clear. It would be interesting
to examine whether taking measures to increase body
mass index slows down the progression of neurological
symptoms. Trejo and co-workers36 showed that a higher
caloric intake stabilises body weight in patients with HD,
but they did not specically address whether disease
progression was aected. With regard to possible eects
www.thelancet.com/neurology Vol 8 August 2009
Personal View
of treating impaired glucose tolerance in HD, animal
studies have provided some information. Chronic
treatment with glibenclamide, a hypoglycaemic drug
that induces release of insulin, does not improve the
disease or the lifespan in R6/2 mice.53 By contrast,
metformin does increase lifespan and reduces motor
symptoms such as hind-limb clasping in male
R6/2 mice.103 The antidiabetic glucagon-like peptide 1
receptor agonist exendin-4 also exerts benecial eects
in R6/2 mice; this drug suppresses pathology in the
pancreas and the brain, leading to improved motor
function and survival.95 However, the precise mechanism
of action of exendin-4 in these experiments is not clear.
This drug also exerts anti-inammatory eects and
promotes brain plasticity, which could support functional
recovery.104
Strategies to counteract cardiac failure in HD could
possibly both improve quality of life and prevent early
death. In future studies targeting cardiomyopathy in this
neurodegenerative disease, non-invasive ultrasonography
could provide immediate and objective feedback with
regard to possible treatment eects. Treatment of other
peripheral abnormalities, including testicular degeneration has, to our knowledge, never been attempted.
Decreased concentrations of testosterone are generally
associated with decreased libido, osteoporosis, and
wasting of skeletal muscle. These signs and symptoms
have all been detected in either patients or mice with
HD.31,35,72,73,76,77 Reduced testosterone concentrations in the
blood correlate with disease severity, as measured by
motor and functional scores.71 Moreover, low concentrations of testosterone have been associated with
neuronal loss,105 which could possibly contribute to HD
neurodegeneration. Patients might therefore benet
from hormonal replacement therapy.
Discovery of new biomarkers
Biomarkers to monitor or predict disease progression in
HD are needed to assess therapeutic responses during
clinical trials. Assessment of symptoms is typically not
suciently sensitive, because of the protracted course of
the disease, and the paucity of rigorous objective tools to
assess clinical changes means that there is a risk for
observer bias. Moreover, there are no methods to assess
disease-associated changes in gene carriers who do not
have
overt
symptoms.106
Unlike
most
other
neurodegenerative diseases, every patient with HD carries
the mutation and everyone with the mutation will develop
the disease. HD therefore oers unique possibilities to
detect early changes of disease course. Direct measures of
peripherally manifested symptoms, such as xerostomia44
and weight loss,39 could possibly translate into potential
biomarkers. Changes in muscle gene expression, which
are altered during the course of the disorder, could also be
used to track disease progression.19,107 Data from several
studies have suggested that peripheral alterations that
manifest in the blood or plasma could be potential
www.thelancet.com/neurology Vol 8 August 2009
biomarkers of HD progression. Changes in nutritional
and metabolic markers,38,108 as well as markers of endocrine
dysfunction,71,79,109 have been suggested. Plasma
24S-hydroxycholesterol is a promising biomarker that
reects the early progression of neurodegeneration in
HD.110 Proteins of the immune system (eg, clusterin and
interleukin 6) also indicate disease progression.26,84 Altered
function of peripheral blood cells has also been suggested
as a biomarker. For example, adenosine receptor A2A
function in peripheral blood is altered in HD81 and
blood-borne immune cells from patients with HD are
hyperactive in response to stimulation from lipopolysaccharide.26
The idea that the eects of mutant huntingtin in
peripheral cells (eg, blood cells) can be used to monitor
disease progression is appealing. One major criticism of
plasma markers of neurodegenerative diseases is that
they might be non-specic, or that they could be
epiphenomena caused by a general illness state. Certainly,
in advanced HD, nutritional, metabolic, and infective
pathology are likely to confound any ndings. However,
changes that are seen even before the onset of overt
motor symptoms will be of interest.
Conclusions and future directions
Peripheral abnormalities are important features of HD.
Most of these changes have been described in animal
models of HD, but it is gradually becoming evident that
similar abnormalities occur in peripheral tissues of
patients. Such defects might result directly from mutant
huntingtin expression in peripheral tissues and might be
involved in HD symptoms. Recent studies on peripheral
pathology in HD have provided important insights into
underlying disease mechanisms. In the future, these
studies might pave the way for new therapies and new
markers to monitor disease progression. Finally,
treatment of peripheral symptoms in HD could prove
important, and positively aect quality of life and prevent
early death.
Search strategy and selection criteria
References for this Review were identied through searches
of PubMed with the search term Huntingtons disease,
crossreferenced with the terms adipose tissue, adipocyte,
biomarker, blood, cardiac, cholesterol, diabetes,
gastrointestinal, heart, huntingtin, hypothalamus,
kidney, liver, lymphocyte, muscle, osteoporosis,
pancreas, thyroid, testis, and weight loss, from 1965
to April, 2009. We mainly selected articles from the past
15 years, but we did not exclude commonly referenced older
publications. We also searched the reference lists of articles
identied by this search strategy and selected those that we
judged relevant. Several review articles were included as they
provide comprehensive overviews of some subtopics that are
included in this article.
771
Personal View
Contributors
JMMvdB initiated the project, drafted the rst version of the Review, and
composed the panel and gures. MB and PB contributed with additional
sections and, together with JMMvdB, edited later editions of the paper.
All authors agreed on the nal version of the review.
Conicts of interest
We have no conicts of interest.
