Documente Academic
Documente Profesional
Documente Cultură
No. Procedure Experiment Observation Result Reaction Conclusion
Încărcat de
diyah0 evaluări0% au considerat acest document util (0 voturi)
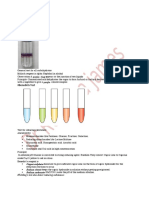
14 vizualizări2 paginiExperiment 6 observed that adding acetone or isopropyl alcohol to sodium hydroxide and iodine produced a yellow precipitate with an odor like betadine. This indicates that acetone and isopropyl alcohol can undergo the haloform reaction to produce haloforms.
Experiment 7 observed that mixing acetaldehyde and sodium hydroxide produced a yellow solution with an acetaldehyde odor. Boiling the mixture produced an orange solution with a stronger crotonaldehyde odor, indicating that acetaldehyde underwent aldol condensation to form crotonaldehyde.
The experiments demonstrated the haloform reaction and aldol condensation reactions through observation of color changes and distinctive odors produced.
Descriere originală:
hasil dari pengamatan
Titlu original
Hasil pengamatan
Drepturi de autor
© © All Rights Reserved
Formate disponibile
DOCX, PDF, TXT sau citiți online pe Scribd
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentExperiment 6 observed that adding acetone or isopropyl alcohol to sodium hydroxide and iodine produced a yellow precipitate with an odor like betadine. This indicates that acetone and isopropyl alcohol can undergo the haloform reaction to produce haloforms.
Experiment 7 observed that mixing acetaldehyde and sodium hydroxide produced a yellow solution with an acetaldehyde odor. Boiling the mixture produced an orange solution with a stronger crotonaldehyde odor, indicating that acetaldehyde underwent aldol condensation to form crotonaldehyde.
The experiments demonstrated the haloform reaction and aldol condensation reactions through observation of color changes and distinctive odors produced.
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca DOCX, PDF, TXT sau citiți online pe Scribd
0 evaluări0% au considerat acest document util (0 voturi)
14 vizualizări2 paginiNo. Procedure Experiment Observation Result Reaction Conclusion
Încărcat de
diyahExperiment 6 observed that adding acetone or isopropyl alcohol to sodium hydroxide and iodine produced a yellow precipitate with an odor like betadine. This indicates that acetone and isopropyl alcohol can undergo the haloform reaction to produce haloforms.
Experiment 7 observed that mixing acetaldehyde and sodium hydroxide produced a yellow solution with an acetaldehyde odor. Boiling the mixture produced an orange solution with a stronger crotonaldehyde odor, indicating that acetaldehyde underwent aldol condensation to form crotonaldehyde.
The experiments demonstrated the haloform reaction and aldol condensation reactions through observation of color changes and distinctive odors produced.
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca DOCX, PDF, TXT sau citiți online pe Scribd
Sunteți pe pagina 1din 2
No.
Procedure experiment Observation result Reaction Conclusion
1.
