Documente Academic
Documente Profesional
Documente Cultură
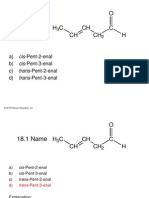
Chemistry DPP 2
Încărcat de
ashaTitlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Chemistry DPP 2
Încărcat de
ashaDrepturi de autor:
Formate disponibile
CHEMISTRY
Daily Practice Problems
Target -JEE /PMT
CLASS : XI MARKS : 35 TIME : 45 MIN. DPP. NO.-2
Concentrate on your Target and win your Target
Only One correct :
t
Q.1 The number of molecules of water in 333 g of Al2(SO4)3. 18H2O is [3]
oin
(A) 18.0 6.02 1023 (B) 9.0 6.02 1023
(C) 18.0 (D) 36.0
Q.2
(A) 4 gm oxygen
(C) 35.5 gm chlorine
yP
Which of the following have largest number of atoms?
(B) 16 gm sulphur
(D) 14 gm lithium
[3]
tud
Q.3 Weight of oxygen molecules which is equal to weight of 20 molecules of SO3. [3]
(A) 100 (B) 50 (C) 15 (D) 8
Q.4 The weight of 3.2 105 atoms of an element is 8.0 1018 gm. The atomic weight of the element should
S
be about [3]
(A) 2.5 1022 (B) 15 (C) 1.5 (D) 150
me
Q.5 The number of neutrons present in 9 mg of O18 is [3]
(A) 10 (B) 5NA (C) 0.005 NA (D) 0.0005 NA
More than One may correct :
Ac
Q.6 Which of the following contain same number of total atoms. [5]
(A) 9 gm C2H6 (B) 18.0 gm C6H12O6
(C) 9.8 gm H2SO4 (D) 48 gm SO3
Q.7 Select the incorrect statement(s) : [5]
(A) Mass of 6.022 1025 molecule of SO3 is 8 kg.
(B) Number of oxygen atoms in 4.8 gm of O3 are 18.06 1023
(C) Mass of 22.7 ml of C2H6 at 1 bar pressure & 273 K is 30 gm
(D) Volume of 51 mg of NH3 at 1 bar pressure & 273 K is 68.1 l .
Assertion and Reasoning type question:
Question No. 8 to 9 ( 2 questions)
Questions below consist of an Assertion in column I and Reason in column II. Use the following
key to choose the appropriate answer
(A) If both Assertion and Reason are correct and Reason is the correct explanation of the
Assertion.
(B) If Assertion and Reason are correct, but Reason is not the correct explanation of the Assertion.
(C) If Assertion is correct, but Reason is incorrect.
t
(D) If Assertion is incorrect, but Reason is correct.
oin
Q.8 Assertion : Equal mass of H2 and O2 contain equal number of atoms of H & O respectively.
Reason : Equal moles of H2 and O2 contain equal number of atoms of H & O respectively.
[3]
Q.9 Assertion :
Reason :
Graphite is an element.
yP
An element contains only one type of atoms.
