Documente Academic
Documente Profesional
Documente Cultură
GMP For Brewing
Încărcat de
Nikka LopezTitlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
GMP For Brewing
Încărcat de
Nikka LopezDrepturi de autor:
Formate disponibile
Good Manufacturing Practices (GMPs)
What are GMPs?
Good Manufacturing Practices (GMPs) are the set of standards used to determine if a manufacturer is
maintaining practices set by federal, state and county regulations. GMPs include industry
recommendations. These easy to understand and execute standards allow companies to operate in
confidence that their facility is acting in good faith to satisfy requirements from any regulatory body that
may come and inspect the facility. Current U.S. Food and Drug Administration (FDA) regulations require
all breweries to have an active GMP program in place.
GMPs
cover
various
subjects.
Good
Manufacturing
Practices
for
Craft
Brewers
(GMPCBs)
are
representative
of
standards
needed
for
brewing
facilities.
There
are
more
GMPs
for
other
types
of
food
and
beverage
manufacturing
plants.
GMPCBs
include
establishing
strong
quality
management
systems,
obtaining
appropriate
quality
raw
materials,
establishing
standard
operating
procedures,
detecting
and
investigating
product
quality
deviations,
maintaining
reliable
laboratory
tests,
meeting
sanitary
and
processing
requirements,
and
packaging
and
labeling
according
to
approved
standards.
GMPCBs
help
to
prevent
contamination,
mix-‐
ups,
deviations,
failures
and
errors
and
are
the
foundation
for
any
brewery’s
quality
assurance
program.
These standards help to audit the facility, are set in checklist form, and are organized under these topics:
• Plants and Grounds
• Equipment and Utensils
• Sanitary Facilities and Controls
• Sanitary Operations
• Processes and Controls
• Personnel
These
standards
will
help
keep
the
facility
clean,
safe
and
pest-‐free
as
well
as
ensure
good
employee
hygiene.
What is HACCP?
Hazard Analysis and Critical Control Point (HACCP) is a management system in which food safety is
addressed through the analysis and control of biological, chemical and physical hazards from raw
material production including procurement and handling, manufacturing, distribution, and consumption
of the finished product. HACCP is designed for use in all segments of the food industry from growing,
harvesting, processing, manufacturing, distributing and merchandising to preparing food for
consumption. Prerequisite programs such as GMPCBs are an essential foundation for the development
and implementation of successful HACCP plans.
The U.S. FDA does not currently require breweries to have HACCP in place. However, it is strongly
recommended that a robust HACCP plan should be a routine part of all breweries’ standard operating
procedures.
This
document
was
created
by
the
Quality
subcommittee
to
the
Technical
Committee
of
the
Brewers
Association
to
help
all
American
brewers
maintain
good
practices
that
comply
with
regulations
as
well
as
create
a
safe
and
pride-‐filled
workplace.
The
Master
Brewers
Association
of
the
Americas
has
produced
a
similar
set
of
parameters
referred
to
as
Good
Brewing
Practices.
Though
another
good
reference,
this
piece
is
not
meant
to
have
exact
parity.
Why Good Manufacturing Practices for Craft Brewers are Necessary
Good Manufacturing Practices for Craft Brewers (GMPCBs) are the foundation for all quality, food safety
and organizational initiatives in any brewery. GMPCBs should be clearly defined and established before
a brewery begins production, regardless of production volume. They require continual improvement and
ongoing employee education.
GMPCBs are a critical aspect of any brewery environment for the following reasons:
• They
are
required
for
all
food
and
beverage
manufacturers
of
any
size
by
the
United
States
Food
and
Drug
Administration
(FDA)
and
can
also
be
enforced
by
state
and/or
local
health
departments.
• They
establish
clear
guidelines
for
the
hygiene
and
cleanliness
of
the
workers
and
their
workplace.
• They
are
the
foundation
of
any
quality
or
food
safety
program
in
any
manufacturing
setting.
• They
dramatically
decrease
the
food
safety
risk
for
the
brewery’s
customers.
