Documente Academic
Documente Profesional
Documente Cultură
Testing Negative Ions
Încărcat de
Gail AidDrepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Testing Negative Ions
Încărcat de
Gail AidDrepturi de autor:
Formate disponibile
Inspirational chemistry
177
Testing for negative ions
Index 7.3.1
2 sheets
Index 7.3.2
1 sheet
This activity is in two parts in the first, students make observations while carrying out
the tests for various negative ions. In the second, they use their observations to help
them identify the negative ions present in a number of unknown solutions. To make
the second part of the exercise more challenging, tests for positive ions could be
introduced and students could be asked to identify both the positive and negative ions
present in a solution.
Equipment required
The exact concentrations of the test solutions are not important. Use approximately
0.10.5 mol dm3 for salt solutions and 0.51.0 mol dm3 for acid solutions, except for
nitric acid, which is corrosive at such concentrations (use 0.4 mol dm3 instead).
Test-tubes
Dropping pipettes (these can be used just for the carbon dioxide testing or also for
dispensing solutions; if the latter, far more pipettes will be required)
Nitric acid 0.4 mol dm3 (Irritant)
3
Silver nitrate solution 0.1 mol dm
Barium chloride solution 0.1 mol dm3 (Harmful)
Hydrochloric acid
Aluminium powder (Highly flammable)
3
Sodium hydroxide solution less than 0.5 mol dm
Limewater
Red litmus paper
3
Ammonia solution 0.4 mol dm .
For the initial observations
Sodium or potassium chloride solution
Sodium or potassium bromide solution
Sodium or potassium iodide solution
Sulfate solution, eg sodium sulfate
(Irritant)
178
Inspirational chemistry
Carbonate solution, eg potassium carbonate
Nitrate solution, eg potassium nitrate.
For testing unknowns
The number of unknowns required depends on the time available. It is a good idea to
use at least four solutions to ensure students are challenged. Label the solutions A, B,
C etc and make sure you know which is which.
Health and safety
Wear eye protection.
Barium chloride solid is toxic; the 0.1 mol dm3 solution is harmful. Wash your hands
after use and warn students to do the same.
Ammonia solution is an irritant when concentrated but not at the concentrations used
by students in this activity. However, it can give off ammonia vapour, which can
irritate the eyes and lungs. Keep the lid on the bottle when not in use.
Nitric acid is an irritant.
Silver nitrate solution can stain skin and clothes.
Further problem solving ideas
There are several suggestions in C. Wood, Creative Problem Solving in Chemistry,
London: Royal Society of Chemistry, 1993.
Inspirational chemistry
179
Tests for negative ions expected observations
Negative ion
Cl
chloride
Test
Observations
Add a few drops of dilute nitric acid
followed by a few drops of silver nitrate
solution. Let the mixture stand for a
few minutes and then add some
ammonia solution.
A white precipitate forms which discolours on
standing. The precipitate is soluble in ammonia
solution.
Br
bromide
A cream precipitate forms which discolours a little
on standing. The precipitate is slightly soluble in
ammonia solution.
I
iodide
A yellow precipitate forms which does not
discolour on standing. The precipitate is insoluble
in ammonia solution.
SO42
sulfate
Add a few drops of barium chloride
solution and then a few drops of
hydrochloric acid.
A white precipitate forms.
CO32
carbonate
Put a small amount of limewater into
a test-tube (no more than 1 cm3). Put
your sample in a separate test-tube and
add a few drops of hydrochloric acid.
Using a pipette, collect the gas given off
and bubble it through the limewater.
(Note: you can also do this test on a
solid sample.)
Bubbles of gas form. The gas turns the limewater
milky, which shows that it is carbon dioxide.
NO3
nitrate
Add a few drops of sodium hydroxide
solution and a little aluminium powder.
Warm the solution in a Bunsen flame
and test any gas given off using red
litmus paper.
A gas is given off which turns the litmus
blue. This shows that the gas is ammonia.