[3]
tud
Match the column:
Q.10 Column I Column II [4]
S
(No. of moles of )
(A) Oxygen atom in 0.5 mol Ca3(PO4)2 (P) 8
me
(B) Carbon atom in 112 lit. CH4 at 1 atm & 273 K. (Q) 4
(C) Sulphur in 192 gm of SO2 (R) 5
Ac
(D) Cl in 24.088 1023 molecule Cl2O (S) 3
S-ar putea să vă placă și
- Bridge Couse-Basic Concept-4Document2 paginiBridge Couse-Basic Concept-4dhruvch2021Încă nu există evaluări
- Mole Concept DPP-1 PDFDocument1 paginăMole Concept DPP-1 PDFNo Rest MassÎncă nu există evaluări
- Mole CPP Combine PDFDocument13 paginiMole CPP Combine PDFPiyushÎncă nu există evaluări
- Mole Concept DPP-2 PDFDocument1 paginăMole Concept DPP-2 PDFNo Rest MassÎncă nu există evaluări
- Bridge Course Assig - Mole ConceptsDocument2 paginiBridge Course Assig - Mole ConceptsSubs BadaoÎncă nu există evaluări
- Chemistry DPP 3Document2 paginiChemistry DPP 3ashaÎncă nu există evaluări
- Total Success Mole Concept and RedoxDocument8 paginiTotal Success Mole Concept and RedoxVengefulÎncă nu există evaluări
- Mole ConceptDocument12 paginiMole ConceptJahan MarfatiaÎncă nu există evaluări
- Part - I: Subjective Questions: Introduction To ChemistryDocument7 paginiPart - I: Subjective Questions: Introduction To ChemistryMohini DeviÎncă nu există evaluări
- Exercise With Answer Key EnglishDocument7 paginiExercise With Answer Key Englishaliasiya929Încă nu există evaluări
- Mole Concept DPP-1 - 501352Document2 paginiMole Concept DPP-1 - 501352Vatsal BhargavaÎncă nu există evaluări
- DDT-1 To 4 English ITCDocument5 paginiDDT-1 To 4 English ITCSujal DwivediÎncă nu există evaluări
- DDT-1 To 4 English ITCDocument5 paginiDDT-1 To 4 English ITCv20687606Încă nu există evaluări
- Class IX - MOLE CONCEPT ONLINEDocument5 paginiClass IX - MOLE CONCEPT ONLINEavijayprasad2207Încă nu există evaluări
- Assignment No. 3: AnswerDocument2 paginiAssignment No. 3: Answertech.anand2211Încă nu există evaluări
- 01 Unit# 1Document3 pagini01 Unit# 1Muhammad Bilal ChemIstÎncă nu există evaluări
- Mole Concept DPP-4 - 501352 PDFDocument1 paginăMole Concept DPP-4 - 501352 PDFJEENEET CHEMISTRYÎncă nu există evaluări
- DPP - 01-11 - PH. CHEM - Abhimanyu (SC)Document23 paginiDPP - 01-11 - PH. CHEM - Abhimanyu (SC)Rohan VayaÎncă nu există evaluări
- Chemistry Assignment: (Single Correct Choice Type) Q.1Document3 paginiChemistry Assignment: (Single Correct Choice Type) Q.1Samridh GuptaÎncă nu există evaluări
- Iconic Chemistry Classes Mole Concept: Iitian Sushil KumarDocument2 paginiIconic Chemistry Classes Mole Concept: Iitian Sushil KumarsushanjÎncă nu există evaluări
- JA DPP No.A1 To A11 P PC bJx3HDYDocument17 paginiJA DPP No.A1 To A11 P PC bJx3HDYAkkaldevi Saivinayak CRÎncă nu există evaluări
- Mole Concept Exercise-1Document10 paginiMole Concept Exercise-1Game TechÎncă nu există evaluări
- Chemistry 14Document3 paginiChemistry 14BALA GANESHÎncă nu există evaluări
- Many Gram in This: (Cuso Containg AreDocument2 paginiMany Gram in This: (Cuso Containg AreShubham SinghÎncă nu există evaluări
- Final 01 A Some Basic Concept of Chemistry PDFDocument22 paginiFinal 01 A Some Basic Concept of Chemistry PDFSwastik TripathiÎncă nu există evaluări
- Stepchem 11 PDFDocument13 paginiStepchem 11 PDFSyeda Fatima ZahraÎncă nu există evaluări
- Some Basic Concepts of Chemistry - Advanced Questions PDFDocument14 paginiSome Basic Concepts of Chemistry - Advanced Questions PDFAditya AggarwalÎncă nu există evaluări