• They
are
easy
to
execute
and
maintain.
Failure to implement GMPCBs can result in the following outcomes:
• Penalties
for
noncompliance
with
FDA
requirements
to
institute
GMPCBs
can
range
from
the
issuance
of
a
letter
notifying
the
brewery
of
a
violation
and
requesting
correction,
to
criminal
prosecution
of
the
individual
or
firm.
Other
FDA
regulatory
actions
can
include
seizure
of
products,
injunctions
to
refrain
from
production,
and
criminal
fines.
• Publicity
associated
with
an
FDA
enforcement
action
or
a
product
recall
created
by
a
lack
of
GMPCBs
can
result
in
decreased
public
confidence
and
loss
of
income.
Implementation
of
Good
Manufacturing
Practices
for
Craft
Brewers
will
instill
a
sense
of
pride
and
ownership
in
both
the
brewery’s
work
environment
and
its
standing
in
the
community.
S-ar putea să vă placă și
- Brewing Cleaning and SanitationDocument1 paginăBrewing Cleaning and SanitationJo SagaralÎncă nu există evaluări
- Enzymes in BrewingDocument9 paginiEnzymes in BrewingAdriana Fu Vivian100% (1)
- Best Practices Guide: QualityDocument24 paginiBest Practices Guide: QualityDavid HernandezÎncă nu există evaluări
- Bhs Brewing Ind ShierDocument69 paginiBhs Brewing Ind Shieralmstick2076Încă nu există evaluări
- Sustainable Brewing Concept Desalination ChemicalsDocument5 paginiSustainable Brewing Concept Desalination ChemicalsAndalusia Martha Novaline SiboroÎncă nu există evaluări
- Review Beer AgeingDocument25 paginiReview Beer AgeingTimothy WestÎncă nu există evaluări
- Brewing ProcessDocument1 paginăBrewing ProcessEly StrÎncă nu există evaluări
- Turbidity of BeerDocument39 paginiTurbidity of BeerManuel PlataÎncă nu există evaluări
- NR 23 Strictly Anaerobic Beer Spoilage Bacteria With Special Emphasis On New and Emerging Species Riikka JuvonenDocument37 paginiNR 23 Strictly Anaerobic Beer Spoilage Bacteria With Special Emphasis On New and Emerging Species Riikka JuvonenIvana NikolicÎncă nu există evaluări
- The Raise of Brettanomyces Yeast Species For Beer ProductionDocument6 paginiThe Raise of Brettanomyces Yeast Species For Beer ProductionJuan Antonio DiazÎncă nu există evaluări
- Topped With Hops Double IPADocument2 paginiTopped With Hops Double IPABautista John DexterÎncă nu există evaluări
- Lautering Back To BasicsDocument3 paginiLautering Back To BasicsVohinh NgoÎncă nu există evaluări
- Haccp and MicrobreweriesDocument14 paginiHaccp and MicrobreweriesJohn Henry Wells100% (5)
- Chemistry of Beer AgingDocument25 paginiChemistry of Beer AgingAntonio Imperi100% (1)
- Brewing ScienceDocument36 paginiBrewing ScienceVohinh NgoÎncă nu există evaluări
- Chemical Composition in Barley Grains and Malt Quality: July 2010Document37 paginiChemical Composition in Barley Grains and Malt Quality: July 2010Roberta AguiarÎncă nu există evaluări
- Enzymes in BrewingDocument8 paginiEnzymes in BrewingAlexandru TarțianÎncă nu există evaluări
- 32005mashing Unmalted 109Document15 pagini32005mashing Unmalted 109Pulbere NeagraÎncă nu există evaluări
- Barley For Brewing - Characteristic Changes During Malting BrewinDocument13 paginiBarley For Brewing - Characteristic Changes During Malting BrewinMariana EsquivelÎncă nu există evaluări
- BeerDocument21 paginiBeermichol20140% (1)
