Documente Academic
Documente Profesional
Documente Cultură
Reaction Kinetics and Reactor Design - John B Butt
Încărcat de
Putri Rahimi100%(1)100% au considerat acest document util (1 vot)
471 vizualizări1 paginăReaction Kinetics and Reactor Design - John B Butt
Drepturi de autor
© © All Rights Reserved
Formate disponibile
PDF sau citiți online pe Scribd
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentReaction Kinetics and Reactor Design - John B Butt
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PDF sau citiți online pe Scribd
100%(1)100% au considerat acest document util (1 vot)
471 vizualizări1 paginăReaction Kinetics and Reactor Design - John B Butt
Încărcat de
Putri RahimiReaction Kinetics and Reactor Design - John B Butt
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PDF sau citiți online pe Scribd
Sunteți pe pagina 1din 1
Apparent Reaction Kinetics 49
which has the interesting property of approaching infinity as the magnitudes of r,
and ry become the same. Physically this means an explosion, so r, = ry defines an
explosion limit for the branching chain reaction. The branching chain theory has
been used to interpret explosions in the low-pressure region for reactions such as
HO, and CO-O;. This sort of explosion is different from the normal thermal
explosion in which the heat evolved in a reaction cannot be removed from the
system, leading to increasing temperatures, rates of reaction, heat evolution, noise,
and a puff of greasy blue smoke.
After you finish Chapter 3 come back here and
show me how the chain-growth polymerization
and the Fischer-Tropsch synthesis add up to
about the same in terms of product distribution
1.7 Chemical Equilibrium
The logical joining point between the study of chemical kinetics and chemical ther-
modynamics is the point, in an elementary step, where the rate of the forward
reaction is equal to the rate of the reverse reaction. This is the point of chemical
equilibrium and is implied in the relationships of equations (1-22) and (1-23). While
we cannot conduct a short course here on the thermodynamics of chemical equili-
brium, some review will be useful. The reader may also wish to refer to a favorite text
on thermodynamics for amplification of our condensed presentation.
171 Equi
ium in Single-Phase Systems
The criterion for chemical equilibrium in a single-phase, single-reaction system is
stated in terms of the minimization of the free energy of the system. In terms of the
reaction, then
Loe,
(1-138)
where v; are the stoichiometric coefficients written in the same way as for equation
(1-49), positive for products and negative for reactants, and G; are partial molar free
energies. Following Sandler (8.1. Sandler, Chemical Engineering Thermodynamics,
John Wiley, New York, 1989), let us follow the development for a typical reaction
CO; + Hy + CO+H0
occurring in a closed system (in the thermodynamic sense) at constant pressure and
some suitable temperature. If the pressure is low enough to permit the assumption of
S-ar putea să vă placă și
- Page 1 For Layout of Design (Actual) of D:/My Dokumen/Bahan Kuliah/Semester 5/Statistik/Dx7Trial/Mydesign - Dx7 11:15 Am Nov 14, 2017Document1 paginăPage 1 For Layout of Design (Actual) of D:/My Dokumen/Bahan Kuliah/Semester 5/Statistik/Dx7Trial/Mydesign - Dx7 11:15 Am Nov 14, 2017Putri RahimiÎncă nu există evaluări
- Spesifikasi Ethylene PT. Candra Asri PetrochemicalDocument1 paginăSpesifikasi Ethylene PT. Candra Asri PetrochemicalPutri RahimiÎncă nu există evaluări
- Flowsheet PDFDocument1 paginăFlowsheet PDFPutri RahimiÎncă nu există evaluări
- Anhui Conch Cement Co LTDDocument1 paginăAnhui Conch Cement Co LTDPutri RahimiÎncă nu există evaluări
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (400)
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (74)
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (121)