Acknowledgments
We are active in a research environment that is sponsored by three
network grants that have HD as one of their themes, namely
NeuroFortis (Swedish Research Council Strong Research Environment),
The Nordic Center of Excellence on Neurodegeneration, and
BAGADILICO (Swedish Research Council Linnaus Grant). All these
grants have contributed to the intellectual environment that has made
this article possible.
References
1
Walker FO. Huntingtons disease. Lancet 2007; 369: 21828.
2
Hoogeveen AT, Willemsen R, Meyer N, et al. Characterization and
localization of the Huntington disease gene product.
Hum Mol Genet 1993; 2: 206973.
3
Li SH, Schilling G, Young WS 3rd, et al. Huntingtons disease gene
(IT15) is widely expressed in human and rat tissues. Neuron 1993;
11: 98593.
4
Trottier Y, Devys D, Imbert G, et al. Cellular localization of the
Huntingtons disease protein and discrimination of the normal and
mutated form. Nat Genet 1995; 10: 10410.
5
Cattaneo E, Zuccato C, Tartari M. Normal huntingtin function: an
alternative approach to Huntingtons disease. Nat Rev Neurosci
2005; 6: 91930.
6
Zeitlin S, Liu JP, Chapman DL, Papaioannou VE, Efstratiadis A.
Increased apoptosis and early embryonic lethality in mice
nullizygous for the Huntingtons disease gene homologue.
Nat Genet 1995; 11: 15563.
7
Lumsden AL, Henshall TL, Dayan S, Lardelli MT, Richards RI.
Huntingtin-decient zebrash exhibit defects in iron utilization
and development. Hum Mol Genet 2007; 16: 190520.
8
Parker WD Jr, Boyson SJ, Luder AS, Parks JK. Evidence for a defect
in NADH: ubiquinone oxidoreductase (complex I) in Huntingtons
disease. Neurology 1990; 40: 123134.
9
Sawa A, Wiegand GW, Cooper J, et al. Increased apoptosis of
Huntington disease lymphoblasts associated with repeat
length-dependent mitochondrial depolarization. Nat Med 1999;
5: 119498.
10 Lodi R, Schapira AH, Manners D, et al. Abnormal in vivo skeletal
muscle energy metabolism in Huntingtons disease and
dentatorubropallidoluysian atrophy. Ann Neurol 2000; 48: 7276.
11 Panov AV, Gutekunst CA, Leavitt BR, et al. Early mitochondrial
calcium defects in Huntingtons disease are a direct eect of
polyglutamines. Nat Neurosci 2002; 5: 73136.
12 Saft C, Zange J, Andrich J, et al. Mitochondrial impairment in
patients and asymptomatic mutation carriers of Huntingtons
disease. Mov Disord 2005; 20: 67479.
13 Mormone E, Matarrese P, Tinari A, et al. Genotype-dependent
priming to self- and xeno-cannibalism in heterozygous and
homozygous lymphoblasts from patients with Huntingtons
disease. J Neurochem 2006; 98: 109099.
14 Almeida S, Sarmento-Ribeiro AB, Januario C, Rego AC,
Oliveira CR. Evidence of apoptosis and mitochondrial
abnormalities in peripheral blood cells of Huntingtons disease
patients. Biochem Biophys Res Commun 2008; 374: 599603.
15 Panov AV, Lund S, Greenamyre JT. Ca+-induced permeability
transition in human lymphoblastoid cell mitochondria from normal
and Huntingtons disease individuals. Mol Cell Biochem 2005;
269: 14352.
16 Squitieri F, Cannella M, Sgarbi G, et al. Severe ultrastructural
mitochondrial changes in lymphoblasts homozygous for
Huntington disease mutation. Mech Ageing Dev 2006;
127: 21720.
17 Gizatullina ZZ, Lindenberg KS, Harjes P, et al. Low stability of
Huntington muscle mitochondria against Ca2+ in R6/2 mice.
Ann Neurol 2006; 59: 40711.
772
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
Luthi-Carter R, Hanson SA, Strand AD, et al. Dysregulation of
gene expression in the R6/2 model of polyglutamine disease:
parallel changes in muscle and brain. Hum Mol Genet 2002;
11: 191126.
Strand AD, Aragaki AK, Shaw D, et al. Gene expression in
Huntingtons disease skeletal muscle: a potential biomarker.
Hum Mol Genet 2005; 14: 186376.
Borovecki F, Lovrecic L, Zhou J, et al. Genome-wide expression
proling of human blood reveals biomarkers for Huntingtons
disease. Proc Natl Acad Sci USA 2005; 102: 110238.
Anderson AN, Roncaroli F, Hodges A, Deprez M, Turkheimer FE.
Chromosomal proles of gene expression in Huntingtons disease.
Brain 2008; 131: 38188.
Chaturvedi RK, Adhihetty P, Shukla S, et al. Impaired
PGC-1{alpha} function in muscle in Huntingtons disease.
Hum Mol Genet 2009; published online May 21. DOI:10.1093/hmg/
ddp243.
Sathasivam K, Hobbs C, Turmaine M, et al. Formation of
polyglutamine inclusions in non-CNS tissue. Hum Mol Genet 1999;
8: 81322.
Tanaka M, Machida Y, Niu S, et al. Trehalose alleviates
polyglutamine-mediated pathology in a mouse model of
Huntington disease. Nat Med 2004; 10: 14854.
Orth M, Cooper JM, Bates GP, Schapira AH. Inclusion formation
in Huntingtons disease R6/2 mouse muscle cultures. J Neurochem
2003; 87: 16.
Bjorkqvist M, Wild EJ, Thiele J, et al. A novel pathogenic pathway of
immune activation detectable before clinical onset in Huntingtons
disease. J Exp Med 2008; 205: 186977.
