Documente Academic
Documente Profesional
Documente Cultură
Ann Bot-1965-EVANS-205-17
Încărcat de
Vrushali PatilDescriere originală:
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Ann Bot-1965-EVANS-205-17
Încărcat de
Vrushali PatilDrepturi de autor:
Formate disponibile
Intercalary Growth in the Aerial Shoot of
Eleocharis acuta R. Br. Prodr.
I. Structure of the Growing Zone1
BY
P. S. EVANS2
With six Figures in the Text
ABSTRACT
The numbers and distribution of cell division and the cell lengths have been
determined for the various tissues in the intercalary meristem of Eleocharis acuta
R. Br. Prodr. Cell divisions are most numerous in the chlorenchyma tissue and
fewest in the plate parenchyma. In all tissues examined except the plate parenchyma, cell length is inversely correlated with numbers of cell divisions. The
cell divisions which occur in the vascular tissue in the intercalary meristem are
regarded as being a part of the intercalary growth and not as a normal development of a procambium. Mature xylem and phloem are seen to be present in the
larger vascular bundles right through the meristematic zone, and the xylem
tracheids are shown to be conductive. The transverse strands connecting adjacent
vascular bundles are considered to be important in the conduction of water.
INTRODUCTION
NTERCALARY growth is widespread among vascular plants, being a
particularly important constituent of the growth of monocotyledons, but
little work has been done on it. Eleocharis acuta provides suitable material for
study since the greater part of the aerial shoot consists of a single internode
formed by an intercalary meristem, and each plant produces a succession of
similar shoots. It is therefore a relatively simple matter to obtain intercalary
meristems at any desired stage of development.
The Nature of the Intercalary Meristem
The intercalary meristem is a rib meristem; i.e. the cells are formed in
vertical columns by transverse divisions in a dividing zone at some point in
the columns. Esau (1953) has described it as a meristematic zone inserted
between more or less differentiated tissue regions. She points out that since
differentiated tissue elements are present and the zone is eventually transformed into mature tissues, it is not of the same rank as the apical and lateral
meristems. She does not regard the activity of intercalary meristems as an
1
This study is part of a thesis presented in part fulfilment of the requirements for the degree
of Master of Science to the University of New Zealand, 1061.
1
Present address, Grasslands Division, Department of Scientific and Industrial Research,
Palmerston North, New Zealand.
[Annals of Botany, N.S. Vol. 39, No. 114, 1965.]
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
Botany Department, University of Canterbury, Christchurch, New Zealand
206
EvansIntercalary Growth in Eleocharis
Anatomy of Eleocharis acuta
Eleocharis acuta consists of a complex of long or short rhizomes which give
rise to numerous tufts of scapose aerial shoots (haulms) 2-45 cm or more in
length and up to 5 mm in diameter, ending either in a dead tip enclosed
within one to several scale leaves, or in a flowering spikelet. The basal
portion of the haulm is surrounded by a scale leaf and two closely appressed
sheaths.
The haulm below the insertion of the first sheath consists of an inner and
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
isolated phenomenon but merely as an intensified and localized expression of
primary growth.
Jacobs (1947) makes a distinction between the nature of the intercalary
meristem of Arachis and those of grasses. That of Arackis he calls a 'resumptive' meristem; i.e. it has resumed growth after a period of quiescence. Those
of grasses he refers to as detached from the apical meristem, but growing continually without any resting stage. It is doubtful if this distinction is significant, for the internodes of grasses may elongate some time after they are laid
down.
The first detailed account of intercalary growth seems to be that of Buchholz (1920), whose work was subsequently extended by Schmalfuss (1930).
These two authors have given surveys of previous observations. Both monocotyledon leaves and stems where the growing zone occurs at the base of the
internode, and dicotyledon flowering stems where it occurs just below the
inflorescence, were examined. Schmalfuss studied the direction and extent of
cell divisions and also cell lengths. He stressed that intercalary growth is predominantly in a longitudinal direction, and that cell division and cell elongation are both important to varying degrees. Miltenyi (1931) has described the
development of the intercalary meristem of several cereals, and Kaufman
(1959) has done the same for Oryza sativa. Golub and Wetmore (1948) have
described the comparable phenomenon in Equisetum arvense.
The first analytical account was that of Jacobs (1947), who examined the
extent of cell divisions in the various tissues of the gynophore of Arackis
hypogaea. Sachs, Lang, Bretz, and Roach (i960) have mapped the distribution
of cell divisions in the flowering stem of Chrysanthemum morifolium, and
Denne (i960) has given a comprehensive account of both cell elongation and
cell division in the leaf and sheath of Narcissus pseudonarcissus.
The way in which vascular continuity is maintained has been examined by
most of the authors who have investigated intercalary growth. Buchholz
(1920) and Schmalfuss (1930) used a dye to trace the xylary connexions through
the zone. They, Jacobs (1947), Golub and Wetmore (1948), and Denne (i960)
examined the intercalary growing zone for mature vascular tissue.