- Mole Concept DPP-2 - 501352Document1 paginăMole Concept DPP-2 - 501352Vatsal BhargavaÎncă nu există evaluări
- 9 Science Exemplar Chapter 3Document7 pagini9 Science Exemplar Chapter 3Ashish GuptaÎncă nu există evaluări
- DPP 01Document2 paginiDPP 01urmomÎncă nu există evaluări
- Sicmyb - DPP Mole ConceptDocument6 paginiSicmyb - DPP Mole ConceptBorn to fightÎncă nu există evaluări
- Chap# 01, 01Document2 paginiChap# 01, 01Faisal IzharÎncă nu există evaluări
- Mole Concept DPP 1 To 10-20240305104637328629Document18 paginiMole Concept DPP 1 To 10-20240305104637328629singhishant0011Încă nu există evaluări
- Mole Concept Worksheet 2 PDFDocument7 paginiMole Concept Worksheet 2 PDFKripa DeviÎncă nu există evaluări
- Moleconcept (E L)Document3 paginiMoleconcept (E L)ramanji1021Încă nu există evaluări
- Moleconcept (E-L)Document3 paginiMoleconcept (E-L)ramanji1021Încă nu există evaluări
- MHT Cet 20221662555106Document7 paginiMHT Cet 20221662555106Some random nameÎncă nu există evaluări
- Exercise 2 QuestionsDocument5 paginiExercise 2 Questionsyuen lok hinÎncă nu există evaluări
- Chapter Wise 11th ChemistryDocument44 paginiChapter Wise 11th Chemistryirfanranjha733Încă nu există evaluări
- Basic Concepts of Chemistry A0x49QVl3BT5Ko2KDocument20 paginiBasic Concepts of Chemistry A0x49QVl3BT5Ko2Krineeth22745Încă nu există evaluări
- Unit 8 AP Chem - Organic and ComplexesDocument20 paginiUnit 8 AP Chem - Organic and ComplexesMinnie InarapmasÎncă nu există evaluări
- 332a080528defc8b129e46e17d91b283b42b97b9963238b01c17e6310a3f4682Document61 pagini332a080528defc8b129e46e17d91b283b42b97b9963238b01c17e6310a3f4682bharadwajnavneet599Încă nu există evaluări
- Test Paper - 1 (Arihant)Document7 paginiTest Paper - 1 (Arihant)kumarsanu2905Încă nu există evaluări
- Solved Examples: Acc-Ch-Mole ConceptDocument11 paginiSolved Examples: Acc-Ch-Mole ConceptTushar SinghÎncă nu există evaluări
- NCERT Exemplar Solution Class 9 Chapter 3Document20 paginiNCERT Exemplar Solution Class 9 Chapter 3HhggfvÎncă nu există evaluări
- Multiple Choice QuestionsDocument7 paginiMultiple Choice QuestionsArya AnupamÎncă nu există evaluări
- Class 9 Science 3Document7 paginiClass 9 Science 3chandralok_kumarÎncă nu există evaluări
- Mole Concept Full Chapter Practice Sheet - 220627 - 1 - 220627 - 155952Document7 paginiMole Concept Full Chapter Practice Sheet - 220627 - 1 - 220627 - 1559522005anushkasharmaÎncă nu există evaluări
- QB-DPPDocument54 paginiQB-DPPd anjilappa100% (1)
- Mole Concept & Redox Reaction Class - 11 Jee PackageDocument52 paginiMole Concept & Redox Reaction Class - 11 Jee Packagevergadiaarnav2106Încă nu există evaluări
- QP - Chem-07-FEB 2024Document5 paginiQP - Chem-07-FEB 2024Tanuj MohiteÎncă nu există evaluări
- 11th Some Basic MCQDocument40 pagini11th Some Basic MCQsheik abdullahÎncă nu există evaluări
- JEE Main Online Exam 2019: Questions & Solutions (Memory Based)Document5 paginiJEE Main Online Exam 2019: Questions & Solutions (Memory Based)Ihtisham Ul HaqÎncă nu există evaluări
- 2018 Chemistry Standardised Test For Science Stream (SPM)Document7 pagini2018 Chemistry Standardised Test For Science Stream (SPM)carnationÎncă nu există evaluări
- DPP (1 TO) Physical ChemistryDocument47 paginiDPP (1 TO) Physical ChemistryRaju SinghÎncă nu există evaluări
- Basic Physical Chemistry 3Document6 paginiBasic Physical Chemistry 3Rupam HaloiÎncă nu există evaluări