Sathasivam K, Woodman B, Mahal A, et al. Centrosome
disorganization in broblast cultures derived from R6/2
Huntingtons disease (HD) transgenic mice and HD patients.
Hum Mol Genet 2001; 10: 242535.
Pattison JS, Robbins J. Protein misfolding and cardiac disease:
establishing cause and eect. Autophagy 2008; 4: 82123.
Pattison JS, Sanbe A, Maloyan A, Osinska H, Klevitsky R,
Robbins J. Cardiomyocyte expression of a polyglutamine
preamyloid oligomer causes heart failure. Circulation 2008;
117: 274351.
Sanberg PR, Fibiger HC, Mark RF. Body weight and dietary factors
in Huntingtons disease patients compared with matched controls.
Med J Aust 1981; 1: 40709.
Farrer LA, Meaney FJ. An anthropometric assessment of
Huntingtons disease patients and families. Am J Phys Anthropol
1985; 67: 18594.
Farrer LA, Yu PL. Anthropometric discrimination among aected,
at-risk, and not-at-risk individuals in families with Huntington
disease. Am J Med Genet 1985; 21: 30716.
Stoy N, McKay E. Weight loss in Huntingtons disease. Ann Neurol
2000; 48: 13031.
Djousse L, Knowlton B, Cupples LA, Marder K, Shoulson I,
Myers RH. Weight loss in early stage of Huntingtons disease.
Neurology 2002; 59: 132530.
Trejo A, Tarrats RM, Alonso ME, Boll MC, Ochoa A, Velasquez L.
Assessment of the nutrition status of patients with Huntingtons
disease. Nutrition 2004; 20: 19296.
Trejo A, Boll MC, Alonso ME, Ochoa A, Velasquez L. Use of oral
nutritional supplements in patients with Huntingtons disease.
Nutrition 2005; 21: 88994.
Robbins AO, Ho AK, Barker RA. Weight changes in Huntingtons
disease. Eur J Neurol 2006; 13: e7.
Mochel F, Charles P, Seguin F, et al. Early energy decit in
Huntington disease: identication of a plasma biomarker traceable
during disease progression. PLoS ONE 2007; 2: e647.
Aziz NA, van der Burg JM, Landwehrmeyer GB, et al. Weight loss
in Huntington disease increases with higher CAG repeat number.
Neurology 2008; 71: 150613.
Morales LM, Estevez J, Suarez H, Villalobos R, Chacin de Bonilla L,
Bonilla E. Nutritional evaluation of Huntington disease patients.
Am J Clin Nutr 1989; 50: 14550.
van der Burg JM, Bacos K, Wood NI, et al. Increased metabolism in
the R6/2 mouse model of Huntingtons disease. Neurobiol Dis 2008;
29: 4151.
www.thelancet.com/neurology Vol 8 August 2009
Personal View
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
Goodman AO, Murgatroyd PR, Medina-Gomez G, et al. The
metabolic prole of early Huntingtons disease--a combined human
and transgenic mouse study. Exp Neurol 2008; 210: 69198.
Myers RH, Sax DS, Koroshetz WJ, et al. Factors associated with
slow progression in Huntingtons disease. Arch Neurol 1991;
48: 80004.
Wood NI, Goodman AO, van der Burg JM, et al. Increased thirst
and drinking in Huntingtons disease and the R6/2 mouse.
Brain Res Bull 2008; 76: 7079.
Podolsky S, Leopold NA, Sax DS. Increased frequency of diabetes
mellitus in patients with Huntingtons chorea. Lancet 1972;
1: 135658.
Podolsky S, Leopold NA. Abnormal glucose tolerance and arginine
tolerance tests in Huntingtons disease. Gerontology 1977; 23: 5563.
Farrer LA. Diabetes mellitus in Huntington disease. Clin Genet
1985; 27: 6267.
Hurlbert MS, Zhou W, Wasmeier C, Kaddis FG, Hutton JC,
Freed CR. Mice transgenic for an expanded CAG repeat in the
Huntingtons disease gene develop diabetes. Diabetes 1999;
48: 64951.
Duan W, Guo Z, Jiang H, Ware M, Li XJ, Mattson MP. Dietary
restriction normalizes glucose metabolism and BDNF levels, slows
disease progression, and increases survival in huntingtin mutant
mice. Proc Natl Acad Sci USA 2003; 100: 291116.
Bjorkqvist M, Fex M, Renstrom E, et al. The R6/2 transgenic
mouse model of Huntingtons disease develops diabetes due to
decient {beta}-cell mass and exocytosis. Hum Mol Genet 2005;
14: 56574.
Josefsen K, Nielsen MD, Jorgensen KH, et al. Impaired glucose
tolerance in the R6/1 transgenic mouse model of Huntingtons
disease. J Neuroendocrinol 2008; 20: 16572.
Lalic NM, Maric J, Svetel M, et al. Glucose homeostasis in
Huntington disease: abnormalities in insulin sensitivity and
early-phase insulin secretion. Arch Neurol 2008; 65: 47680.
Hunt MJ, Morton AJ. Atypical diabetes associated with inclusion
formation in the R6/2 mouse model of Huntingtons disease is not
improved by treatment with hypoglycaemic agents. Exp Brain Res
2005; 166: 22029.
Andreassen OA, Dedeoglu A, Stanojevic V, et al. Huntingtons
disease of the endocrine pancreas: insulin deciency and diabetes
mellitus due to impaired insulin gene expression. Neurobiol Dis
2002; 11: 41024.
Bacos K, Bjorkqvist M, Petersen A, et al. Islet beta-cell area and
hormone expression are unaltered in Huntingtons disease.
Histochem Cell Biol 2008; 129: 62329.