Transverse strands connecting adjacent longitudinal vascular bundles have
been recorded in monocotyledon leaves (Buchholz, 1920; Percival, 1921;
Kaufman, 1959) and stems (Plowman, 1906; Buchholz, 1920). Both Plowman
and Kaufman found them associated with pith plates.
EvansIntercalary Growth in Eleocharis
207
The vascular system consists of a series of closed collateral bundles with
centrifugal phloem. In contrast to the rest of the axis, annular thickening is
found in the protoxylem of the internode produced by the intercalary meristem.
The protoxylem is more abundant than elsewhere and breaks down to form a
lacuna. Also in this internode, the larger bundles have two lateral vessels.
The tracheary elements are longer than elsewhere.
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
an outer region of isodiametric parenchyma cells separated by a multiplelayer endodermis. Vascular bundles, each with their own single-layer endodermis, are scattered within the inner region. The epidermal cells are all of
one type and no stomata have been observed. Just inside the epidermis are
numerous small fibre bundles.
In the region of insertion of the two sheaths the multiple-layer endodermis
disappears and the vascular bundles move outwards. The outermost bundles
are the traces leading to the two sheaths. The remainder continue up the
haulm three to four cells inside the epidermis.
Immediately above the insertion of the second
sheath lies the intercalary meristem from
which develops the major part of the haulm.
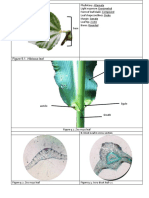
Fig. 1 shows the structure of the base of the
pp
haulm.
In the internode produced by the intercalary
meristem (see Fig. 2) there is never a ring of
endodermis, and in the region of the active
meristem the endodermis of the individual
vascular bundles is not distinguishable. In
contrast to the lower regions, the epidermis
here consists of several types of cells, and stomata are present. The adjacent parenchyma
is modified to form a chlorenchymatous layer
two to five cells deep in which are embedded
the vascular bundles. The centre of the internode, instead of being solid, consists of a
regular series of air spaces separated by ribs
=E
and plates of parenchyma. The ribs consist of
elongated cells of varying sizes whereas the FIG. 1. Structure of the haulm
cells of the plates are short and stellate. The air base. The intercalary meriatem is
situated above the insertion of the
spaces are formed by the tearing apart of the inner sheath. A = air spaces, PP =
plate parenchyma. The sub-epidermal fibre parenchyma plates. PR = parenbundles are similar to those of the lower regions. chyma rib. is = inner sheath.
OS = outer sheath, vu = vascular
This internode, which forms the main part of supply to upper part of haulm,
the haulm, ends in a scale leaf or bract. The vs = vascular supply to sheaths.
E = endodermis.
haulm here narrows abruptly and then tapers
to the tip. The structure is more like that of the base of the haulm than that
of the region directly below. Where the haulm continues into a flowering
spikelet, there is a maze of bract and flower traces.
EvansIntercalary Growth in Eleocharis
208
Flo. 2. Sections through the base of an intemode showing positions
of the various tissues, (a) transverse section. (6) longitudinal section
along line X
X in (a). E = epidermis. F = fibre bundle. CH =
chlorenchyma. v = vascular bundles, PP = plate parenchyma, RP =>
rib parenchyma. ( X 200).
At regular intervals up the main internode, there occur transverse strands of
elongated parenchyma cells connecting adjacent longitudinal vascular bundles.
The cells of these strands arise by division of the inner chlorenchyma and
outer rib-parenchyma cells. Although not developmentaUy part of the vascular
system they will be shown to have a conductive function. They are seen to be
associated with the pith plates.
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
(b)
EvansIntercalary Growth in Eleocharis
209
MATERIALS AND M E T H O D S
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
Material for microscopic examination was obtained locally, fixed in formalin-aceto-alcohol for several days and then preserved in 70 per cent ethyl
alcohol. Microtome sections cut at 10/x were stained with safranin and fast
green.
In order to determine the rate and region of elongation in the intercalary
meristem by marking the epidermis as did Buchholz (1920) and Jacobs (1947),
plants were grown in culture solution in flasks, wrapped in black paper to
prevent algal growth.
Preliminary investigations showed that all cell divisions and almost all cell
elongation occurred in the first 2 cm of the internode, and so two 1-cm lengths
of the base of the internode were embedded and sectioned. Two haulms 23 and
24 cm long (approximately half of the final length) from different plants were
used. One was sectioned longitudinally and the other transversely.
There are several ways of estimating cell divisions. Jacobs (1947) and Sachs
Lang, Bretz, and Roach (i960) using serial longitudinal sections, measured
their position by means of an ocular micrometer grid and graph paper corresponding to segments of the stem. Jacobs (1947) attempted to relate the
total number of cell divisions to the number of cells in a transverse section to
determine the relationship between cell division and cell length. Brown and
Rickless (1949) macerated root tip tissue and examined the maceration for the
percentage of cells dividing; a distinction between tissues cannot be easily made
using this method. Denne (i960) examined longitudinal sections, counting the
percentage of cells dividing in each tissue in each segment of the leaf, a thousand
cells being counted each time.