- DPP - 12-21 - PH. CHEM - Abhimanyu - (Sol.)Document21 paginiDPP - 12-21 - PH. CHEM - Abhimanyu - (Sol.)GEETA JUNAWAÎncă nu există evaluări
- Mole Concept 11 PDFDocument26 paginiMole Concept 11 PDFSamyak Jha100% (1)
- 159 PDF PDFDocument7 pagini159 PDF PDFashaÎncă nu există evaluări
- Special RelativityDocument17 paginiSpecial RelativityDiego M GranziolÎncă nu există evaluări
- High Resistance by LeakageDocument1 paginăHigh Resistance by LeakageRavi Kanth M NÎncă nu există evaluări
- 34556-11820-JEST Sample Question PhysicsDocument1 pagină34556-11820-JEST Sample Question PhysicsTrisha BanerjeeÎncă nu există evaluări
- III-B.Sc Practical: I Set-Experiments ListDocument1 paginăIII-B.Sc Practical: I Set-Experiments ListashaÎncă nu există evaluări
- Nanoparticles: Preparation, Characterization, Applications: Wet-Chemical MethodsDocument2 paginiNanoparticles: Preparation, Characterization, Applications: Wet-Chemical MethodsashaÎncă nu există evaluări
- White GrapheneDocument4 paginiWhite GrapheneashaÎncă nu există evaluări
- Self Inductance of A Given Coil by Anderson's BridgeDocument2 paginiSelf Inductance of A Given Coil by Anderson's BridgeashaÎncă nu există evaluări
- Parallel Resonance: Frequency in HZ Voltage in VDocument2 paginiParallel Resonance: Frequency in HZ Voltage in VashaÎncă nu există evaluări
- Helmoltz GalvanometerDocument3 paginiHelmoltz GalvanometerashaÎncă nu există evaluări
- Magnetic Field On The Axis of A CoilDocument1 paginăMagnetic Field On The Axis of A CoilashaÎncă nu există evaluări
- Sushmitha Iind BSCDocument1 paginăSushmitha Iind BSCashaÎncă nu există evaluări
- Grating Normal DeviationDocument2 paginiGrating Normal DeviationashaÎncă nu există evaluări
- Self Inductance of A Given Coil by Anderson's BridgeDocument2 paginiSelf Inductance of A Given Coil by Anderson's BridgeashaÎncă nu există evaluări
- 3.e by M by Thomson MethodDocument4 pagini3.e by M by Thomson MethodashaÎncă nu există evaluări
- Physicsmanual 5Document2 paginiPhysicsmanual 5ashaÎncă nu există evaluări
- Low Pass and High PassDocument1 paginăLow Pass and High PassashaÎncă nu există evaluări
- Mode ConstantsDocument25 paginiMode ConstantsashaÎncă nu există evaluări
- Unit Wise Analysis of Previous Question Papers of Csir-Ugc (Net/Jef) Exam (New Pattern)Document1 paginăUnit Wise Analysis of Previous Question Papers of Csir-Ugc (Net/Jef) Exam (New Pattern)ashaÎncă nu există evaluări
- Sushmitha Iind BSCDocument1 paginăSushmitha Iind BSCashaÎncă nu există evaluări
- High Resistance by LeakageDocument1 paginăHigh Resistance by LeakageRavi Kanth M NÎncă nu există evaluări
- State Eligibility Test: Physical Sciences Previous Question PapersDocument1 paginăState Eligibility Test: Physical Sciences Previous Question PapersashaÎncă nu există evaluări
- Torsional PendulumDocument1 paginăTorsional PendulumashaÎncă nu există evaluări
- Chapterwise Analysis PDFDocument1 paginăChapterwise Analysis PDFashaÎncă nu există evaluări
- IIT-JAM Chemistry Test Series Answer KeyDocument4 paginiIIT-JAM Chemistry Test Series Answer KeyashaÎncă nu există evaluări
- Solid State Physics Full - 3Document7 paginiSolid State Physics Full - 3ashaÎncă nu există evaluări
- Unit Wise Analysis of Previous Question Papers of Csir-Ugc (Net/Jef) Exam (New Pattern)Document1 paginăUnit Wise Analysis of Previous Question Papers of Csir-Ugc (Net/Jef) Exam (New Pattern)ashaÎncă nu există evaluări