Chiang MC, Chen HM, Lee YH, et al. Dysregulation of C/EBPalpha
by mutant Huntingtin causes the urea cycle deciency in
Huntingtons disease. Hum Mol Genet 2007; 16: 48398.
Fain JN, Del Mar NA, Meade CA, Reiner A, Goldowitz D.
Abnormalities in the functioning of adipocytes from R6/2 mice that
are transgenic for the Huntingtons disease mutation.
Hum Mol Genet 2001; 10: 14552.
Phan J, Hickey MA, Zhang P, Chesselet MF, Reue K. Adipose
tissue dysfunction tracks disease progression in two Huntingtons
disease mouse models. Hum Mol Genet 2009; 18: 100616.
Ribchester RR, Thomson D, Wood NI, et al. Progressive
abnormalities in skeletal muscle and neuromuscular junctions of
transgenic mice expressing the Huntingtons disease mutation.
Eur J Neurosci 2004; 20: 3092114.
Beal MF. Mitochondria take center stage in aging and
neurodegeneration. Ann Neurol 2005; 58: 495505.
Arenas J, Campos Y, Ribacoba R, et al. Complex I defect in muscle
from patients with Huntingtons disease. Ann Neurol 1998;
43: 397400.
Ciammola A, Sassone J, Alberti L, et al. Increased apoptosis,
Huntingtin inclusions and altered dierentiation in muscle cell
cultures from Huntingtons disease subjects. Cell Death Dier 2006;
13: 206878.
Turner C, Cooper JM, Schapira AH. Clinical correlates of
mitochondrial function in Huntingtons disease muscle. Mov Disord
2007; 22: 171521.
Lanska DJ, Lanska MJ, Lavine L, Schoenberg BS. Conditions
associated with Huntingtons disease at death. A case-control study.
Arch Neurol 1988; 45: 87880.
www.thelancet.com/neurology Vol 8 August 2009
65
66
67
68
69
70
71
72
73
74
75
76
77
78
79
80
81
82
83
84
85
86
87
88
Bar KJ, Boettger MK, Andrich J, et al. Cardiovagal modulation upon
postural change is altered in Huntingtons disease. Eur J Neurol
2008; 15: 86971.
Mihm MJ, Amann DM, Schanbacher BL, Altschuld RA, Bauer JA,
Hoyt KR. Cardiac dysfunction in the R6/2 mouse model of
Huntingtons disease. Neurobiol Dis 2007; 25: 297308.
Strong TV, Tagle DA, Valdes JM, et al. Widespread expression of the
human and rat Huntingtons disease gene in brain and nonneural
tissues. Nat Genet 1993; 5: 25965.
Pridmore SA, Adams GC. The fertility of HD-aected individuals in
Tasmania. Aust N Z J Psychiatry 1991; 25: 26264.
Markianos M, Panas M, Kalfakis N, Vassilopoulos D. Plasma
testosterone in male patients with Huntingtons disease: relations
to severity of illness and dementia. Ann Neurol 2005; 57: 52025.
Van Raamsdonk JM, Murphy Z, Selva DM, et al. Testicular
degeneration in Huntington disease. Neurobiol Dis 2007; 26: 51220.
Saleh N, Moutereau S, Durr A, et al. Neuroendocrine disturbances
in Huntingtons disease. PLoS ONE 2009; 4: e4962.
Leavitt BR, Guttman JA, Hodgson JG, et al. Wild-type huntingtin
reduces the cellular toxicity of mutant huntingtin in vivo.
Am J Hum Genet 2001; 68: 31324.
Papalexi E, Persson A, Bjorkqvist M, et al. Reduction of GnRH and
infertility in the R6/2 mouse model of Huntingtons disease.
Eur J Neurosci 2005; 22: 154146.
Van Raamsdonk JM, Pearson J, Rogers DA, et al. Loss of wild-type
huntingtin inuences motor dysfunction and survival in the
YAC128 mouse model of Huntington disease. Hum Mol Genet 2005;
14: 137992.
Mangiarini L, Sathasivam K, Seller M, et al. Exon 1 of the HD gene
with an expanded CAG repeat is sucient to cause a progressive
neurological phenotype in transgenic mice. Cell 1996; 87: 493506.
Bonelli C, Bonelli R, Eichinger M, et al. Bone density and bone
turnover in Huntingtons disease. Osteoporosis Int 2002; 13: S64.
Otti DV, Hdl AK, Bonelli CM, et al. Osteoporosis in
Huntingtons disease. Neurodegenerative Dis 2007;
4 (suppl 1): 304 (abstract 914).
Bjorkqvist M, Petersen A, Bacos K, et al. progressive alterations in
the hypothalamic-pituitary-adrenal axis in the R6/2 transgenic
mouse model of Huntingtons disease. Hum Mol Genet 2006;
15: 171321.
Aziz NA, Pijl H, Frolich M, van der Graaf AW, Roelfsema F,
Roos RA. Increased hypothalamic-pituitary-adrenal axis activity in
Huntingtons disease. J Clin Endocrinol Metab 2009; 94: 122328.
Maglione V, Cannella M, Gradini R, Cislaghi G, Squitieri F.
Huntingtin fragmentation and increased caspase 3, 8 and 9
activities in lymphoblasts with heterozygous and homozygous
Huntingtons disease mutation. Mech Ageing Dev 2006;
127: 21316.
Varani K, Abbracchio MP, Cannella M, et al. Aberrant A2A receptor
function in peripheral blood cells in Huntingtons disease.
Faseb J 2003; 17: 214850.
Varani K, Bachoud-Levi AC, Mariotti C, et al. Biological
abnormalities of peripheral A(2A) receptors in a large
representation of polyglutamine disorders and Huntingtons
disease stages. Neurobiol Dis 2007; 27: 3643.