This last method, which has the advantage over that of Jacobs (1947) and
Sachs, Lang, Bretz, and Roach (i960) in that serial sections are not required,
was used in examining the longitudinal sections (Fig. 3). Alternate sections
were examined to avoid counting the same divisions twice. The percentage of
cells dividing was calculated for every o-5-mm segment, except in the plate
parenchyma in which 0-25-11101 segments were used because the divisions extended only over a small distance. The number and direction of cell divisions
were recorded for every tenth serial transverse section as a check on the
method, and also to see if any divisions were longitudinal since Eleocharis has
polycentric chromosomes and it was impossible to distinguish between a
polar view of metaphase or anaphase and an equatorial view of prophase. For
this reason too all stages of cell division were counted. Jacobs (1947) did not
include prophases. Successive groups of five totals were added to give a 10
per cent sample of the cell divisions in each 0'5-mm segment (Fig. 4). From
longitudinal sections the cell length (average of 100 cells) was measured for
each tissue except the vascular in each o-5-mm segment (0-25 mm in the plate
parenchyma) from the base of the internode to the level where cell divisions
ceased. Cell length counts were made only to the point in each tissue where
cell division ceased, because beyond this the rib parenchyma begins to break
EvansIntercalary Growth in Eleocharis
210
Chlorenchyma
o Vascular
Epidermis
Rib parenchyma
Plate parenchyma
F I G . 3. Percentage of cells dividing (from longitudinal sections).
350
ChJorenchyma
300
o Vascular
Epidermis
250
Rib parenchyma
Plate parenchyma
=1200
150
100
50
4
5
6
Distance from base in mm
F I G . 4. Number of cells dividing (from transverse sections).
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
3
4
5
Distance from base In mm
EvansIntercalary Growth in Eleocharis
211
down and the epidermis differentiates into several types of cell of different
lengths which could not easily be distinguished from one another. No
measurements were made in the vascular tissue because of its complexity.
The results are shown in Fig. 5.
To trace the vascular tissue, longitudinal hand-sections were cut from the
bases of young haulms. These were stained in o-oi per cent aqueous safranin
Cfilorenchyma
Rib parenchyma
Plate parenchyma
20
8
s
I i
12
I < 8
- I
I
I
3
4
5
Distance from base in mm.
F I G . 5. Average cell lengths.
for a few minutes then cleared and mounted in polyvinyl alcohol lactophenol
(Downs, in Gray, 1954). To determine the way water conduction occurs
across the intercalary meristem, young haulms were cut under water just
below the intercalary meristem and the bases immersed in 0-2 per cent
aqueous basic fuchsin. The haulms were placed in strong light for about ten
minutes and then transverse hand-sections were made through the meristem
and at i-cm intervals up the haulm. To determine if the meristem restricted
the rate of water movement, mature haulms were also examined. These were
cut to the length of the immature haulms to give a comparable surface area
for transpiration.
RESULTS
As Buchholz (1920) found, the partial removal of the sheaths necessary in
order to mark the haulm caused death or distortion and the only information
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
Epidermis
24
212
EvansIntercalary Growth in Eleocharis
Cell Division and Cell Elongation
The majority of cell divisions were transverse, leading to the formation of
longitudinal columns of cells. Occasional divisions in the plate and rib
parenchyma and the chlorenchyma were, however, longitudinal. Tangentiallongitudinal divisions were frequent in the epidermis in the first 0-5 mm. of the
internode. These give rise to the fibre initials which divide longitudinally
several times, producing a bundle of fibres approximately six cells in crosssection. This occurs in about every fourth row of epidermal cells around the
haulm. From the 2'5-mm level onwards, about one-quarter of the cell divisions in the epidermis were radial-longitudinal. This was the region in which
the stomata were formed, each stoma mother cell dividing longitudinally three
times to form two guard cells and two subsidiary cells. As there were no
divisions in which the spindle axis was longitudinal, it was assumed that all
divisions here were longitudinal and concerned with stomata formation. The
procambium consists of an outer zone of cells with transverse end walls and
an inner zone of narrower cells with oblique end walls. The cells of the
outer zone, which divide transversely, develop into the vascular parenchyma
and xylem vessels. Divisions of the celb of the inner zone may be transverse,
longitudinal, or longitudinal-oblique. These cells develop into the tracheids,
sieve tubes, and companion cells.
Fig. 3 shows that the percentage of cells dividing increases rapidly to a
maximum level and then diminishes at a slower rate from the base towards the
top of the internode. The percentage of cells dividing and the extent of cell
divisions are greatest in the chlorenchyma and least in the plate parenchyma.
The extent of cell divisions is the same in the two haulms examined except
that in the transversely sectioned haulm the divisions extend 1 mm further
in the chlorenchyma. In all tissues, the cell length (Fig. 5) decreases from
the base towards the top of the internode as the percentage of cells dividing
increases, and increases once more as the percentage of cells dividing decreases.