- Manual Analysis Methods For The Brewery Industry Pharo 2011-03Document70 paginiManual Analysis Methods For The Brewery Industry Pharo 2011-03Pablo Arostegui BarriosÎncă nu există evaluări
- Alternatives To Malt BrewingDocument9 paginiAlternatives To Malt BrewingPrincy AgnihotriÎncă nu există evaluări
- 01.30.55.110-Yeast Propagation and Management 2008 PDFDocument23 pagini01.30.55.110-Yeast Propagation and Management 2008 PDFÁnhXuânPhạmÎncă nu există evaluări
- Hops 2013Document30 paginiHops 2013odairnettoÎncă nu există evaluări
- Balling Formula Scrutiny of A Brewing DogmaDocument8 paginiBalling Formula Scrutiny of A Brewing DogmaEvis SandovalÎncă nu există evaluări
- The Main Fermentation in the Beer Brewing Process - Selected QuestionsDe la EverandThe Main Fermentation in the Beer Brewing Process - Selected QuestionsEvaluare: 5 din 5 stele5/5 (1)
- Beyond Malt HopsDocument46 paginiBeyond Malt Hopsbartolomej7Încă nu există evaluări
- The Art of Brewing and The Biology of Lager Yeast: Tom Pugh Miller Brewing CompanyDocument49 paginiThe Art of Brewing and The Biology of Lager Yeast: Tom Pugh Miller Brewing CompanyElly SufriadiÎncă nu există evaluări
- Malt & The Malting Process: Kelly J. Kuehl - National Sales Director The Country Malt GroupDocument33 paginiMalt & The Malting Process: Kelly J. Kuehl - National Sales Director The Country Malt GroupOleg Burlac100% (1)
- Brewing Technology ProtocolDocument56 paginiBrewing Technology ProtocolCeci Cruz PalmaÎncă nu există evaluări
- Craft Brewers Guide to Building a Sensory PanelDocument10 paginiCraft Brewers Guide to Building a Sensory PanelSaurabh BhawsarÎncă nu există evaluări
- Inspection MasterDocument36 paginiInspection MasterThì ĐặngÎncă nu există evaluări
- 13 M.sc. I & II Wine, Brewing and Alcohol TechnologyDocument41 pagini13 M.sc. I & II Wine, Brewing and Alcohol TechnologyCojocaru GeorgeÎncă nu există evaluări
- Forty Years of Brewing ResearchDocument27 paginiForty Years of Brewing ResearchAngélica Durán MuñozÎncă nu există evaluări
- Saccharomyces Yeasts Contribute On Innovative Brewing Fermentations - Innovacion en CervezasDocument9 paginiSaccharomyces Yeasts Contribute On Innovative Brewing Fermentations - Innovacion en CervezasemannuellyÎncă nu există evaluări
- VLB Labotech Delivery Programme enDocument78 paginiVLB Labotech Delivery Programme enbajricaÎncă nu există evaluări
- First Wort Hopping SummaryDocument5 paginiFirst Wort Hopping SummaryRodrigo Jacy Monteiro MartinsÎncă nu există evaluări
- BEER HOP EXTRACTION WITH SUPERCRITICAL CO2Document4 paginiBEER HOP EXTRACTION WITH SUPERCRITICAL CO2MaxPostvanderBurgÎncă nu există evaluări
- CBC Online Seminar Presentation Draught Beer Quality Workshop Addressing Draught Beer Dispense IssuesDocument40 paginiCBC Online Seminar Presentation Draught Beer Quality Workshop Addressing Draught Beer Dispense IssuesAnh TuanÎncă nu există evaluări
- Brewers Guide Access e PDFDocument182 paginiBrewers Guide Access e PDFAnonymous Y2J3QRfd100% (1)
- Nigeria Breweries Exam Papers Missing in TransitDocument28 paginiNigeria Breweries Exam Papers Missing in TransitAdam May100% (1)
- Arduino Controlled BrewingDocument5 paginiArduino Controlled BrewingJuliana ChavesÎncă nu există evaluări