Norman TR, Chiu E, French MA. Platelet monoamine
oxidase activity in patients with Huntingtons disease.
Clin Exp Pharmacol Physiol 1987; 14: 54750.
Dalrymple A, Wild EJ, Joubert R, et al. Proteomic proling of plasma
in Huntingtons disease reveals neuroinammatory activation and
biomarker candidates. J Proteome Res 2007; 6: 283340.
Fong Y, Moldawer LL, Marano M, et al. Cachectin/TNF or IL-1 alpha
induces cachexia with redistribution of body proteins.
Am J Physiol 1989; 256(3 Pt 2): R65965.
Langen RC, Schols AM, Kelders MC, Wouters EF,
Janssen-Heininger YM. Inammatory cytokines inhibit myogenic
dierentiation through activation of nuclear factor-kappaB.
Faseb J 2001; 15: 116980.
Spiegelman BM, Hotamisligil GS. Through thick and thin: wasting,
obesity, and TNF alpha. Cell 1993; 73: 62527.
Aziz NA, van der Marck MA, Pijl H, Olde Rikkert MG, Bloem BR,
Roos RA. Weight loss in neurodegenerative disorders. J Neurol
2008; 255: 187280.
773
Personal View
89
Petersen A, Bjorkqvist M. Hypothalamic-endocrine aspects in
Huntingtons disease. Eur J Neurosci 2006; 24: 96167.
90 Aziz NA, Swaab DF, Pijl H, Roos RA. Hypothalamic dysfunction
and neuroendocrine and metabolic alterations in Huntingtons
disease: clinical consequences and therapeutic implications.
Rev Neurosci 2007; 18: 22351.
91 Petersen A, Gil J, Maat-Schieman ML, et al. Orexin loss in
Huntingtons disease. Hum Mol Genet 2005; 14: 3947.
92 Aziz A, Fronczek R, Maat-Schieman M, et al. hypocretin and
melanin-concentrating hormone in patients with Huntington
disease. Brain Pathol 2008; 18: 47483.
93 Kotliarova S, Jana NR, Sakamoto N, et al. Decreased expression of
hypothalamic neuropeptides in Huntington disease transgenic
mice with expanded polyglutamine-EGFP uorescent aggregates.
J Neurochem 2005; 93: 64153.
94 Fahrenkrug J, Popovic N, Georg B, Brundin P, Hannibal J. Decreased
VIP and VPAC2 receptor expression in the biological clock of the
R6/2 Huntingtons disease mouse. J Mol Neurosci 2007; 31: 13948.
95 Martin B, Golden E, Carlson OD, et al. Exendin-4 improves
glycemic control, ameliorates brain and pancreatic pathologies, and
extends survival in a mouse model of Huntingtons disease.
Diabetes 2009; 58: 31828.
96 Cannella M, Maglione V, Martino T, et al. DNA instability in
replicating Huntingtons disease lymphoblasts. BMC Med Genet
2009; 10: 11.
97 Valenza M, Rigamonti D, Goredo D, et al. Dysfunction of the
cholesterol biosynthetic pathway in Huntingtons disease.
J Neurosci 2005; 25: 993239.
98 Valenza M, Carroll JB, Leoni V, et al. Cholesterol biosynthesis
pathway is disturbed in YAC128 mice and is modulated by
huntingtin mutation. Hum Mol Genet 2007; 16: 218798.
99 Valenza M, Leoni V, Tarditi A, et al. Progressive dysfunction of the
cholesterol biosynthesis pathway in the R6/2 mouse model of
Huntingtons disease. Neurobiol Dis 2007; 28: 13342.
100 Marullo M, Valenza M, Mariotti C, Di Donato S, Cattaneo E,
Zuccato C. Analysis of the repressor element-1 silencing
transcription factor/neuron-restrictive silencer factor occupancy of
non-neuronal genes in peripheral lymphocytes from patients with
Huntingtons disease. Brain Pathol 2008; published online
December 23. DOI:10.1111/j.1750^=3639.2008.00249.x.
774
101 Cui L, Jeong H, Borovecki F, Parkhurst CN, Tanese N, Krainc D.
Transcriptional repression of PGC-1alpha by mutant huntingtin
leads to mitochondrial dysfunction and neurodegeneration. Cell
2006; 127: 5969.
102 Weydt P, Pineda VV, Torrence AE, et al. Thermoregulatory and
metabolic defects in Huntingtons disease transgenic mice
implicate PGC-1alpha in Huntingtons disease neurodegeneration.
Cell Metab 2006; 4: 34962.
103 Ma TC, Buescher JL, Oatis B, et al. Metformin therapy in a
transgenic mouse model of Huntingtons disease. Neurosci Lett
2007; 411: 98103.
104 Perry T, Greig NH. The glucagon-like peptides: a double-edged
therapeutic sword? Trends Pharmacol Sci 2003; 24: 37783.
105 Veiga S, Melcangi RC, Doncarlos LL, Garcia-Segura LM, Azcoitia I.
Sex hormones and brain aging. Exp Gerontol 2004; 39: 162331.
106 Wild EJ, Tabrizi SJ. Predict-HD and the future of therapeutic trials.
Lancet Neurol 2006; 5: 72425.
107 Luthi-Carter R, Strand AD, Hanson SA, et al. Polyglutamine and
transcription: gene expression changes shared by DRPLA and
Huntingtons disease mouse models reveal context-independent
eects. Hum Mol Genet 2002; 11: 192737.
108 Hersch SM, Gevorkian S, Marder K, et al. Creatine in Huntington
disease is safe, tolerable, bioavailable in brain and reduces serum
8OH2dG. Neurology 2006; 66: 25052.