6. Haloform Reaction Before Acetone and isopropyl
- Sodium hydroxide : alcohol can produce a
3 mL of 5% Sodium colorless haloform and has an odor
Hydroxide solution - Iodium : brownish red like betadine (medicine)
- Acetone : colorless
- Pouring into test tube 1 and test - Isopropyl alcohol :
tube 2 colorless
- Adding 5 drops of acetone into
test tube 1 and test tube 2 After
- Adding 10 mL iodium solution - Sodium hydroxide +
into test tube 1 and test tube 2 acetone : colorless
- Shaking until the colour of - Sodium hydroxide +
iodium isnt lost acetone + iodium :
- Observing yellow precipitate, has
- Smelling it an odor like betadine
(medicine)
Yellow sediment & - Sodium hydroxide +
has an odor isopropyl alcohol :
colorless
- Sodium hydroxide +
isopropyl alcohol +
iodium : yellow
precipitate, has an odor
like betadine (medicine)
7. Condensation of Aldol Before An aldehyde (acetaldehyde)
- NaOH : Colorless can produce an aldol with
- Acetaldehyde : yellow crotonaldehyde odor
After
4 mL of 1% Sodium Hydroxide - NaOH + acetaldehyde :
solution yellow , has an odor
- NaOH + acetaldehyde +
- Pouring into test tube boling : orange , has a
- Adding 0,5 mL acetaldehyde stronger odor
solution (crotonaldehyde)
- Shaking
- Smelling it (acetaldehyde)
- Boiling until 3 minutes
- Observing
- Smelling it again
Has an odor
(smell crotonaldehyde)
S-ar putea să vă placă și
- Identification of Alcohols: Methanol Ethanol Glycerol (Glycerin)Document10 paginiIdentification of Alcohols: Methanol Ethanol Glycerol (Glycerin)lox agencyÎncă nu există evaluări
- Iodimetry and IodometryDocument6 paginiIodimetry and IodometrythereseÎncă nu există evaluări
- Identifying Salts LabDocument14 paginiIdentifying Salts LabSantiago Macías100% (1)
- Activity No 9 - AcetoneDocument3 paginiActivity No 9 - Acetonepharmaebooks100% (5)
- 1 - X Acids & BasesDocument8 pagini1 - X Acids & BasesYogy YÎncă nu există evaluări
- Experiment 7 Report SheetDocument21 paginiExperiment 7 Report SheetDiane Princess SultanÎncă nu există evaluări
- AlcoholsDocument32 paginiAlcoholsheshammohamed44148Încă nu există evaluări
- Systematic Analysis of Organic CompoundsDocument12 paginiSystematic Analysis of Organic CompoundsM Bharath ReddyÎncă nu există evaluări
- Title: 1) Preliminary TestsDocument5 paginiTitle: 1) Preliminary TestsAkash PawarÎncă nu există evaluări
- Exp 4 Postlab Worksheet Identification of An Unknown Through Qualitative AnalysisDocument4 paginiExp 4 Postlab Worksheet Identification of An Unknown Through Qualitative AnalysisKarl CarandangÎncă nu există evaluări
- Experiment Result Nu. Procedure Observationt Result Reaction Conclusion BeforeDocument6 paginiExperiment Result Nu. Procedure Observationt Result Reaction Conclusion BeforeAnggraini Nugroho PÎncă nu există evaluări
- Qualitative Tests For CarbohydratesDocument22 paginiQualitative Tests For CarbohydratesHelal HamadÎncă nu există evaluări
- Physical Examination A. Physical State Liquid B. Color Colorless C. Odor Strong and Pungent Odor D. Ignition Test FlammableDocument3 paginiPhysical Examination A. Physical State Liquid B. Color Colorless C. Odor Strong and Pungent Odor D. Ignition Test FlammableYen BumÎncă nu există evaluări
- Activity No. 1:: Inorganic Compounds VS Organic CompoundsDocument15 paginiActivity No. 1:: Inorganic Compounds VS Organic CompoundsChristine RanoaÎncă nu există evaluări
- Expt. No. 5 Identification of Organic CompoundsDocument3 paginiExpt. No. 5 Identification of Organic CompoundsMae DinDinÎncă nu există evaluări