- 0 05 Syllabus For NET With Tips FinalDocument14 pagini0 05 Syllabus For NET With Tips FinalashaÎncă nu există evaluări
- Cover Page - IIT JAM Chemistry - MPDocument2 paginiCover Page - IIT JAM Chemistry - MPashaÎncă nu există evaluări
- Solid State Physics Full - 250Document5 paginiSolid State Physics Full - 250ashaÎncă nu există evaluări
- StrippersDocument6 paginiStrippersDiliniÎncă nu există evaluări
- Chemistry SQP 1Document8 paginiChemistry SQP 1Purnima PandaÎncă nu există evaluări
- Compressed Gas Association CGA V-1 Cylinder Valve ConnectionsDocument4 paginiCompressed Gas Association CGA V-1 Cylinder Valve ConnectionsmauroÎncă nu există evaluări
- Cisplatin in Anticancer DrugsDocument22 paginiCisplatin in Anticancer DrugswatiÎncă nu există evaluări
- Safety - What Is %LEL - %UEL and PID - Lower Explosive Limit (LEL) and The Upper Explosive Limit (UEL) - PhotoIonization DetectorDocument4 paginiSafety - What Is %LEL - %UEL and PID - Lower Explosive Limit (LEL) and The Upper Explosive Limit (UEL) - PhotoIonization DetectorlavkeshÎncă nu există evaluări
- Crude Oil GradesDocument5 paginiCrude Oil GradesibrahimkhansahilÎncă nu există evaluări
- Advanced Organometallic Chemistry and Its Applications in Industry and Catalytic Reactions PDFDocument144 paginiAdvanced Organometallic Chemistry and Its Applications in Industry and Catalytic Reactions PDFdustinthewindÎncă nu există evaluări
- Rhesperse - RM100 - Rheological ModifierDocument3 paginiRhesperse - RM100 - Rheological ModifierteddydeÎncă nu există evaluări
- Ciulu, 2018 ReviewDocument20 paginiCiulu, 2018 ReviewLia PuspitasariÎncă nu există evaluări
- FA-derivatyzacja AChromDocument13 paginiFA-derivatyzacja AChromReza AzghadiÎncă nu există evaluări
- Used Motor Oil Treatment: Turning Waste Oil Into Valuable ProductsDocument12 paginiUsed Motor Oil Treatment: Turning Waste Oil Into Valuable ProductsBoulos NassarÎncă nu există evaluări
- Chem 243 Midterm ReviewDocument152 paginiChem 243 Midterm ReviewProfAnonymous100% (1)
- MCQs Dr. Laila - Lipogenesis - Lipolysis - FA OxidationDocument26 paginiMCQs Dr. Laila - Lipogenesis - Lipolysis - FA OxidationYoussef Mohamed100% (2)
- Capstone Chapter 1 3Document15 paginiCapstone Chapter 1 3De-Andrie GotuatoÎncă nu există evaluări
- Excelsize 22 (Visco VS Solid%)Document2 paginiExcelsize 22 (Visco VS Solid%)Ardita Rizki FauziÎncă nu există evaluări
- 1.3. Other Means of Generating Enolates: Osime Meli O Li + SimeDocument14 pagini1.3. Other Means of Generating Enolates: Osime Meli O Li + SimeVirendra Singh RajputÎncă nu există evaluări
- Chemistry Topics MypDocument6 paginiChemistry Topics MypMaxwell WamaithaÎncă nu există evaluări
- What Is Hydrocarbons?Document6 paginiWhat Is Hydrocarbons?Kathelyn Ruiz-SumandoÎncă nu există evaluări
- IAL Chemistry Unit-02 Samplepaper - 03 PDFDocument17 paginiIAL Chemistry Unit-02 Samplepaper - 03 PDFDimuthu SandaruwanÎncă nu există evaluări
- Viscosity, Relaxation and Stability of Natural RubberDocument4 paginiViscosity, Relaxation and Stability of Natural RubberHuỳnh TGÎncă nu există evaluări
- PRODUCTIONOF60000MTPAOFOLEOCHEMICALMETHYLESTERFROMRBDPALMKERNELOILDocument586 paginiPRODUCTIONOF60000MTPAOFOLEOCHEMICALMETHYLESTERFROMRBDPALMKERNELOILKevin Fernando PratamaÎncă nu există evaluări