109 Markianos M, Panas M, Kalfakis N, Vassilopoulos D. Plasma
testosterone, dehydroepiandrosterone sulfate, and cortisol in female
patients with Huntingtons disease. Neuro Endocrinol Lett 2007;
28: 199203.
110 Leoni V, Mariotti C, Tabrizi SJ, et al. Plasma 24S-hydroxycholesterol
and caudate MRI in pre-manifest and early Huntingtons disease.
Brain 2008; 131 (Pt 11): 285159.
www.thelancet.com/neurology Vol 8 August 2009
Reproduced with permission of the copyright owner. Further reproduction prohibited without permission.
S-ar putea să vă placă și
- Huntington’s Chorea, A Simple Guide To The Condition, Treatment And Related ConditionsDe la EverandHuntington’s Chorea, A Simple Guide To The Condition, Treatment And Related ConditionsEvaluare: 1 din 5 stele1/5 (1)
- Huntington’s Disease: Pathogenic Mechanisms and Implications for TherapeuticsDe la EverandHuntington’s Disease: Pathogenic Mechanisms and Implications for TherapeuticsX. William YangÎncă nu există evaluări
- Reviews: Normal Huntingtin Function: An Alternative Approach To Huntington'S DiseaseDocument12 paginiReviews: Normal Huntingtin Function: An Alternative Approach To Huntington'S DiseaseDaniel SchmidtÎncă nu există evaluări
- Huntington's Disease: A Clinical ReviewDocument32 paginiHuntington's Disease: A Clinical ReviewDewi NofiantiÎncă nu există evaluări
- "Huntington Disease: Clinical Features and Diagnosis" "Huntington Disease: Management"Document36 pagini"Huntington Disease: Clinical Features and Diagnosis" "Huntington Disease: Management"Gilbert LimenÎncă nu există evaluări
- Apuntes Caso ClínicoDocument9 paginiApuntes Caso ClínicoViviana LunaÎncă nu există evaluări
- OT6 - Huntington's DiseaseDocument1 paginăOT6 - Huntington's DiseaseAnnbe BarteÎncă nu există evaluări
- Huntington Disease Research PaperDocument36 paginiHuntington Disease Research PaperJingyun WuÎncă nu există evaluări
- BMB-22-157 1 1Document30 paginiBMB-22-157 1 1Jingyun WuÎncă nu există evaluări
- LC3 GrantDocument3 paginiLC3 GrantAj KeefeÎncă nu există evaluări
- Immunohistochemistry Electron Microscopy Subcellular Fractionation Vesicles Microtubules Mitochondria Vesicle Clathrin Endocytosis Epithelial PolarityDocument2 paginiImmunohistochemistry Electron Microscopy Subcellular Fractionation Vesicles Microtubules Mitochondria Vesicle Clathrin Endocytosis Epithelial PolarityAmna SiddiquiÎncă nu există evaluări
- Research Paper On Huntingtons DiseaseDocument4 paginiResearch Paper On Huntingtons Diseasemkcewzbnd100% (1)
- High and Low Levels of An NTRK2-Driven Genetic Profile Affect Motor - and Cognition-Associated Frontal Gray Matter in Prodromal Huntington's DiseaseDocument24 paginiHigh and Low Levels of An NTRK2-Driven Genetic Profile Affect Motor - and Cognition-Associated Frontal Gray Matter in Prodromal Huntington's DiseaseJennifer CiarochiÎncă nu există evaluări
- Correlation Between Overt Hyperthyroid and Subclinical Hyperthyroid and Cognitive Impairment in Dr. Sardjito General Hospital, Yogyakarta, IndonesiaDocument8 paginiCorrelation Between Overt Hyperthyroid and Subclinical Hyperthyroid and Cognitive Impairment in Dr. Sardjito General Hospital, Yogyakarta, IndonesiaNintha TariganÎncă nu există evaluări
- 1034 Full PDFDocument9 pagini1034 Full PDFZaki AlloucheÎncă nu există evaluări
- ÜldiseloomustusDocument2 paginiÜldiseloomustusKristjan KiviÎncă nu există evaluări
- Essay - Nanou - Neurogenerative Disorders Discussing Huntingtons Disease and Its Possible Treatments - Biol - Basics - Psychol - 2022 - Spring.Document10 paginiEssay - Nanou - Neurogenerative Disorders Discussing Huntingtons Disease and Its Possible Treatments - Biol - Basics - Psychol - 2022 - Spring.Maria NaÎncă nu există evaluări
- Journal of Advanced Scientific Research: Review ArticleDocument7 paginiJournal of Advanced Scientific Research: Review ArticleYacoba PatandiananÎncă nu există evaluări
- Hungitons DiseaseDocument267 paginiHungitons DiseasePeter Osundwa Kiteki100% (1)
- Huntingtons Disease Research PaperDocument6 paginiHuntingtons Disease Research Paperafeaynwqz100% (1)
- Advances in The PharmacologicalDocument12 paginiAdvances in The PharmacologicalpaijosusenoÎncă nu există evaluări
- DBS HuntingtonDocument10 paginiDBS HuntingtonEdith SteinÎncă nu există evaluări
- Huntington's DiseaseDocument9 paginiHuntington's DiseaseBARATHY A/P RAMAN KPM-GuruÎncă nu există evaluări
- Research Paper Huntingtons DiseaseDocument5 paginiResearch Paper Huntingtons Diseasejcrfwerif100% (1)
- Bordelon 13 HuntingtonDocument10 paginiBordelon 13 HuntingtonMARIA MONTSERRAT SOMOZA MONCADAÎncă nu există evaluări
- Hungtington DiseaseDocument46 paginiHungtington DiseasewarunkumarÎncă nu există evaluări