- Expt. No. 5 Identification of Organic CompoundsDocument3 paginiExpt. No. 5 Identification of Organic CompoundsYen BumÎncă nu există evaluări
- Experiment 19Document3 paginiExperiment 19Beenu SinghÎncă nu există evaluări
- A2as Chem Revised Support 18964Document12 paginiA2as Chem Revised Support 18964Neen NaazÎncă nu există evaluări
- Chem Procedure-Inorg Salt - 2021Document9 paginiChem Procedure-Inorg Salt - 2021S3er IgÎncă nu există evaluări
- Aldehyde-Ketone TestsDocument3 paginiAldehyde-Ketone TestsNorazma ZainuddinÎncă nu există evaluări
- ReaksiDocument12 paginiReaksiLaila RoikhatulÎncă nu există evaluări
- Acidbasenotes 100505143921 Phpapp02Document15 paginiAcidbasenotes 100505143921 Phpapp02shelter musasaÎncă nu există evaluări
- Fongrsy - Acids Bases and AlkalisDocument2 paginiFongrsy - Acids Bases and AlkalisDinangaÎncă nu există evaluări
- Title: 1) Preliminary TestsDocument6 paginiTitle: 1) Preliminary TestsAkash PawarÎncă nu există evaluări
- Maha FaridDocument49 paginiMaha FaridRamY El NahasÎncă nu există evaluări
- Chemistry Qualitative Analysis NotesDocument9 paginiChemistry Qualitative Analysis NotesLim Yan Peng GaryÎncă nu există evaluări
- Yanzon Post Lab 4Document4 paginiYanzon Post Lab 4anon_862023761Încă nu există evaluări
- Acids Bases and SaltsDocument8 paginiAcids Bases and SaltsNeeraj Poddar100% (1)
- Acid and Alkali: 6.1 Properties of Acids and AlkalisDocument2 paginiAcid and Alkali: 6.1 Properties of Acids and AlkalisKalesware MuniandyÎncă nu există evaluări
- Let Me Give Some General DetailsDocument7 paginiLet Me Give Some General DetailsNimra IqbalÎncă nu există evaluări
- Acid Bases SaltDocument4 paginiAcid Bases SaltPhạm Quốc HảiÎncă nu există evaluări
- ESSENTIAL CHEMESTRIY Final 45Document14 paginiESSENTIAL CHEMESTRIY Final 45Syrus ZambiaÎncă nu există evaluări
- Experiment 2 - POST LAB DISCUSSIONDocument6 paginiExperiment 2 - POST LAB DISCUSSIONMina BiancaÎncă nu există evaluări
- Chem 503 - Activity 4Document6 paginiChem 503 - Activity 4Aries Jay ReyesÎncă nu există evaluări
- AsdasdDocument1 paginăAsdasdArthur Estevan KusnadiÎncă nu există evaluări
- Experiment 8B - Chemistry 31.1.Document82 paginiExperiment 8B - Chemistry 31.1.JoachimÎncă nu există evaluări
- Biochem 4Document9 paginiBiochem 4Clint Danniel GuarinÎncă nu există evaluări
- Organic Chemistry 2: Welcome BackDocument67 paginiOrganic Chemistry 2: Welcome BackYoussef AtefÎncă nu există evaluări
- Identification Class DyesDocument38 paginiIdentification Class DyesZikriaKMuhammadÎncă nu există evaluări
- Experiment N0.5 - Alcohols and PhenolsDocument3 paginiExperiment N0.5 - Alcohols and PhenolsVida HumadasÎncă nu există evaluări
- Classification of Alcohol-Oxidation TestDocument10 paginiClassification of Alcohol-Oxidation TestFatimatuzzahra' Binti Hardiyono FISÎncă nu există evaluări
- Laporan Amali Kimia 1Document13 paginiLaporan Amali Kimia 1malarÎncă nu există evaluări
- QC 1 Lec Chapter 9Document3 paginiQC 1 Lec Chapter 9Jielline Mia CañasÎncă nu există evaluări
- Qualitative Analysis HandbookDocument16 paginiQualitative Analysis HandbookPranaliÎncă nu există evaluări
- Acids and AlkalisDocument34 paginiAcids and AlkalisMoh AmedÎncă nu există evaluări
- Glycerin IpDocument1 paginăGlycerin IpHector Santiago Lopez AcostaÎncă nu există evaluări
- Aldehydes and KetoneDocument18 paginiAldehydes and KetoneAli AlisonÎncă nu există evaluări
- Vii. Result of The ExperimentDocument4 paginiVii. Result of The Experimentulfa afifahÎncă nu există evaluări