The chlorenchyma which has the greatest maximum rate of cell division has
the lowest minimum cell length. In the plate parenchyma, cell length decreases
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
gained was that, as will be described below from longitudinal sections, most of
the growth occurred in the first 2 cm of the internode. However, measurement
of the pith lacunae did provide some rough measurement of elongation. In
the mature haulm these lacunae are of approximately the same length between
the same set of ribs. It was found in the region of the meristem that the length
of these lacunae increases steadily to a maximum from 1-3 mm above the
base of the internode, where they are formed by the separation of the layers
of the plate parenchyma. This indicates that elongation of a segment proceeds
at a steady rate with increasing distance from the base of the internode at
least from the i-3-mm level.
No difficulty was experienced in distinguishing cells of the different tissues
except in the first 0-25 mm. In this region they were all of uniform size and
appearance and could be identified only by their position.
EvansIntercalary Growth in Eleocharis
213
DISCUSSION
In Eleocharis the cell divisions within a column of cells showed periodicity.
The cells increased in length up the column to a maximum; there were then
usually two or three cells in various stages of division, and following these a
number of small cells which had apparently divided recently. Continuing up
the column, the cell size increased once more and the cycle was repeated so
that there were alternate groups of long and short cells. However, the different
0S0.2
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
to a minimum but does not increase again, so that, because of tensions set up
by cell elongation in the other tissues, the layers of cells are torn apart, thus
producing the large air spaces.
Vascular Continuity
Fig. 6 shows transverse sections through large and small vascular bundles,
above and below the intercalary meristem and at two positions within it.
There are groups of up to six protoxylem tracheids present in about half of the
bundles in the intercalary meristem. At the upper point of insertion of the
second sheath, there is an abrupt transition in most rows of elements from
xylem to procambium. Only the xylem nearest to the centre of the axis is
continuous. The first-formed protoxylem tracheids, which are the most
stretched, have spiral thickening. Subsequent tracheids have annular thickening and the distance between thickening rings indicates that the tracheids can
stretch approximately tenfold before they collapse to form a xylary lacuna
which increases in size up the internode. New tracheids differentiate from the
procambium at the same rate as the older ones become ineffective through
collapse.
Water Conduction
Immersing the bases of the haulms in stain suggested that only the tracheids
are responsible for conduction since in the meristem no stain was present in
the small bundles lacking mature tissue or in the undifferentiated tissue of the
larger bundles. Attempts to compare the rate of conduction in mature and
immature haulms gave inconclusive results. The upward movement of the
stain was in most instances greater in the mature haulms, but the presence of
the meristem did not appear seriously to limit the conduction of water up the
larger vascular bundles. The transverse connecting strands supply the smaller
bundles which have no mature tissue in the region of the meristem. Transport
occurred along these strands from the large bundles to the small ones some
time after the dye reached that level in the large bundles. This suggested that
diffusion through the intervening xylem parenchyma was slow compared with
its passage up the tracheids, but was much more rapid than its diffusion
through other tissues. The mature vessels also conducted the dye but the
xylary lacunae did not appear to do so. As with the xylem, there appear to be
mature phloem elements present in the larger vascular bundles right through
the meristem. In mature parts of the haulm, the protophloem is crushed but
no lacuna is formed as in the xylem.
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
FIG. 6. Transverse sections of large and small vascular bundles (X 400).
(1) At base; (2) 1 mm above base; (3) 3-5 mm above base; (4) 12-5 mm above base. P =
mature phloem, u = undifferentiated tissue, x = mature xylem. L = xylem lacuna. In 1 and
4 only mature tissue is present. In 2 and 3 (region of cell division), there is some mature xylem
and phloem only in the large bundle. Note the increase in size of the xylem lacuna.
EvansIntercalary Growth in Eleocharis
215
columns of cells were not in phase with one another, and at any level all stages
could be seen, so overall periodicity of cell division would not be expected.
Therefore the irregularities in the graphs of cell division (Figs. 3 and 4) are not
considered to be significant.
Vascular System
The way in which vascular continuity is maintained and water conduction
occurs in Eleocharis is the same as in other species, except that in contrast to the
findings of Buchholz (1920) and Golub and Wetmore (1948) the xylary lacuna
does not appear to be conductive.
The relationship of the vascular tissue to the meristem warrants some attention. In Equisetum, Golub and Wetmore (1948) state that the vascular bundles
pass through the meristematic region, and that the latter does not contribute
to them. Denne (i960) also does not seem to regard the two as being closely
related. Jacobs (1947), on the other hand, has described a zone of cell divisions
in the vascular tissue similar to that in the other tissues in the meristem of
Arachis. In the vascular tissue he noted that about one-third of the mitoses
were transverse and the rest were longitudinal or longitudinal-oblique. In
Eleocharis a parallel situation is found. The important point is whether this
zone of cell divisions should be regarded as a manifestation of normal vascular
growth or as a function of the intercalary meristem. The intercalary meristem
is in form a rib meristem, i.e. the cells are formed in vertical columns by
transverse divisions. In Eleocharis this is also seen to be the case in the
vessels and the vascular parenchyma. In the other xylem and phloem initials
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
Mechanical Support
The mature regions of the haulm are mechanically supported by the bundles
of sub-epidermal fibres. At the base, where the meristematic growth is occurring, the developing fibres are unlikely to fulfil this function, but the haulm is
here supported by the surounding sheaths which are closely appressed to it.