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)
- Chem Review 2022Document14 paginiChem Review 2022SangvenkatÎncă nu există evaluări
- Baldwins RulesDocument3 paginiBaldwins Rulesaggelisgeorge8546Încă nu există evaluări
- Lab Report Enzyme KineticsDocument5 paginiLab Report Enzyme KineticsABEGAIL JOY MAURICIO LAPITANÎncă nu există evaluări
- Curt inDocument4 paginiCurt inAnkit JainÎncă nu există evaluări
- Kin + Sur + Pol Ques + Notes PDFDocument170 paginiKin + Sur + Pol Ques + Notes PDFAakash SrivastavaÎncă nu există evaluări
- Hess's Law ProblemsDocument2 paginiHess's Law ProblemsTjrjfjdjÎncă nu există evaluări
- Chem 17 Formal Report 2Document6 paginiChem 17 Formal Report 2Patricia Frances P. FloresÎncă nu există evaluări
- CH 19Document85 paginiCH 19Dan McÎncă nu există evaluări
- Chemistry Notes: Rate of Reaction:-Is The Change in The Concentration of A Reactant or Product Per Unit TimeDocument2 paginiChemistry Notes: Rate of Reaction:-Is The Change in The Concentration of A Reactant or Product Per Unit TimeKathryn TindallÎncă nu există evaluări
- Chee4367 HW051231Document2 paginiChee4367 HW051231kimhoang_16927574Încă nu există evaluări
- Half Life and Activation Energy PPQ PDFDocument9 paginiHalf Life and Activation Energy PPQ PDFAngus AnizÎncă nu există evaluări
- Total Synthesis of (+) - Acutiphycin: Ryan M. Moslin and Timothy F. JamisonDocument10 paginiTotal Synthesis of (+) - Acutiphycin: Ryan M. Moslin and Timothy F. JamisonTạ Đình TrungÎncă nu există evaluări
- Test Bank For Organic Chemistry Structure and Function 6th Edition Vollhardt DownloadDocument18 paginiTest Bank For Organic Chemistry Structure and Function 6th Edition Vollhardt Downloadchristopherwilliamsmcrjpeynzf100% (22)
- IIT JEE Main Advanced Physical Chemistry 12th Chemical KineticsDocument44 paginiIIT JEE Main Advanced Physical Chemistry 12th Chemical KineticsT sidharth100% (3)
- Ch14 TestbankDocument53 paginiCh14 TestbankJeremy Martin100% (11)
- KNC1063 Organic Chemistry: DR Md. Rezaur RahmanDocument19 paginiKNC1063 Organic Chemistry: DR Md. Rezaur RahmanKunashiny RamashÎncă nu există evaluări
- Organic ReactionsDocument16 paginiOrganic Reactionsyie_793Încă nu există evaluări
- Kinetics 2Document43 paginiKinetics 2noelÎncă nu există evaluări
- Benzoin CondensationDocument3 paginiBenzoin CondensationsazinaÎncă nu există evaluări
- 11 Stoichiometry Calculations Based On Chemical EquationsDocument38 pagini11 Stoichiometry Calculations Based On Chemical EquationsMohamed TarekÎncă nu există evaluări
- Chemical Kinetics (Complete Unit)Document73 paginiChemical Kinetics (Complete Unit)SAFDAR HafizÎncă nu există evaluări
- CHEM 210 CH 06 SubstitutionDocument14 paginiCHEM 210 CH 06 SubstitutionAnonymous vRpzQ2BL100% (1)
- CHP 6 Class Review ChemDocument15 paginiCHP 6 Class Review ChemNatalie RussellÎncă nu există evaluări
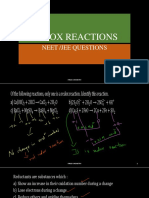
- Redox Reactions: Neet /jee QuestionsDocument27 paginiRedox Reactions: Neet /jee QuestionsAsher LaurierÎncă nu există evaluări
- Palaganas Hw2 Che 197Document5 paginiPalaganas Hw2 Che 197Elah Palaganas100% (1)
- Hydrocarbons Nitesh Devnani 2Document9 paginiHydrocarbons Nitesh Devnani 2nandkishorjadhav673Încă nu există evaluări
- 123.312 Advanced Organic Chemistry: Retrosynthesis: TutorialDocument10 pagini123.312 Advanced Organic Chemistry: Retrosynthesis: TutorialĐàoTrungHiếuÎncă nu există evaluări
- Deriving The Rate Equations For Product Inhibition Patterns in Bisubstrate Enzyme ReactionsDocument18 paginiDeriving The Rate Equations For Product Inhibition Patterns in Bisubstrate Enzyme ReactionsvijayalakshmiramanÎncă nu există evaluări
- Assignment 1 SolutionDocument4 paginiAssignment 1 Solutionliyanaraof100% (4)
- 4MBBS101 Lecture 5 Properties of Enzymes - Enzyme KineticsDocument51 pagini4MBBS101 Lecture 5 Properties of Enzymes - Enzyme KineticsArm UdomratÎncă nu există evaluări