- Analysis of Salts Acid Radicals/Anions - : 1. For - CO, So, S, No, CH COODocument1 paginăAnalysis of Salts Acid Radicals/Anions - : 1. For - CO, So, S, No, CH COO123764yÎncă nu există evaluări
- Distinction TestsDocument4 paginiDistinction TestsAntriksh SharmaÎncă nu există evaluări
- Acid, Base and Salt: by Iin Indriyati Biology Teacher of SMP 1 WonosariDocument23 paginiAcid, Base and Salt: by Iin Indriyati Biology Teacher of SMP 1 Wonosariivans augerÎncă nu există evaluări
- Acid Base SaltDocument6 paginiAcid Base SaltShaheed AnwerÎncă nu există evaluări
- Carbohydrates TestsDocument3 paginiCarbohydrates TestsKings PrideÎncă nu există evaluări
- Experiment 9 Title: Aldehyde and Ketones: Characterization of An Unknown ObjectiveDocument8 paginiExperiment 9 Title: Aldehyde and Ketones: Characterization of An Unknown Objectivebabywenn100% (6)
- Chemistry Igcse Atp Important NotesDocument12 paginiChemistry Igcse Atp Important Notesrazan nazeer86% (7)
- Purifying Palladium Precipitated From Dirty SolutoinsDocument5 paginiPurifying Palladium Precipitated From Dirty SolutoinsAFLAC ............Încă nu există evaluări
- Iodine, Iodism Cornstarch Sodium Thiosulfate Lugol's Solution Expectorant, Antimicrobial Antibacteria LUGOL'S SOLUTION/Strong Iodine SolutionDocument2 paginiIodine, Iodism Cornstarch Sodium Thiosulfate Lugol's Solution Expectorant, Antimicrobial Antibacteria LUGOL'S SOLUTION/Strong Iodine SolutionJoanna Carla Marmonejo Estorninos-WalkerÎncă nu există evaluări
- Chem 211-212 AspirinDocument15 paginiChem 211-212 AspirinDyana SyazwaniÎncă nu există evaluări
- PROTEINDocument41 paginiPROTEINdiyahÎncă nu există evaluări
- Orlab TiaraDocument3 paginiOrlab TiaradiyahÎncă nu există evaluări
- 4 X 6 In.Document1 pagină4 X 6 In.diyahÎncă nu există evaluări
- Tugas 1Document2 paginiTugas 1diyahÎncă nu există evaluări
- Tugas 1Document2 paginiTugas 1diyahÎncă nu există evaluări
- Dastwr KarbohidratDocument5 paginiDastwr KarbohidratdiyahÎncă nu există evaluări
- Hydrolysis of Ethyl Acetate in Weak AcidDocument2 paginiHydrolysis of Ethyl Acetate in Weak AciddiyahÎncă nu există evaluări
- 5126 Metpen-10Document20 pagini5126 Metpen-10diyahÎncă nu există evaluări
- CalculationDocument6 paginiCalculationdiyahÎncă nu există evaluări
- G. Analysis and Explanations: O CH IDocument2 paginiG. Analysis and Explanations: O CH IdiyahÎncă nu există evaluări
- Lesson Plan Classroom Discussion Buzz Group Method "Water Hardness"Document10 paginiLesson Plan Classroom Discussion Buzz Group Method "Water Hardness"diyahÎncă nu există evaluări
- Ginger OilDocument2 paginiGinger OildiyahÎncă nu există evaluări
- Self Assessment Sheet Spiritual AttitudeDocument14 paginiSelf Assessment Sheet Spiritual AttitudediyahÎncă nu există evaluări
- Soal Koloid English FixDocument10 paginiSoal Koloid English FixdiyahÎncă nu există evaluări
- Application of Infrared Thermography Technique in Uilding Finish EvaluationDocument9 paginiApplication of Infrared Thermography Technique in Uilding Finish EvaluationNicolas De NadaiÎncă nu există evaluări
- Drilling and Blasting As A Tunnel Excavation MethodDocument26 paginiDrilling and Blasting As A Tunnel Excavation MethodMIGUEL FERNANDO AGUIRRE QUISPE100% (1)
- 2561 Rubber Based Adhesives For Automobile IndustryDocument14 pagini2561 Rubber Based Adhesives For Automobile IndustryKaushik SenguptaÎncă nu există evaluări
- Metalsferrousandnonferrous 131023154745 Phpapp01Document29 paginiMetalsferrousandnonferrous 131023154745 Phpapp01Prasad RaikarÎncă nu există evaluări
