Documente Academic
Documente Profesional
Documente Cultură
Closon
Încărcat de
rahi6055Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Closon
Încărcat de
rahi6055Drepturi de autor:
Formate disponibile
Closon
Betamethasone dipropionate BP 0.5mg + Clotrimazole BP 10mg
COMPOSITION
Closon cream : Each gram cream contains 0.50 mg Betamethasone as Dipropionate and 10 mg
clotrimazole.
PHARMACOLOGY
Clotrimazole is a broad-spectrum antifungal agent used for the treatment of superficial infections
caused by species of pathogenic dermatophytes, yeasts and Malassezia furfur. The mechanism of
action involves inhibition of the synthesis of ergosterol, a major sterol in the fungal cell
membrane. This leads to instability of the cell membrane and eventual death of the fungus.
Betamethasone dipropionate is a corticosteroid with anti-inflammatory, antipruritic, and
vasoconstrictive properties. But the exact mechanism of action of corticosteroids is not clearly
known.
INDICATION
Closoncream is indicated for the topical treatment of inflammatory dermal infections like tinea
pedis, tinea cruris, tinea corporis, etc.
DOSAGE AND ADMINISTRATION
Sufficient cream should be applied onto the affected and surrounding skin areas twice a day, in
the morning and evening, for 2 weeks in tinea cruris and tinea corporis and for 4 weeks in tinea
pedis. The use of cream for longer than four weeks is not recommended.
CONTRAINDICATION AND PRECAUTION
The preparation is contraindicated to those patients who are sensitive to any of its components or
to other corticosteroids or to imidazoles. If irritation or sensitization develops with the use of the
cream, treatment should be discontinued and appropriate therapy instituted.
The cream is contraindicated in facial rosacea, acne vulgaris, perioral dermatits, perianal and
genital pruritus, napkin eruptions and bacterial or viral infections. Systemic absorption of topical
corticosteroides can produce reversible hypothalmic-pituitary-adrenal (HPA) axis suppression. If
HPA axis suppression is noted, an attempt should be made to withdraw the drug or to reduce the
frequency of application. Pediatric patients may be more susceptible to systemic toxicity from
equivalent doses due to their large skin surface to body mass ratios.
SIDE EFFECT
Adverse reactions reported for the preparation in clinical trials were paresthesia in 1.9% of
patients, rash, edema and secondary infection, each in less than 1% of patients. Other adverse
reactions reported with the preparation were burning and dry skin in 1.6% of patients and
stinging in less than 1% of patients.
USE IN PREGNANCY AND LACTATION
There is inadequate evidence of safety in pregnancy. Clotrimazole has no teratogenic effect in
animals, but is foetotoxic at high oral doses. Topical administration of corticosteroids to pregnant
animals can cause abnormalities of fetal development. Hence the cream should only be used in
pregnancy, if the benefit justifies the potential risk to the fetus and such use should not be
extensive, i.e. in large amounts or for long periods. It is not known whether the components of
the preparation are excreted in human milk and therefore caution should be exercised when
treating nursing mothers.
USE IN CHILDREN
The safety and effectiveness of the preparation has not been established in children below the age
of 12 years.
OVERDOSE
Acute overdose with the cream is unlikely and would not be expected to lead to a life-threatening
situation. The cream should not be used for longer than the prescribed time period.
STORAGE CONDITION
Store at or below 30C. Do not freeze.
HOW SUPPLIED
Closon cream: Each pack has a tube containing 10 gm of the cream.
Manufactured by:
MEDICON Pharmaceuticals Ltd.
Mirpur, Dhaka, Bangladesh
S-ar putea să vă placă și
- Welcome To The PresentationDocument23 paginiWelcome To The Presentationrahi6055Încă nu există evaluări
- Bogra Depot Office: 654, Hafizar Rahmanroad, Jaleswaritola, BograDocument1 paginăBogra Depot Office: 654, Hafizar Rahmanroad, Jaleswaritola, Bograrahi6055Încă nu există evaluări
- IncentiveDocument2 paginiIncentiverahi6055Încă nu există evaluări
- Doctor Visit SccheduleDocument1 paginăDoctor Visit Scchedulerahi6055Încă nu există evaluări
- Bsmmu PG Hospital Investigations FeesDocument5 paginiBsmmu PG Hospital Investigations Feesrahi6055Încă nu există evaluări
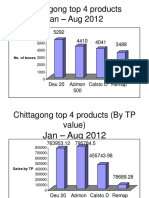
- Chittagong Top 4 Products Jan - Aug 2012: No. of BoxesDocument4 paginiChittagong Top 4 Products Jan - Aug 2012: No. of Boxesrahi6055Încă nu există evaluări
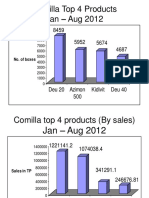
- Comilla Top 4 ProductsDocument4 paginiComilla Top 4 Productsrahi6055Încă nu există evaluări
- Monicopharma Limited AM, Evaluation Test-2011: Time: 40 Minutes Marks: 40Document3 paginiMonicopharma Limited AM, Evaluation Test-2011: Time: 40 Minutes Marks: 40rahi6055Încă nu există evaluări