- TECH-392 - European HACCP AttachDocument64 paginiTECH-392 - European HACCP AttachMiguel Meneses100% (2)
- Chapter 3 Brewing ProcessDocument16 paginiChapter 3 Brewing ProcessmaniiiiiiiiÎncă nu există evaluări
- Draught Beer Quality Manual - 2nd EditionDocument82 paginiDraught Beer Quality Manual - 2nd EditionMSalesÎncă nu există evaluări
- Brewers Diary PreviewDocument20 paginiBrewers Diary PreviewNicolás Ariel LázaroÎncă nu există evaluări
- Fundamentals of Beer and Hop ChemistryDocument12 paginiFundamentals of Beer and Hop ChemistryRbn SanchoÎncă nu există evaluări
- Parkin, Shellhammer - 2017 - Toward Understanding The Bitterness of Dry-Hopped BeerDocument6 paginiParkin, Shellhammer - 2017 - Toward Understanding The Bitterness of Dry-Hopped BeerPablo Perez DonosoÎncă nu există evaluări
- UB Micro ManualDocument95 paginiUB Micro ManualRiyan100% (1)
- Recipes for Brewing Different Types of Beers and AlesDe la EverandRecipes for Brewing Different Types of Beers and AlesÎncă nu există evaluări
- BJCPExamForDummies2018 Rev2Document67 paginiBJCPExamForDummies2018 Rev2ronsup67% (3)
- Brewing Technology From BriggsDocument14 paginiBrewing Technology From BriggsDuy NguyenÎncă nu există evaluări
- Advanced Chemistry of Beer and Brewing-Compatibility-Mode1 PDFDocument24 paginiAdvanced Chemistry of Beer and Brewing-Compatibility-Mode1 PDFRavi Radhoe100% (3)
- Brewing Materials and Processes: A Practical Approach to Beer ExcellenceDe la EverandBrewing Materials and Processes: A Practical Approach to Beer ExcellenceCharles W BamforthEvaluare: 2 din 5 stele2/5 (1)
- Brewery Lab & Quality Control: Rewery ABDocument29 paginiBrewery Lab & Quality Control: Rewery ABAlija IbrisevicÎncă nu există evaluări
- REVIVAL OF DRY HOPPING TECHNIQUESDocument3 paginiREVIVAL OF DRY HOPPING TECHNIQUESAlison AbreuÎncă nu există evaluări
- Monthly Fire Extinguisher Maintenance Log: Green Environmental Management System (Gems)Document2 paginiMonthly Fire Extinguisher Maintenance Log: Green Environmental Management System (Gems)Nikka LopezÎncă nu există evaluări
- Fire Safety Inspection ChecklistDocument8 paginiFire Safety Inspection ChecklistNikka LopezÎncă nu există evaluări
- Good To Great in Gods Eyes PDFDocument45 paginiGood To Great in Gods Eyes PDFNikka Lopez50% (2)
- Ch6 SolutionsDocument66 paginiCh6 SolutionsAtikah Rosely100% (1)
- We Will Start in 15 Minutes!!!Document18 paginiWe Will Start in 15 Minutes!!!Nikka LopezÎncă nu există evaluări
- 50 Important Physics Q&ADocument15 pagini50 Important Physics Q&Aamankumar sahuÎncă nu există evaluări
- Food Safety Training Guidance: WWW - Improveltd.co - UkDocument29 paginiFood Safety Training Guidance: WWW - Improveltd.co - UkNikka LopezÎncă nu există evaluări
- Checklist On PpeDocument3 paginiChecklist On PpeNikka LopezÎncă nu există evaluări
- WelcomeDocument7 paginiWelcomeNikka LopezÎncă nu există evaluări
- Level Sensor PDFDocument7 paginiLevel Sensor PDFNikka LopezÎncă nu există evaluări
- GratitudeDocument1 paginăGratitudeNikka LopezÎncă nu există evaluări
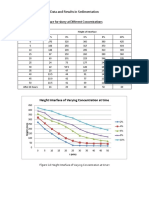
- Height Interface of Varying Concentration at Time: Data and Results in SedimentationDocument18 paginiHeight Interface of Varying Concentration at Time: Data and Results in SedimentationNikka LopezÎncă nu există evaluări