- Planilha Sem TítuloDocument56 paginiPlanilha Sem TítuloAnonymous T.I.Încă nu există evaluări
- Pesticides Formulations As On 01.06.2023Document25 paginiPesticides Formulations As On 01.06.2023Atish WagheleÎncă nu există evaluări
- Carbonyl ChemsketchDocument3 paginiCarbonyl ChemsketchJenica RiaÎncă nu există evaluări
- 50 Olefin ReductionDocument2 pagini50 Olefin ReductiondimkuhÎncă nu există evaluări
- Biochemistry Book 2Document119 paginiBiochemistry Book 2Jugnu JugnuÎncă nu există evaluări
- Gen & Genom TumbuhanDocument33 paginiGen & Genom Tumbuhanmonocotil9879Încă nu există evaluări
- Benzofused HeterocyclesDocument6 paginiBenzofused HeterocyclesMohini Bajaj50% (2)
- Kqd7009 - Tutorial 8Document3 paginiKqd7009 - Tutorial 8taned8787Încă nu există evaluări
- Cs Se 0006 HumastarDocument2 paginiCs Se 0006 Humastarluisoft88Încă nu există evaluări
- AP World History: Modern Premium, 2024: Comprehensive Review with 5 Practice Tests + an Online Timed Test OptionDe la EverandAP World History: Modern Premium, 2024: Comprehensive Review with 5 Practice Tests + an Online Timed Test OptionEvaluare: 5 din 5 stele5/5 (1)
- AP Microeconomics/Macroeconomics Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP Microeconomics/Macroeconomics Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- Medical English Dialogues: Clear & Simple Medical English Vocabulary for ESL/EFL LearnersDe la EverandMedical English Dialogues: Clear & Simple Medical English Vocabulary for ESL/EFL LearnersÎncă nu există evaluări
- AP Physics 1 Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP Physics 1 Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- AP English Language and Composition Premium, 2024: 8 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP English Language and Composition Premium, 2024: 8 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- GMAT Prep 2024/2025 For Dummies with Online Practice (GMAT Focus Edition)De la EverandGMAT Prep 2024/2025 For Dummies with Online Practice (GMAT Focus Edition)Încă nu există evaluări
- AP Biology Premium, 2024: Comprehensive Review With 5 Practice Tests + an Online Timed Test OptionDe la EverandAP Biology Premium, 2024: Comprehensive Review With 5 Practice Tests + an Online Timed Test OptionÎncă nu există evaluări
- GMAT Foundations of Verbal: Practice Problems in Book and OnlineDe la EverandGMAT Foundations of Verbal: Practice Problems in Book and OnlineÎncă nu există evaluări
- Digital SAT Reading and Writing Practice Questions: Test Prep SeriesDe la EverandDigital SAT Reading and Writing Practice Questions: Test Prep SeriesEvaluare: 5 din 5 stele5/5 (2)
- College Level Anatomy and Physiology: Essential Knowledge for Healthcare Students, Professionals, and Caregivers Preparing for Nursing Exams, Board Certifications, and BeyondDe la EverandCollege Level Anatomy and Physiology: Essential Knowledge for Healthcare Students, Professionals, and Caregivers Preparing for Nursing Exams, Board Certifications, and BeyondÎncă nu există evaluări
- AP Physics 2 Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP Physics 2 Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- How to Be a High School Superstar: A Revolutionary Plan to Get into College by Standing Out (Without Burning Out)De la EverandHow to Be a High School Superstar: A Revolutionary Plan to Get into College by Standing Out (Without Burning Out)Evaluare: 4.5 din 5 stele4.5/5 (11)