Prat (1935) made extensive measurements of the strength of various portions of
the internodes of grasses. His work showed that the resistance of mature
internodes to cutting was many times that of the basal meristematic region and
that the leaf sheaths considerably increased the resistance. According to Holtturn (1955) the presence of this sheath in monocotyledons permits the continued growth of the stem in a way which would otherwise be impossible. The
case of the flowering stems in dicotyledons is slightly different; in these the
meristematic region (which is usually without a supporting sheath) occurs at
the top of the internode, where it has the least weight to support. It also has
usually completed its elongation before the flowering head matures. Nevertheless, the meristematic region must be mechanically much stronger than in
the case of monocotyledon stems. In these it need only be sufficiently strong
to resist the downward pressure of the stem above. The importance of the
sheaths] in Eleocharis was shown by the death or distortion of the meristem
when part of the sheaths was removed.
2i6
EvansIntercalary Growth in Eleocharis
Transverse Connecting Strands
Although transverse strands connecting adjacent vascular bundles have
been observed by several authors, only Buchholz (1920) noted that they
connect bundles which have mature vascular tissue in the intercalary meristem
with those which do not, and that they conduct water. Kaufman (1959) considered that in Oryza they are composed of tracheids. In Eleocharis they are
much more similar to the vascular parenchyma. It appears that the transverse
connecting strands help to compensate for the restrictions imposed upon
water conduction by the intercalary meristem by increasing the lateral distribution above it. In Eleocharis, although they would seem unnecessary when
the haulm has matured because all the vascular bundles then have continuous
mature xylem, they still appear to function.
ACKNOWLEDGEMENTS
I wish to acknowledge the guidance of Professor W. R. Philipson, Botany
Department, University of Canterbury, under whom this work was carried out.
Appreciation is expressed to Mr. A. C. Glenday, Applied Mathematics
Laboratory, D.S.I.R., for advice on statistical treatment, and to Miss M. E.
Soulsby, D.S.I.R., for photography.
LITERATURE C I T E D
BROWN, R., and RICKLESS, P., 1949. A New Method for the Study of Cell Division and Cell
Extension with some Preliminary Observations on the Effect of Temperature and of
Nutrients. Proc. Roy. Soc. B, 136, 110-25.
BUCHHOUZ, M., 1920. Uber die Wasserleitungsbahnen in den interkakren Wachstumszonen
monokotyler Sprosse. Flora, 114, 11986.
DENNE, M. P., i960. Leaf Development in Nardstus pseudonarcissus L. II. The Comparative
Development of Scale and Foliage Leaves. Ann. Bot., NA 34, 32-47.
ESAU, K., 1953. Plant Anatomy. John Wiley & Sons, New York.
GOLUB, S. J., and WETMORE, R. H., 1948. Studies of Development in the Vegetative Shoot of
Equisetum arvense L. II. The Mature Shoot. Amer.J. Bot. 35, 767-81.
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
divisions are mainly longitudinal-oblique (although both longitudinal and
transverse divisions do occur) and the cells are not formed in columns but
because of the shape of their end walls overlap somewhat. Some of the longitudinal divisions would be those giving rise to the sieve cells and companion
cells. The most significant fact, however, is that cell division in this region is
almost entirely directed towards the elongation of the vascular bundle. There
is some radial increase of vascular tissue but this is only in compensation for
the mature tissue being destroyed through stretching. It is considered that
the extent of cell divisions in the vascular tissue in the meristematic region of
Eleocharis, and probably also in Arachis, is such that they should be regarded
as part of the intercalary meristem. That continuous mature tissue is present in
only some of the bundles in Eleocharis and also other species (Buchholz, 1920;
Denne, i960) is regarded as support for this view.
EvansIntercalary Growth in Eleocharis
217
SACHS, R. M., LANG, A., BRBTZ, C. F., and ROACH, J., i960. Shoot Histogenesis: Subapical
Meristematic Activity in a Caulescent Plant and the Action of Gibberellic Acid and Amo1618. Amer. J. Bot. 47, 260-6.
SCHMAUUSS, K., 1930. Untersuchungen iiber die interkalare Wachstumszone an Glumifloren
und dicotylen BlQtenschaften. Flora, N.s. 24, 333-66.
Downloaded from http://aob.oxfordjournals.org/ at University of Edinburgh on October 14, 2016
GRAY, P., 1954. The Microtomist's Formulary and Guide. Constable & Co., London.
HABERLANDT, G., 1914. Phytiological Plant Anatomy. Trans. Drummond, M. Macmillan &
Co., London.
HOLTTUM, R. E., 1955. Growth Habits of MonocotyledonsVariations on a Theme. Phytomorphology, 5, 309-413.
JACOBS, W. P., 1947. The Development of the Gynophore of the Peanut Plant, Arachu hypogaea
L. I. The Distribution of Mitoses, the Region of Greatest Elongation, and the Maintenance of Vascular Continuity in the Intercalary Meristem. Ainer. J. Bot. 34, 361-70.
KAUFMAN, P. B., 1959. Development of the Shoot of Oryza tativa L. II. Leaf Histogenesis.