- Training Manual Food Safety Supervisor Advance (Level 2) : Retail & DistributionDocument42 paginiTraining Manual Food Safety Supervisor Advance (Level 2) : Retail & DistributionNikka LopezÎncă nu există evaluări
- Disease-Resistant and Herbicide-Resistant CropsDocument20 paginiDisease-Resistant and Herbicide-Resistant CropsNikka LopezÎncă nu există evaluări
- Foodmanual PDFDocument22 paginiFoodmanual PDFshavetadattÎncă nu există evaluări
- Campylobacter Organism Causes PDFDocument4 paginiCampylobacter Organism Causes PDFNikka LopezÎncă nu există evaluări
- ISO 22000 Food Safety Management Quality Manual PDFDocument46 paginiISO 22000 Food Safety Management Quality Manual PDFsadbad656% (9)
- Audit Checklist SampleDocument150 paginiAudit Checklist SampleNikka LopezÎncă nu există evaluări
- An Introduction to Food Safety Risk AnalysisDocument119 paginiAn Introduction to Food Safety Risk Analysisssaxena007709492% (12)
- Food Safety and Food QualityDocument173 paginiFood Safety and Food Qualitymacauboy_8980% (5)
- 2017 35 15038 Review of Microbial Pathogen Inactivation Relevant To Sous...Document58 pagini2017 35 15038 Review of Microbial Pathogen Inactivation Relevant To Sous...Nikka LopezÎncă nu există evaluări
- Audit Checklist SampleDocument2 paginiAudit Checklist SampleEnakhifo Victor100% (1)
- 2014 16 Risk Profile Listeria Monocytogenes in Raw MilkDocument156 pagini2014 16 Risk Profile Listeria Monocytogenes in Raw MilkNikka LopezÎncă nu există evaluări
- 2012 30 Standardisation of Hazard Analysis and Critical Control PointDocument12 pagini2012 30 Standardisation of Hazard Analysis and Critical Control PointNikka LopezÎncă nu există evaluări
- 2016 16 Risk Profile Campylobacter ST6964 With SISDocument57 pagini2016 16 Risk Profile Campylobacter ST6964 With SISNikka LopezÎncă nu există evaluări
- 2014 13 Assessment of Effects of Pasteurisation On Claimed Nutrition and Health Benefits of Raw MilkDocument24 pagini2014 13 Assessment of Effects of Pasteurisation On Claimed Nutrition and Health Benefits of Raw MilkNikka LopezÎncă nu există evaluări
- 2012 27 Clostridium Botulinum in ShellfishDocument84 pagini2012 27 Clostridium Botulinum in ShellfishNikka LopezÎncă nu există evaluări
- 2015 37 Risk Assessment of Fish Oil and Oxidised Fish OilDocument19 pagini2015 37 Risk Assessment of Fish Oil and Oxidised Fish OilNikka LopezÎncă nu există evaluări
- 2014 16 Risk Profile Listeria Monocytogenes in Raw MilkDocument81 pagini2014 16 Risk Profile Listeria Monocytogenes in Raw MilkNikka LopezÎncă nu există evaluări
- 2014 16 Risk Profile Listeria Monocytogenes in Raw MilkDocument81 pagini2014 16 Risk Profile Listeria Monocytogenes in Raw MilkNikka LopezÎncă nu există evaluări
- Bentham, Jeremy - Offences Against One's SelfDocument25 paginiBentham, Jeremy - Offences Against One's SelfprotonpseudoÎncă nu există evaluări
- Home - ECourt India Services Harpreet Singh by JN PatelDocument2 paginiHome - ECourt India Services Harpreet Singh by JN PatelIasam Groups'sÎncă nu există evaluări
- The Gazzat of PakistanDocument236 paginiThe Gazzat of PakistanFaheem UllahÎncă nu există evaluări
- Prince SmartFit CPVC Piping System Price ListDocument1 paginăPrince SmartFit CPVC Piping System Price ListPrince Raja38% (8)