- Therapeutic Approaches For Inhibition of Protein Aggregation in Huntington's DiseaseDocument9 paginiTherapeutic Approaches For Inhibition of Protein Aggregation in Huntington's DiseaseJorge ElectoÎncă nu există evaluări
- Term Paper On Huntingtons DiseaseDocument6 paginiTerm Paper On Huntingtons Diseaseea8r2tyk100% (1)
- HypothalaDocument13 paginiHypothalaijafetÎncă nu există evaluări
- Hun A Clini 2022 AndhaleDocument7 paginiHun A Clini 2022 AndhaleAngelica Maria PardoÎncă nu există evaluări
- PsychFest - Lipid Droplets in HepatocytesDocument1 paginăPsychFest - Lipid Droplets in HepatocytesAj KeefeÎncă nu există evaluări
- HHS Public Access: Studying Polyglutamine Diseases in DrosophilaDocument39 paginiHHS Public Access: Studying Polyglutamine Diseases in DrosophilaJoeÎncă nu există evaluări
- Huntingtons ChoreaDocument11 paginiHuntingtons ChoreajananiÎncă nu există evaluări
- Huntingtons Treatment PDFDocument8 paginiHuntingtons Treatment PDFave123izationÎncă nu există evaluări
- Paper 8 SidiquiDocument31 paginiPaper 8 SidiquiNICOLE HSIAO-SANCHEZÎncă nu există evaluări
- Reversible Dementia: by Ahmed Abdul GhaniDocument41 paginiReversible Dementia: by Ahmed Abdul GhaniAnjjÎncă nu există evaluări
- Huntington'S Disease: Update and Review Neuropsychiatric AspectsDocument20 paginiHuntington'S Disease: Update and Review Neuropsychiatric AspectsDewi NofiantiÎncă nu există evaluări
- Hypoglycemia and Jaundice in A Newborn Revealing Pituitary Stalk Interruption SyndromeDocument4 paginiHypoglycemia and Jaundice in A Newborn Revealing Pituitary Stalk Interruption SyndromeIJAR JOURNALÎncă nu există evaluări
- What Is HDDocument8 paginiWhat Is HDMichael Andrew OlsenÎncă nu există evaluări
- 19 Salas Milagros - SnowdenDocument14 pagini19 Salas Milagros - SnowdenJose Bryan GonzalezÎncă nu există evaluări
- What A SeminarDocument51 paginiWhat A Seminarapi-3741769Încă nu există evaluări
- Huntington's DiseaseDocument4 paginiHuntington's DiseaseJ. L.Încă nu există evaluări
- HuntingtonDocument11 paginiHuntingtonrita3maria2002Încă nu există evaluări
- The Ketogenic Diet As A Non Pharmacological Treatment For HIV Associated Neurocognitive Disorder A Descriptive AnalysisDocument5 paginiThe Ketogenic Diet As A Non Pharmacological Treatment For HIV Associated Neurocognitive Disorder A Descriptive AnalysisPaul HartingÎncă nu există evaluări
- The Study of Psychiatric Disorders in Patients With Thyroid Disorder at The Tertiary Care Centre in Western Region of NepalDocument7 paginiThe Study of Psychiatric Disorders in Patients With Thyroid Disorder at The Tertiary Care Centre in Western Region of NepalAyu PurniawatiÎncă nu există evaluări
- Management of HypopituitarismDocument23 paginiManagement of HypopituitarismMaría JoséÎncă nu există evaluări
- Tasks 3 and 4 Genetics Questions and Problems LEAH DAVIES PDFDocument11 paginiTasks 3 and 4 Genetics Questions and Problems LEAH DAVIES PDFSara SÎncă nu există evaluări
- CN 16 519Document14 paginiCN 16 519John SmithÎncă nu există evaluări
- The PERK-Dependent Molecular Mechanisms As A Novel Therapeutic Target For Neurodegenerative DiseasesDocument40 paginiThe PERK-Dependent Molecular Mechanisms As A Novel Therapeutic Target For Neurodegenerative DiseasesCarlos VSÎncă nu există evaluări
- Genetic Disorder: Huntington's Disease Huntington's Disease: This Disease Was First Recognised As An Inherited Disorder in 1872 When ADocument4 paginiGenetic Disorder: Huntington's Disease Huntington's Disease: This Disease Was First Recognised As An Inherited Disorder in 1872 When Aapi-315693239Încă nu există evaluări
- Understanding Huntington's Disease Causes, Symptoms and DiagnosesDocument10 paginiUnderstanding Huntington's Disease Causes, Symptoms and Diagnosessmithetvin625Încă nu există evaluări
- Huntington's DiseaseDocument18 paginiHuntington's DiseaseZachary DequillaÎncă nu există evaluări
- Azadiradione Restores Protein Quality Control and Ameliorates The Disease Pathogenesis in A Mouse Model of Huntington 'S DiseaseDocument10 paginiAzadiradione Restores Protein Quality Control and Ameliorates The Disease Pathogenesis in A Mouse Model of Huntington 'S DiseaseAnirban MannaÎncă nu există evaluări
- Age Dependent and Gender Dependent RegulDocument33 paginiAge Dependent and Gender Dependent RegulandreÎncă nu există evaluări
- Clinical Characteristics For The Relationship Between Type-2 Diabetes Mellitus and Cognitive Impairment: A Cross-Sectional StudyDocument9 paginiClinical Characteristics For The Relationship Between Type-2 Diabetes Mellitus and Cognitive Impairment: A Cross-Sectional StudyNathaly Baldeon CamposÎncă nu există evaluări