Phytomorphology, 9, 277-311.
MLLTKNYI, L., 1931. Histologisch-Entwicklungsgeschichtliche Untersuchungen an Getreidearten. Bot. KSzl. 38, 1-51.
PERCIVAL, J., 1921. The Wheat Plant. Duckworth & Co., London.
PLOWMAN, A. B., 1006. The Comparative Anatomy and Phylogeny of the Cyperaceae. Ann.
Bot. ao, 1-33.
PRAT, H., 1935. Recherches sur la structure et la mode de croissance des chaumes. Ann.
Sci. Nat. Bot. x, 17, 81-145.
S-ar putea să vă placă și
- Thoracic and Coracoid Arteries In Two Families of Birds, Columbidae and HirundinidaeDe la EverandThoracic and Coracoid Arteries In Two Families of Birds, Columbidae and HirundinidaeÎncă nu există evaluări
- Origin and Development of The Vascular Bundle of Zea Mays L.Document9 paginiOrigin and Development of The Vascular Bundle of Zea Mays L.Afrida NandaÎncă nu există evaluări
- Lab 1Document8 paginiLab 1JeemCarloFagelaPulaÎncă nu există evaluări
- Journal of Entomology and Zoology Volume Eleven, Number Two, June 1919De la EverandJournal of Entomology and Zoology Volume Eleven, Number Two, June 1919Încă nu există evaluări
- New Insights on the Development of the Vascular SystemDe la EverandNew Insights on the Development of the Vascular SystemÎncă nu există evaluări
- Anatomy of Palms R Hap IsDocument22 paginiAnatomy of Palms R Hap IsBelem AlejandroÎncă nu există evaluări
- Stelar System of PlantDocument6 paginiStelar System of Plantworlds tourÎncă nu există evaluări
- Equisetum: The Horsetail PlantDocument7 paginiEquisetum: The Horsetail PlantChithra VijayanÎncă nu există evaluări
- LAB EXERCISE 8 - 10 MM Pig Embryo - Mesodermal DerivativesDocument5 paginiLAB EXERCISE 8 - 10 MM Pig Embryo - Mesodermal DerivativesGerald Angelo DeguinioÎncă nu există evaluări
- Botn 21n - Exercise 12 - Stems and Secondary GrowthDocument17 paginiBotn 21n - Exercise 12 - Stems and Secondary GrowthABIEGAIL LAMBERTÎncă nu există evaluări
- 307 48 ET V1 S1 - File1 PDFDocument22 pagini307 48 ET V1 S1 - File1 PDFaaaatniÎncă nu există evaluări
- Anatomy RenalDocument12 paginiAnatomy RenalAnnisaastikaÎncă nu există evaluări
- Air Tissue Sifton 1945Document36 paginiAir Tissue Sifton 1945Pilar7100Încă nu există evaluări
- Microsurgical Anatomy of Posterior Fossa VeinsDocument43 paginiMicrosurgical Anatomy of Posterior Fossa VeinsCan EkerÎncă nu există evaluări
- The Comparative Morphology of the Winteraceae VI. Vascular Anatomy of the Flowering ShootDocument18 paginiThe Comparative Morphology of the Winteraceae VI. Vascular Anatomy of the Flowering Shootbarerahjaved479Încă nu există evaluări
- The Origin and Conduction of The Heart BeatDocument43 paginiThe Origin and Conduction of The Heart BeatDjanino Fernandes0% (1)
- Un Estudio de TargioniaDocument13 paginiUn Estudio de TargioniaramongonzaÎncă nu există evaluări
- Strucrtures of Plant PartsDocument95 paginiStrucrtures of Plant Partsfatima100% (1)
- On A New Organ in The Lycoridea, and On The Nephridium in Nereis Diversicolor, O. P. MullDocument18 paginiOn A New Organ in The Lycoridea, and On The Nephridium in Nereis Diversicolor, O. P. Mullelfan titiansyahÎncă nu există evaluări
- Young Stems and RootsDocument11 paginiYoung Stems and RootsHailey CoutuÎncă nu există evaluări
- Plant Physiology LAB REPORT XDocument5 paginiPlant Physiology LAB REPORT XBoyd benson kayomboÎncă nu există evaluări
- Anatomy and Properties of Bamboo CulmsDocument198 paginiAnatomy and Properties of Bamboo Culmsvin_3ldÎncă nu există evaluări
- Vascular Tissue DifferentiationDocument9 paginiVascular Tissue DifferentiationC'estMoiÎncă nu există evaluări
- Fig. 2 B, (Fig. at Of: Histology EyeDocument26 paginiFig. 2 B, (Fig. at Of: Histology EyeraniaÎncă nu există evaluări
- Pteridophytes-Telome Theory and Stelar Evolution by Dr. Prabha DhondiyalDocument39 paginiPteridophytes-Telome Theory and Stelar Evolution by Dr. Prabha DhondiyalAbhishek Singh ChandelÎncă nu există evaluări
- The Vascular Endothelium: A Holistic Approach for OncologyDe la EverandThe Vascular Endothelium: A Holistic Approach for OncologyÎncă nu există evaluări