- Recommended Practices ON Static Electricity: OISD-110 OISD - 110 (Rev.1)Document35 paginiRecommended Practices ON Static Electricity: OISD-110 OISD - 110 (Rev.1)manuppm100% (1)
- UC's MotionsDocument37 paginiUC's MotionsjssherkowÎncă nu există evaluări
- Gypsum ProductsDocument53 paginiGypsum ProductsKiran KumarÎncă nu există evaluări
- Asme B16.34Document16 paginiAsme B16.34Thomas Martin100% (1)
- Head Regulator DesignDocument112 paginiHead Regulator Designpraveena61Încă nu există evaluări
- Synergism in Solvent ExtractionDocument26 paginiSynergism in Solvent ExtractionabrahanÎncă nu există evaluări
- BEIKE - Tripod Catalogue 2014Document30 paginiBEIKE - Tripod Catalogue 2014BaliÎncă nu există evaluări
- NTSE Question Bank & Solutions (Complete Syllabus) PDFDocument7 paginiNTSE Question Bank & Solutions (Complete Syllabus) PDFSaksham50% (2)
- HAZOP For Blasting ChamberDocument5 paginiHAZOP For Blasting Chambersepri manerson sinaga0% (1)
- X-MET3000TX+ PMI BrochureDocument4 paginiX-MET3000TX+ PMI BrochureYulfikaenis MachroniÎncă nu există evaluări
- Halal and Haram IngredientsDocument26 paginiHalal and Haram IngredientsMuhammad A RehmanÎncă nu există evaluări
- Activity 2 3 2 Tensile Testing With The SsaDocument9 paginiActivity 2 3 2 Tensile Testing With The Ssaapi-277473504Încă nu există evaluări
- Physico-Chemical Standardization and Phytochemical Screening of Potential Medicinal Herb: Vetiveria Zizanioides (Roots)Document6 paginiPhysico-Chemical Standardization and Phytochemical Screening of Potential Medicinal Herb: Vetiveria Zizanioides (Roots)xiuhtlaltzinÎncă nu există evaluări
- Chapter 5: Analytic Techniques: by Julia C. Drees, Matthew S. Petrie, Alan H.B. WuDocument15 paginiChapter 5: Analytic Techniques: by Julia C. Drees, Matthew S. Petrie, Alan H.B. WuTanveerÎncă nu există evaluări
- Temperature Dependence and ZTC Bias Point Evaluation of Sub 20nm Bulk Multigate DevicesDocument16 paginiTemperature Dependence and ZTC Bias Point Evaluation of Sub 20nm Bulk Multigate DevicesYgor AguiarÎncă nu există evaluări
- 22 6Document13 pagini22 6Francisco M. RamosÎncă nu există evaluări
- MME 3518-Powder Metallurgy-1-2Document80 paginiMME 3518-Powder Metallurgy-1-2Mohammed GhisheerÎncă nu există evaluări
- The Philippine Chemical Industry ProfileDocument7 paginiThe Philippine Chemical Industry ProfileEddie Resurreccion Jr.Încă nu există evaluări
- Advanced Laboratory Testing in Research and Practice: The 2nd Bishop LectureDocument30 paginiAdvanced Laboratory Testing in Research and Practice: The 2nd Bishop LectureMMM-2012Încă nu există evaluări
- Addition of Ref#2080, #2081, #2082, #2083 and #2084. D D: EB-455Wi/465i/450W/450Wi/460/460iDocument15 paginiAddition of Ref#2080, #2081, #2082, #2083 and #2084. D D: EB-455Wi/465i/450W/450Wi/460/460ifefotroncitoÎncă nu există evaluări
- 28 Amazing Benefits and Uses For Hydrogen Peroxide Wake Up WorldDocument6 pagini28 Amazing Benefits and Uses For Hydrogen Peroxide Wake Up WorldStephen AdamsÎncă nu există evaluări
- Incorporation of Waste Plastic in Asphalt Binders To Improve TheirDocument9 paginiIncorporation of Waste Plastic in Asphalt Binders To Improve TheirSandor ManesesÎncă nu există evaluări
- Astm 578Document5 paginiAstm 578ASWANTH T S100% (1)
- Bongsuwan D 2008Document7 paginiBongsuwan D 2008Liona MargaritaÎncă nu există evaluări
- Generalized Guidelines For Structural Steel Welding InspectionDocument19 paginiGeneralized Guidelines For Structural Steel Welding InspectionHariPrasath100% (1)
- BenzilDocument5 paginiBenzildeviycÎncă nu există evaluări