- Disinvestment Policy of IndiaDocument7 paginiDisinvestment Policy of Indiahusain abbasÎncă nu există evaluări
- Napoleon Edwards Amended Complaint - RedactedDocument13 paginiNapoleon Edwards Amended Complaint - Redactedthe kingfishÎncă nu există evaluări
- Evidence Case List 1 STDocument1 paginăEvidence Case List 1 STEmmanuel Gomez de Peralta Jr.Încă nu există evaluări
- CHAPTER 2 FINANCIAL ANALYSIS AND PLANNINGDocument85 paginiCHAPTER 2 FINANCIAL ANALYSIS AND PLANNINGTarekegn DemiseÎncă nu există evaluări
- PCM10 LA6741 schematics documentDocument37 paginiPCM10 LA6741 schematics documentrmartins_239474Încă nu există evaluări
- Oliver Wyman Insurance Insights Edition 15 EnglishDocument7 paginiOliver Wyman Insurance Insights Edition 15 EnglishChiara CambriaÎncă nu există evaluări
- 2019 Graduation MessagesDocument4 pagini2019 Graduation MessagesKevin ProvendidoÎncă nu există evaluări
- Criminal Attempt: Meaning, Periphery, Position Explained Under The Indian Penal CodeDocument27 paginiCriminal Attempt: Meaning, Periphery, Position Explained Under The Indian Penal CodePrabhash ChandÎncă nu există evaluări
- ResultGCUGAT 2018II - 19 08 2018Document95 paginiResultGCUGAT 2018II - 19 08 2018Aman KhokharÎncă nu există evaluări
- Quotation Eoi/Rfp/Rft Process Checklist Goods And/Or ServicesDocument7 paginiQuotation Eoi/Rfp/Rft Process Checklist Goods And/Or ServicesTawanda KurasaÎncă nu există evaluări
- Cloud Control Basic Installation GuideDocument184 paginiCloud Control Basic Installation GuidesoftdesireÎncă nu există evaluări
- Schnittke - The Old Style Suite - Wind Quintet PDFDocument16 paginiSchnittke - The Old Style Suite - Wind Quintet PDFciccio100% (1)
- Trs EnrollmentDocument1 paginăTrs EnrollmentBrandon MillsÎncă nu există evaluări
- Bank StatementDocument2 paginiBank StatementZakaria EL MAMOUNÎncă nu există evaluări
- Marraige and CompatabilityDocument3 paginiMarraige and CompatabilityRamesh Menon100% (1)
- Emt - Aqeel AbbasDocument1 paginăEmt - Aqeel AbbasMuhammad Jawwad AhmedÎncă nu există evaluări
- Universal Principles of Biomedical EthicsDocument16 paginiUniversal Principles of Biomedical EthicsMEOW41100% (3)
- Follow Up of DCPS Emergency Response Planning and ReadinessDocument12 paginiFollow Up of DCPS Emergency Response Planning and ReadinessABC7NewsÎncă nu există evaluări
- Unit 6 LessonsDocument25 paginiUnit 6 Lessonsapi-300955269Încă nu există evaluări
- Hacienda Fatima, Et Al. v. National Federation of Sugarcane Workers-Food and General Trade, G.R. No. 149440, Jan. 28, 2003Document8 paginiHacienda Fatima, Et Al. v. National Federation of Sugarcane Workers-Food and General Trade, G.R. No. 149440, Jan. 28, 2003Martin SÎncă nu există evaluări
- Penaranda Vs Bangaga Plywood CorpDocument4 paginiPenaranda Vs Bangaga Plywood CorpDeniseEstebanÎncă nu există evaluări
- Jazz - Score and PartsDocument44 paginiJazz - Score and PartsMaanueel' Liizaamaa'Încă nu există evaluări
- Comparing Police Systems in Southeast AsiaDocument2 paginiComparing Police Systems in Southeast AsiaKrizzia SoguilonÎncă nu există evaluări
- Ra Construction and Development Corp.Document3 paginiRa Construction and Development Corp.janus jarderÎncă nu există evaluări