- NHLBI Workshop Summary: The Mysterious Pulmonary Brush CellDocument4 paginiNHLBI Workshop Summary: The Mysterious Pulmonary Brush CellcholoconvertiÎncă nu există evaluări
- ST Joseph's Institution (Senior School) IBDP1 HL Biology PracticalDocument5 paginiST Joseph's Institution (Senior School) IBDP1 HL Biology PracticalSean NgÎncă nu există evaluări
- Top 3 Shoot Apical Meristem TheoriesDocument11 paginiTop 3 Shoot Apical Meristem TheoriesAbhimanyu Pandey100% (2)
- Theories of Shoot Apical Meristem OKOKOKDocument8 paginiTheories of Shoot Apical Meristem OKOKOKShameel PervezÎncă nu există evaluări
- A4. Thilander H, Bloom G. Cell Contacts in Oral Epithelia. J Perio Res 1968Document16 paginiA4. Thilander H, Bloom G. Cell Contacts in Oral Epithelia. J Perio Res 1968Daniel CubasÎncă nu există evaluări
- The Biology of Lumbriculus variegatusDocument4 paginiThe Biology of Lumbriculus variegatusklipschthx0Încă nu există evaluări
- Carl Claus - XIII.-On The Organization of The CypridesDocument5 paginiCarl Claus - XIII.-On The Organization of The CypridestorigÎncă nu există evaluări
- Chapter 140: Cochlear Anatomy and Central Auditory Pathways Peter A. Santi, Patrizia ManciniDocument20 paginiChapter 140: Cochlear Anatomy and Central Auditory Pathways Peter A. Santi, Patrizia ManciniJustkillme AlifÎncă nu există evaluări
- Senecio Grandidentatus Ledeb. (Asteraceae) : Anatomical Aspects of The Stem and Leaf ofDocument7 paginiSenecio Grandidentatus Ledeb. (Asteraceae) : Anatomical Aspects of The Stem and Leaf ofBlinda Raluca MariaÎncă nu există evaluări
- Mephitinve, Spilogale, Conepatus, Me Phitis. Mephgtis Conetatus. Mefhlt S: M. Macrura, .M. 2utorlus, MethitlcaDocument18 paginiMephitinve, Spilogale, Conepatus, Me Phitis. Mephgtis Conetatus. Mefhlt S: M. Macrura, .M. 2utorlus, MethitlcaLouis CyferÎncă nu există evaluări
- Anatomia Foramem IntervertebralDocument11 paginiAnatomia Foramem IntervertebralmlmudoÎncă nu există evaluări
- Gross Anatomy II Lecture on Heart and Vascular SystemDocument13 paginiGross Anatomy II Lecture on Heart and Vascular SystemSantosh Bhandari100% (2)
- Journal of Morphology SpermwarteryDocument14 paginiJournal of Morphology SpermwarteryJustin RamonÎncă nu există evaluări
- BOTANYDocument5 paginiBOTANYPrincess Fay LopezÎncă nu există evaluări
- PleuraDocument6 paginiPleurasjs6r8wwv9Încă nu există evaluări
- Jurnal - Christopher Muhamad Zildan Holliday - 215090107111029 - Kelas ADocument17 paginiJurnal - Christopher Muhamad Zildan Holliday - 215090107111029 - Kelas AChristopher Muhamad Zildan HÎncă nu există evaluări
- The Stem and Root Anatomy of Sanmiguelia lewisiiDocument41 paginiThe Stem and Root Anatomy of Sanmiguelia lewisiiShafa NnisaÎncă nu există evaluări
- Miología Apendicular Del Procyon Cancrivorus (Windle, 1888)Document9 paginiMiología Apendicular Del Procyon Cancrivorus (Windle, 1888)Sebastian ChungaÎncă nu există evaluări
- Lecture Learning Objectives for BIO 210 SystemsDocument14 paginiLecture Learning Objectives for BIO 210 SystemsAmber DavisÎncă nu există evaluări
- Ventrilces BrainDocument18 paginiVentrilces BrainVamsi KrishnaÎncă nu există evaluări
- Human Anat 2 Year Volume 4Document122 paginiHuman Anat 2 Year Volume 4HARSHINI PANDIANÎncă nu există evaluări
- VeinsDocument5 paginiVeinsMaximos ManiatisÎncă nu există evaluări
- NyctaDocument18 paginiNyctaAghila SamjiÎncă nu există evaluări
- Tissue and Tissue Systems - STD 9Document14 paginiTissue and Tissue Systems - STD 9Syamala Natarajan100% (1)
- Blood Vessels, AnatomyDocument20 paginiBlood Vessels, AnatomyAbu BakarÎncă nu există evaluări
- ANATOMICAL STUDY OF ANOMALY STRUCTURE IN CucurbitaDocument5 paginiANATOMICAL STUDY OF ANOMALY STRUCTURE IN CucurbitaGaluh Novita Ayu0% (1)
- Chapter 6 Anatomy of Flowering PlantsDocument8 paginiChapter 6 Anatomy of Flowering Plantsswayamdey71Încă nu există evaluări