- Implications of SetroDocument2 paginiImplications of Setrorahi6055Încă nu există evaluări
- Bac and CefurDocument1 paginăBac and Cefurrahi6055Încă nu există evaluări
- Alfex Tab and PFSDocument2 paginiAlfex Tab and PFSrahi6055Încă nu există evaluări
- Leave AplicationDocument1 paginăLeave Aplicationrahi6055Încă nu există evaluări
- Ana Sales FinalDocument1 paginăAna Sales Finalrahi6055Încă nu există evaluări
- Training Manual On Kidivit 200mlDocument11 paginiTraining Manual On Kidivit 200mlrahi6055Încă nu există evaluări
- AlminDocument1 paginăAlminrahi6055Încă nu există evaluări
- Employment (Academic) : Application ForDocument7 paginiEmployment (Academic) : Application Forrahi6055Încă nu există evaluări
- Atocon - 01 02.10.16Document1 paginăAtocon - 01 02.10.16rahi6055Încă nu există evaluări
- RixolDocument2 paginiRixolrahi6055Încă nu există evaluări
- MebelDocument2 paginiMebelrahi6055Încă nu există evaluări
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5795)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (345)
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (74)
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (121)
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)
- Guiding For HospitalDocument14 paginiGuiding For HospitalAnkit Bhatia100% (1)
- Discovering Brainwaves - Beta, Alpha, Theta and DeltaDocument4 paginiDiscovering Brainwaves - Beta, Alpha, Theta and DeltaBer Salazar JrÎncă nu există evaluări
- Non-Pharmacologic Pain Management in OrthodonticsDocument21 paginiNon-Pharmacologic Pain Management in OrthodonticsDr. Kanak100% (1)
- Correlation of Retinal Structural Alteration With Retinal Sensitivity Loss: A Prospective StudyDocument26 paginiCorrelation of Retinal Structural Alteration With Retinal Sensitivity Loss: A Prospective StudyAbhishek KothariÎncă nu există evaluări
- Borderline Personality Disorder 2018Document1 paginăBorderline Personality Disorder 2018Nata Papanatas100% (1)
- Brachial PlexusDocument42 paginiBrachial PlexusPrince DuÎncă nu există evaluări
- Levetiracetam in Refractory Pediatric EpilepsyDocument11 paginiLevetiracetam in Refractory Pediatric EpilepsyAdlinaÎncă nu există evaluări
- Triage in Mental Health A New Model For Acute Inpatient PsychiatryDocument6 paginiTriage in Mental Health A New Model For Acute Inpatient PsychiatryAmit ChawlaÎncă nu există evaluări
- Chapter 14 PDFDocument8 paginiChapter 14 PDFShei BariÎncă nu există evaluări
- ADR Form PDFDocument2 paginiADR Form PDFCha Tuban DianaÎncă nu există evaluări
- Temporal Sphenoidal Line: T4 Gall BladderDocument3 paginiTemporal Sphenoidal Line: T4 Gall Bladdertaichi7Încă nu există evaluări
- Background KarunkelDocument2 paginiBackground KarunkeldatascribdyesÎncă nu există evaluări
- IV Fluid Replacement TherapyDocument12 paginiIV Fluid Replacement TherapyKamran Sheraz100% (1)
- DiureticsDocument16 paginiDiureticsvivianÎncă nu există evaluări
- Naveen Kumar.M M.Pharmacy (Pharmaceutics) - 10Y81SO310Document29 paginiNaveen Kumar.M M.Pharmacy (Pharmaceutics) - 10Y81SO310Benish RasheedÎncă nu există evaluări
- HematuriaDocument42 paginiHematuriaWasim R. IssaÎncă nu există evaluări
- Understanding Neurology - A Problem-Orientated Approach PDFDocument241 paginiUnderstanding Neurology - A Problem-Orientated Approach PDFrys239Încă nu există evaluări
- Drugs TableDocument2 paginiDrugs TableDaryl VillamaterÎncă nu există evaluări
- Biomechanical Management of Children and Adolescents With Down SyndromeDocument8 paginiBiomechanical Management of Children and Adolescents With Down SyndromeMihaela NițulescuÎncă nu există evaluări
- Mental Health Reflection 1Document5 paginiMental Health Reflection 1nasir khan100% (1)
- Appendix and Cecum!Document13 paginiAppendix and Cecum!SilvanaPutriÎncă nu există evaluări
- Laparoscopic Surgery Principles and Procedures Second Edition Revised and Expanded PDFDocument624 paginiLaparoscopic Surgery Principles and Procedures Second Edition Revised and Expanded PDFdrnica100% (1)
- Shibly Rahman PacesDocument356 paginiShibly Rahman PacesMohammadAbdurRahmanÎncă nu există evaluări
- Orthopaedic Study Guide 2014Document126 paginiOrthopaedic Study Guide 2014Yip Kar Jun100% (1)
- Treatment-Guidelines Cornelia de LangeDocument8 paginiTreatment-Guidelines Cornelia de Langeirsyad120Încă nu există evaluări
- Case Study P.TDocument5 paginiCase Study P.TSharon WilliamsÎncă nu există evaluări
- SE5l, Medical-Surgical Review Curse PowerPointDocument104 paginiSE5l, Medical-Surgical Review Curse PowerPointBluebloodboy100% (2)
- Manual Usuario Hill-Rom Care Assist - 118186Document64 paginiManual Usuario Hill-Rom Care Assist - 118186Eugenio MilianÎncă nu există evaluări
- Perspective of Facial Esthetics in Dental Treatment PlanningDocument13 paginiPerspective of Facial Esthetics in Dental Treatment PlanninganasbukhariÎncă nu există evaluări