Equations
NaCl(aq) + AgNO3(aq) NaNO3(aq) + AgCl(s)
Cl(aq) + Ag+(aq) AgCl(s) (and similarly for Br and I)
Na2SO4(aq) + BaCl2(aq) 2 NaCl(aq) + BaSO4(s)
2
2+
SO4 (aq) + Ba (aq) BaSO4(s)
2 HCl(aq) + Na2CO3(aq) 2 NaCl(aq) + H2O(l) + CO2(g)
2
+
CO3 (aq) + 2H (aq) CO2(g) + H2O(l)
For completeness, the reaction with the nitrate ion is shown below. It is unlikely that
students will be able to construct this for themselves and the student sheet does not ask
them to do so.
8Al(s) + 3NO3(aq) + 5OH(aq) + 18H2O(l) 8[Al(OH)4](aq) + 3NH3(g)
Testing for negative ions
making observations
This activity is in two parts. In the first part you observe the reactions of various negative ions and
in the second you use those observations to identify unknown solutions.
Use the table Tests for negative ions to record your observations during each test. Use a clean
test-tube each time or wash up thoroughly between tests using distilled or deionised water to
avoid contamination. Use a small portion of the test solution each time (no more than 1 cm3).
Write balanced symbol equations for the reaction that occurs in each of the tests (except the test
for a nitrate).
Health and safety
Barium chloride solution is harmful; wash your hands after use.
Sodium hydroxide is an irritant.
Ammonia solution can give off ammonia vapour, which can irritate the eyes and lungs. Keep
the lid on the bottle when not in use.
Nitric acid is an irritant.
Silver nitrate can stain skin and clothes.
TO C
OP
Y
Testing for negative ions page 1 of 2, Index 7.3.1
PHO
Wear eye protection. Take extra care when dealing with unknown solutions. At the
concentrations used in this experiment:
Tests for negative ions
Test
Observations
Cl
chloride
Add a few drops of dilute nitric acid
followed by a few drops of silver nitrate
solution. Let the mixture stand for a
few minutes and then add some
ammonia solution.
Br
bromide
Add a few drops of dilute nitric acid
followed by a few drops of silver nitrate
solution. Let the mixture stand for a
few minutes and then add some
ammonia solution.
I
iodide
Add a few drops of dilute nitric acid
followed by a few drops of silver nitrate
solution. Let the mixture stand for a
few minutes and then add some
ammonia solution.
SO42
sulfate
Add a few drops of barium chloride
solution and then a few drops of
hydrochloric acid.
CO32
carbonate
Put a small amount of limewater into
a test-tube (no more than 1 cm3). Put
your sample in a separate test tube and
add a few drops of hydrochloric acid.
Using a pipette, collect the gas given off
and bubble it through the limewater.
(Note: you can also do this test on a
solid sample.)
NO3
nitrate
Add a few drops of sodium hydroxide
solution and a little aluminium powder.
Warm the solution in a Bunsen flame
and test any gas given off using red
litmus paper.
TO C
OP
Y
Testing for negative ions page 2 of 2, Index 7.3.1
PHO
Negative ion
Testing for negative ions
identifying unknowns
Using the observations chart you made in Testing for negative ions making observations,
test the unknown solutions provided and identify the negative ions present. Make careful
observations, including any negative results. You may need to try a number of tests before you
get a positive result. Design a table to record your observations. You may wish to use the
headings: Unknown sample; Test tried; Observations; and Conclusion.
Health and safety
Wear eye protection. Take extra care when dealing with unknown solutions. At the concentration
used in this experiment:
Barium chloride solution is harmful; wash your hands after use.
Sodium hydroxide is an irritant.
Ammonia solution can give off ammonia vapour, which can irritate the eyes and lungs. Keep
the lid on the bottle when not in use.
Nitric acid is an irritant.
Silver nitrate can stain skin and clothes.