- GMAT Foundations of Math: Start Your GMAT Prep with Online Starter Kit and 900+ Practice ProblemsDe la EverandGMAT Foundations of Math: Start Your GMAT Prep with Online Starter Kit and 900+ Practice ProblemsEvaluare: 4 din 5 stele4/5 (7)
- Digital SAT Prep 2024 For Dummies: Book + 4 Practice Tests Online, Updated for the NEW Digital FormatDe la EverandDigital SAT Prep 2024 For Dummies: Book + 4 Practice Tests Online, Updated for the NEW Digital FormatÎncă nu există evaluări
- 55 Successful Harvard Law School Application Essays, 2nd Edition: With Analysis by the Staff of The Harvard CrimsonDe la Everand55 Successful Harvard Law School Application Essays, 2nd Edition: With Analysis by the Staff of The Harvard CrimsonEvaluare: 3 din 5 stele3/5 (1)
- AP Human Geography Premium, 2024: 6 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP Human Geography Premium, 2024: 6 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- SAT Prep Plus: Unlocked Edition 2022 - 5 Full Length Practice Tests - Behind-the-scenes game-changing answer explanations to each question - Top level strategies, tips and tricks for each sectionDe la EverandSAT Prep Plus: Unlocked Edition 2022 - 5 Full Length Practice Tests - Behind-the-scenes game-changing answer explanations to each question - Top level strategies, tips and tricks for each sectionÎncă nu există evaluări
- IELTS Academic Vocabulary Builder: Improve Your Band Score on the IELTS Academic ExamDe la EverandIELTS Academic Vocabulary Builder: Improve Your Band Score on the IELTS Academic ExamÎncă nu există evaluări
- AP U.S. History Premium, 2024: Comprehensive Review With 5 Practice Tests + an Online Timed Test OptionDe la EverandAP U.S. History Premium, 2024: Comprehensive Review With 5 Practice Tests + an Online Timed Test OptionÎncă nu există evaluări
- Digital PSAT/NMSQT Study Guide Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeDe la EverandDigital PSAT/NMSQT Study Guide Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- Indian Polity with Indian Constitution & Parliamentary AffairsDe la EverandIndian Polity with Indian Constitution & Parliamentary AffairsÎncă nu există evaluări
- The Premed Playbook: Guide to the Medical School Personal StatementDe la EverandThe Premed Playbook: Guide to the Medical School Personal StatementEvaluare: 5 din 5 stele5/5 (1)
- English Grammar Exercises With Answers Part 5: Your Quest Towards C2De la EverandEnglish Grammar Exercises With Answers Part 5: Your Quest Towards C2Încă nu există evaluări
- Digital SAT Preview: What to Expect + Tips and StrategiesDe la EverandDigital SAT Preview: What to Expect + Tips and StrategiesEvaluare: 5 din 5 stele5/5 (3)
- AP Calculus Premium, 2024: 12 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP Calculus Premium, 2024: 12 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- AP Environmental Science Premium, 2024: 5 Practice Tests + Comprehensive Review + Online PracticeDe la EverandAP Environmental Science Premium, 2024: 5 Practice Tests + Comprehensive Review + Online PracticeÎncă nu există evaluări
- Finish What You Start: The Art of Following Through, Taking Action, Executing, & Self-DisciplineDe la EverandFinish What You Start: The Art of Following Through, Taking Action, Executing, & Self-DisciplineEvaluare: 4.5 din 5 stele4.5/5 (94)