- Life Cycle and Reproduction of Anthoceros LiverwortDocument24 paginiLife Cycle and Reproduction of Anthoceros LiverwortListiyani Putri AzzahroÎncă nu există evaluări
- Equisetum 180922130237Document34 paginiEquisetum 180922130237Ján ŠormanÎncă nu există evaluări
- Structure: Plant Form and FunctionDocument12 paginiStructure: Plant Form and FunctionPhilippines QatarÎncă nu există evaluări
- Botany I PDFDocument4 paginiBotany I PDFFakiha NadeemÎncă nu există evaluări
- Tamil Nadu 11th Standard Botany Practical ManualDocument43 paginiTamil Nadu 11th Standard Botany Practical Manualsri thamizhan80% (85)
- Northern beech trunk in autumnDocument4 paginiNorthern beech trunk in autumnmansi bavliyaÎncă nu există evaluări
- Exercise 7. Leaf: Distinctive CharacteristicsDocument8 paginiExercise 7. Leaf: Distinctive CharacteristicsTers MedinaÎncă nu există evaluări
- PLANTS: Types, Parts, Uses & How They Make FoodDocument22 paginiPLANTS: Types, Parts, Uses & How They Make FoodSurbhi Bhatia100% (1)
- Aquatic Plants AdaptDocument2 paginiAquatic Plants AdaptKrystallane ManansalaÎncă nu există evaluări
- Transport System in PlantDocument16 paginiTransport System in PlantAbu-Omar50% (4)
- Practical: I Total Marks 50 Five Questions Will Be Asked Each Question Carries 10 MarksDocument3 paginiPractical: I Total Marks 50 Five Questions Will Be Asked Each Question Carries 10 MarksCrazy GamerÎncă nu există evaluări
- Identify The Plant Organ ActivityDocument5 paginiIdentify The Plant Organ Activityapi-246973610Încă nu există evaluări
- 1 - Morphology of The Rice PlantDocument31 pagini1 - Morphology of The Rice PlantThe Municipal Agriculturist100% (1)
- Morphology and Anatomy of Flowering PlantsDocument3 paginiMorphology and Anatomy of Flowering PlantsAyako HayashidaÎncă nu există evaluări
- Vegetative OrgansDocument68 paginiVegetative OrgansAngel GoÎncă nu există evaluări
- Exer 9 LeavesDocument3 paginiExer 9 LeavesColleen Dejan100% (1)
- Struktur Anatomi Batang Tiga Jenis Rotan Genus Calamus Dari Kawasan Tembawang Kabupaten Kubu RayaDocument6 paginiStruktur Anatomi Batang Tiga Jenis Rotan Genus Calamus Dari Kawasan Tembawang Kabupaten Kubu RayaZahra Nur AzizahÎncă nu există evaluări
- AnfistumDocument207 paginiAnfistumjosefannyhahaÎncă nu există evaluări
- ELSC Week13 Plant Survival PDFDocument43 paginiELSC Week13 Plant Survival PDFarriane BaladadÎncă nu există evaluări
- Exercise 2Document7 paginiExercise 2Lendie VillÎncă nu există evaluări
- CBSE Class 11 Biology Chapter 6 Anatomy of Flowering Plants Revision NotesDocument72 paginiCBSE Class 11 Biology Chapter 6 Anatomy of Flowering Plants Revision Noteschakrabortytrisha.2006Încă nu există evaluări
- Bryanston Primary School: Natural Science Cycle TestDocument8 paginiBryanston Primary School: Natural Science Cycle TestErinÎncă nu există evaluări
- Chapter 1 (Part 1) - Plant Anatomy & Physiology PDFDocument89 paginiChapter 1 (Part 1) - Plant Anatomy & Physiology PDFMohammed Ridzuwan94% (16)
- NEET Level Test Questions (29-Jan-23Document25 paginiNEET Level Test Questions (29-Jan-23Unit VIIÎncă nu există evaluări
- Modul Perfect Score SBP Biology SPM 2013 Question and SchemeDocument283 paginiModul Perfect Score SBP Biology SPM 2013 Question and SchemeCikgu Faizal87% (15)
- Cells-Tissues Student GuideDocument2 paginiCells-Tissues Student GuideMark LozoÎncă nu există evaluări
- Plant Body Plant Body: Why Is Histology Important?Document6 paginiPlant Body Plant Body: Why Is Histology Important?ZyreeneNicoleÎncă nu există evaluări
- Plant Cell Specialization and Tissue DifferentiationDocument10 paginiPlant Cell Specialization and Tissue DifferentiationAndrea CruzÎncă nu există evaluări
- 8384 ST PDFDocument20 pagini8384 ST PDFJohn Dave Francisco100% (1)
- Bio MaterialsDocument26 paginiBio MaterialsGeorge SingerÎncă nu există evaluări
- Anatomy of Flowering PlantsDocument19 paginiAnatomy of Flowering PlantsPrerak PatelÎncă nu există evaluări
- CucurbitaDocument5 paginiCucurbitaapi-49891619Încă nu există evaluări