TO C
OP
Y
Testing for negative ions unknowns page 1 of 1, Index 7.3.2
PHO
S-ar putea să vă placă și
- Phytochem OnlyDocument4 paginiPhytochem OnlyGail AidÎncă nu există evaluări
- Clint Ox Lab Anti MotilityDocument3 paginiClint Ox Lab Anti MotilityGail AidÎncă nu există evaluări
- June 2016 Board Exam QuestionsDocument24 paginiJune 2016 Board Exam QuestionsFanBayÎncă nu există evaluări
- Adventist Medical Center College Andres Bonifacio Ave., San Miguel, Iligan City Department of PharmacyDocument5 paginiAdventist Medical Center College Andres Bonifacio Ave., San Miguel, Iligan City Department of PharmacyGail AidÎncă nu există evaluări
- qc2 ch1Document6 paginiqc2 ch1Gail AidÎncă nu există evaluări
- ChapterDocument3 paginiChapterGail AidÎncă nu există evaluări
- Plankton PPT For SurigaoDocument24 paginiPlankton PPT For SurigaoGail AidÎncă nu există evaluări
- 1 Ionic BondsDocument35 pagini1 Ionic BondsNear ChemÎncă nu există evaluări
- 04 Drug Safety Final 08Document40 pagini04 Drug Safety Final 08Gail AidÎncă nu există evaluări
- Pharmaceutical Jurisprudence and EthicsDocument5 paginiPharmaceutical Jurisprudence and EthicsGail Aid100% (1)
- Risk Factors For Common Chronic DiseasesDocument14 paginiRisk Factors For Common Chronic DiseasesGail AidÎncă nu există evaluări
- Shig Ellos IsDocument15 paginiShig Ellos IsGail AidÎncă nu există evaluări
- Sds Navigo 70 MCL enDocument10 paginiSds Navigo 70 MCL enGail AidÎncă nu există evaluări
- Health EthicsDocument18 paginiHealth EthicsGail AidÎncă nu există evaluări
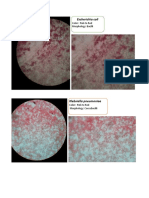
- Escherichia Coli: Color: Pink To Red Morphology: BacilliDocument1 paginăEscherichia Coli: Color: Pink To Red Morphology: BacilliGail AidÎncă nu există evaluări
- CEBUDocument6 paginiCEBUGail AidÎncă nu există evaluări
- 2423L6Document11 pagini2423L6Salman Dinani100% (1)
- Who Trs 908-Annex9Document12 paginiWho Trs 908-Annex9Poorvi KumarÎncă nu există evaluări
- Lec 1Document5 paginiLec 1Gail AidÎncă nu există evaluări
- Crescentia Cujete Calabash-Tree: Fact Sheet ST-216Document3 paginiCrescentia Cujete Calabash-Tree: Fact Sheet ST-216Gail AidÎncă nu există evaluări
- Adverse Drug ReactionDocument4 paginiAdverse Drug Reactionapi-246003035Încă nu există evaluări
- Org ChartDocument1 paginăOrg ChartGail AidÎncă nu există evaluări
- Crescentia Cujete Calabash-Tree: Fact Sheet ST-216Document3 paginiCrescentia Cujete Calabash-Tree: Fact Sheet ST-216Gail AidÎncă nu există evaluări
- PharmacodynamicsDocument62 paginiPharmacodynamicsGail AidÎncă nu există evaluări
- 120 141 Anatomy and PhysiologyDocument3 pagini120 141 Anatomy and PhysiologyTin BabistaÎncă nu există evaluări
- Miracle FruitDocument9 paginiMiracle FruitGail AidÎncă nu există evaluări
- Crescentia CujeteDocument40 paginiCrescentia CujeteGail AidÎncă nu există evaluări
- Miracle FruitDocument11 paginiMiracle FruitGail AidÎncă nu există evaluări
- Status of Macrobenthic Community and Its Relationships To Trace Metals and NaturalDocument8 paginiStatus of Macrobenthic Community and Its Relationships To Trace Metals and NaturalGail AidÎncă nu există evaluări
- Radio PharmaceuticalsDocument31 paginiRadio PharmaceuticalsGail AidÎncă nu există evaluări
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5794)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (895)
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (588)
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (74)
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (344)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1090)