- High Plasma Oxytocin Levels in Men With Hypersexual DisorderDocument7 paginiHigh Plasma Oxytocin Levels in Men With Hypersexual DisorderboxeritoÎncă nu există evaluări
- Huntington's DiseaseDocument13 paginiHuntington's DiseaseGEETA MOHAN100% (1)
- Youssef Et Al 2016 Pathology of The Aging Brain in Domestic and Laboratory Animals and Animal Models of HumanDocument22 paginiYoussef Et Al 2016 Pathology of The Aging Brain in Domestic and Laboratory Animals and Animal Models of HumanISABELAÎncă nu există evaluări
- Bipolar 1 DisorderDocument11 paginiBipolar 1 DisorderGirlly AlcantaraÎncă nu există evaluări
- Ijms 23 03861Document32 paginiIjms 23 03861Surfer LeoÎncă nu există evaluări
- Advs202104006 Sup 0001 SuppmatDocument20 paginiAdvs202104006 Sup 0001 SuppmatLe Victor DungÎncă nu există evaluări
- Pacificgoldenplover Microbe Mission Practice TestDocument5 paginiPacificgoldenplover Microbe Mission Practice TestsandeeptaraÎncă nu există evaluări
- Turn Up The Heat - Unlock The Fat-Burning Power of Your MetabolismDocument378 paginiTurn Up The Heat - Unlock The Fat-Burning Power of Your MetabolismElvis Florentino96% (23)
- Golden File 4 Corrected With Refernces Where Necessary 2 PDFDocument127 paginiGolden File 4 Corrected With Refernces Where Necessary 2 PDFJansher Ali ChohanÎncă nu există evaluări
- Bio 473 Reproductive Endocrinology Lab Compiled LabDocument10 paginiBio 473 Reproductive Endocrinology Lab Compiled Labapi-253602935Încă nu există evaluări
- FM - K36 - Prenatal CareDocument33 paginiFM - K36 - Prenatal CareNatasya Ryani PurbaÎncă nu există evaluări
- Mual MuntahDocument8 paginiMual MuntahRosalindaRezkyÎncă nu există evaluări
- Gene Technology ...Document28 paginiGene Technology ...Jelita AlviolinaÎncă nu există evaluări
- PrimaquineDocument3 paginiPrimaquineVijayakumar NsÎncă nu există evaluări
- Springer - From Messengers To Molecules-Memories Are Made of These (Neuroscience Intelligence Unit)Document638 paginiSpringer - From Messengers To Molecules-Memories Are Made of These (Neuroscience Intelligence Unit)FroyÎncă nu există evaluări
- NleDocument12 paginiNleJohn Haider Colorado Gamol50% (2)
- 5 Psae Egypt 2014Document9 pagini5 Psae Egypt 2014Khoa Vi Sinh BVCRÎncă nu există evaluări
- Excretory ProductsDocument8 paginiExcretory ProductsmatinÎncă nu există evaluări
- Bartter Syndrome: ScapeDocument21 paginiBartter Syndrome: ScapeAl DiÎncă nu există evaluări
- Huntington's DiseaseDocument13 paginiHuntington's DiseaseGEETA MOHAN100% (1)
- Hepatic CellsDocument2 paginiHepatic CellsSteven RiosÎncă nu există evaluări
- Genetic Engineering PDFDocument50 paginiGenetic Engineering PDFBrian Rey L. AbingÎncă nu există evaluări
- Litsearch UreaDocument126 paginiLitsearch Ureaemmet11100% (1)
- First Division (G.R. No. 217036, August 20, 2018)Document12 paginiFirst Division (G.R. No. 217036, August 20, 2018)Emir MendozaÎncă nu există evaluări
- Skin Lesions and DiagnosisDocument25 paginiSkin Lesions and DiagnosisBUCM Class 2023Încă nu există evaluări
- DLL FrontDocument4 paginiDLL FrontAnafemolyn NingascaÎncă nu există evaluări
- Silver PADocument7 paginiSilver PAhitman2288Încă nu există evaluări
- Journal of Neurology Volume 256 Issue 3 2009 (Doi 10.1007/s00415-009-0149-2) T. Lempert H. Neuhauser - Epidemiology of Vertigo, Migraine and Vestibular MigraineDocument6 paginiJournal of Neurology Volume 256 Issue 3 2009 (Doi 10.1007/s00415-009-0149-2) T. Lempert H. Neuhauser - Epidemiology of Vertigo, Migraine and Vestibular MigraineDevi Arnes SimanjuntakÎncă nu există evaluări
- Animal Testing (Black Mamba)Document3 paginiAnimal Testing (Black Mamba)Robespade El PistoleroÎncă nu există evaluări
- Genetics and Genomics in Nursing Health Care 2nd EditionDocument61 paginiGenetics and Genomics in Nursing Health Care 2nd Editionjulia.swanson282100% (44)
- Detailed EoT Coverage 9 ADVDocument50 paginiDetailed EoT Coverage 9 ADVNKAÎncă nu există evaluări
- Tenebrio Molitor Exposed To Environmental Bacteria and Fungi atDocument17 paginiTenebrio Molitor Exposed To Environmental Bacteria and Fungi atDbaltÎncă nu există evaluări
- Annex I Summary of Product CharacteristicsDocument29 paginiAnnex I Summary of Product CharacteristicslisnerisÎncă nu există evaluări
- Luminescent ATP Detection Assay Kit Ab113849 Luminescent ATP Detection Assay Kit Ab113849Document4 paginiLuminescent ATP Detection Assay Kit Ab113849 Luminescent ATP Detection Assay Kit Ab113849carlosÎncă nu există evaluări
- Question Paper Unit f214 01 Communication Homeostasis and EnergyDocument20 paginiQuestion Paper Unit f214 01 Communication Homeostasis and EnergygeorgiaÎncă nu există evaluări