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (121)
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)
- Lecture 6Document23 paginiLecture 6boledi angelaÎncă nu există evaluări
- 3 Asam BasaDocument162 pagini3 Asam BasaainulavidaÎncă nu există evaluări
- CHM 361 Lab Report Exp 2Document5 paginiCHM 361 Lab Report Exp 2Warina 01Încă nu există evaluări
- Relative Masses of Atoms and MoleculesDocument23 paginiRelative Masses of Atoms and MoleculesKris DookharanÎncă nu există evaluări
- Phenol Preparation and Chemical ReactionsDocument13 paginiPhenol Preparation and Chemical Reactions36 : Tushar RajguruÎncă nu există evaluări
- Articulo InteresanteDocument10 paginiArticulo InteresanteMayra Niebles BolivarÎncă nu există evaluări
- ASOE Chemistry 2022 ASDAN Questions OnlyDocument32 paginiASOE Chemistry 2022 ASDAN Questions OnlykastonoÎncă nu există evaluări
- IUPAC Nomenclature of Organic Chemistry: Basic PrinciplesDocument17 paginiIUPAC Nomenclature of Organic Chemistry: Basic PrinciplesSUBHENDU5174124Încă nu există evaluări
- Lecture Notes 13 - Esters88Document4 paginiLecture Notes 13 - Esters88Surendra RamkissoonÎncă nu există evaluări
- Ionic Equilibrium (Chem)Document82 paginiIonic Equilibrium (Chem)Draw with Hassan JamalÎncă nu există evaluări
- Binder 2 EdexcelDocument9 paginiBinder 2 EdexcelahmedÎncă nu există evaluări
- Sodium Cyanide Safety TESTDocument3 paginiSodium Cyanide Safety TESTzyy877rzzgÎncă nu există evaluări
- Chp-4, VSEPR Powerpoint (Autosaved)Document37 paginiChp-4, VSEPR Powerpoint (Autosaved)Ju KaÎncă nu există evaluări
- General Chemistry: Chapter 18: Additional Aspects of Acid-Base EquilibriaDocument20 paginiGeneral Chemistry: Chapter 18: Additional Aspects of Acid-Base EquilibrialotusputihÎncă nu există evaluări
- Predict The Product For The Following Reaction.: I II IIIDocument11 paginiPredict The Product For The Following Reaction.: I II IIIzelalem getachewÎncă nu există evaluări
- Ammonium Sulfuricum: COEI-1-AMMSUL: 2000Document3 paginiAmmonium Sulfuricum: COEI-1-AMMSUL: 2000AnaÎncă nu există evaluări
- Jahn - Teller DistortionDocument24 paginiJahn - Teller DistortionPrabhakara MusturÎncă nu există evaluări
- Organic Chemistry - WikipediaDocument11 paginiOrganic Chemistry - Wikipediatsvmpm1765Încă nu există evaluări
- PH and Buffers 7 NewDocument2 paginiPH and Buffers 7 NewVincent ManganaanÎncă nu există evaluări
- Class X - Micro Plan 2019-2020Document5 paginiClass X - Micro Plan 2019-2020sooraj nikamÎncă nu există evaluări
- Aieee 2012 Chem Sit yDocument4 paginiAieee 2012 Chem Sit yVaibhav SinghÎncă nu există evaluări
- IAL Chemistry SB2 Answers Topic17Document6 paginiIAL Chemistry SB2 Answers Topic17salmaÎncă nu există evaluări
- P Block (Mega)Document38 paginiP Block (Mega)Anant JainÎncă nu există evaluări
- Ester Hydro LDocument4 paginiEster Hydro LShivamGuptaÎncă nu există evaluări
- CH1001 2010 (Language2) NotesDocument18 paginiCH1001 2010 (Language2) Notesbav92Încă nu există evaluări
- PH Worksheet SolutionsDocument3 paginiPH Worksheet Solutionsxdiep10Încă nu există evaluări
- Problem Set 2 KEY PDFDocument5 paginiProblem Set 2 KEY PDFTrường TùngÎncă nu există evaluări
- Umpolung: Carbonyl Synthons: William D. ShipeDocument32 paginiUmpolung: Carbonyl Synthons: William D. ShipeSathiyamoorthy SithurajÎncă nu există evaluări
- Waste ProcessDocument86 paginiWaste ProcessacidoanimalÎncă nu există evaluări
- Final-Laboratory-Reportexperiment-no.1-inting FinalDocument5 paginiFinal-Laboratory-Reportexperiment-no.1-inting FinalChanie Baguio Pitogo100% (1)