Documente Academic
Documente Profesional
Documente Cultură
Chemical Reaction Engineering Ph. D. Qualifier Examination Open Book (Scott Fogler) Exam
Încărcat de
Nicole Anne BorromeoDescriere originală:
Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Chemical Reaction Engineering Ph. D. Qualifier Examination Open Book (Scott Fogler) Exam
Încărcat de
Nicole Anne BorromeoDrepturi de autor:
Formate disponibile
Chemical Reaction Engineering
Ph. D. Qualifier examination
Open book (Scott Fogler) exam
1. A second order irreversible liquid phase reaction A +B C takes place in a Plug
flow reactor (PFR). The feed to the reactor is 50 kmol/hr of each reactant. The
total conversion is 50%. The reaction kinetics is rA = -k CACB where k is 0.5 m3/
kmol/hr. The same reaction is to be performed in a CSTR of equal volume.
Calculate the conversion in the latter case. The concentration of A in the feed is 5
mol/lit.
If the reaction was in gas phase, would the answer be different? If yes, write the
relevant equations (do not solve) to recalculate the conversion. [20]
2. a. For a reaction A B C (all are irreversible and elementary reactions), give
the equations to be solved to obtain the concentration of B in a batch reactor as a
function of time. Give qualitative profiles of all the species as a function of time.
b. For an irreversible reaction the reaction rate doubles when
temperature is increased from 300K to 310 K. Calculate the activation energy.
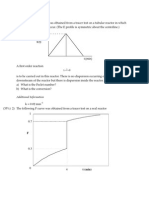
c.For the following graph of a solid-catalytic reaction, in each case which process
governs the overall rate? -intrinsic kinetics, external mass transfer or intraparticle
diffusion. Justify your answer.
A
ln (-rA)
B
1/T
[20]
3. A first order homogeneous liquid-phase reaction is carried out in an ideal stirred
tank reactor. The concentration of the reactant in the feed is 3.0 kmol/m3 and the
volumetric flow rate is 60 x 10-6 m3/sec. The density and specific heat for the
reaction mixture are constant at 1000 kg/m3 and 4.19 kJ/kg.K, respectively. The
reactor volume is 18 lit. There is no product in the feed stream and reactor
operates adiabatically. The heat and rate of reaction are -2.09 x 108 J/kmol and
rA = - 4.48 x 106 CA exp (- 62,800/RT) kmol/m3/sec
where, activation energy is in SI units. CA is the reactant concentration in
kmol/m3. T is in K and R= 8.314 J/mol/K. If the feed is at 273 K what is the
steady state product temperature and conversion? (Hint: the conversion is beyond
90%). [20]
4. The rate of isomerization of n-butane on a silica-alumna spherical catalyst
measured at 5 atm and 50oC, in a laboratory reactor with high turbulence in the
gas phase surrounding the catalyst pellet is given below. Turbulence ensures that
the external diffusion resistances are negligible. Kinetic studies indicate that the
rate is first order and irreversible. The effective diffusivity is 0.08 cm2/sec at the
reaction conditions, and the density of the catalyst pellet is 1 g/cm3 regardless of
the size. The measured rates when pure n-butane surrounds the catalyst are as
follows:
Particle 1/16 1/8 ¼ 3/8 1
diameter (in)
Rate 4.88 x 10-4 4.85 x 10-4 4.01 x 10-4 3.54 x 10-4 1 x 10-5
mol/sec/g of
catalyst
From the above data, determine the intrinsic first order rate constant in the
absence of intraparticle diffusion. Also predict the rate if the particle size is 1.5
inch. It may be assumed that effectiveness factor () is inverse of Thiele modulus
() (i.e. = 1/) when 0.25. [20]
5. A reactor of irregular geometry offers following E curve. Calculate the average
residence time. Also determine the conversion in each of the following cases (i
and ii) if complete segregation of feed streams is assumed to take place.
i) For a first order reaction rA = -0.1 CA (kmols/hr/m3)
ii) For a second order reaction in problem 1,
Also, what will be the conversion for the first order reaction in case-i for the
maximum mixedness case?
E(t) hr-1
2.0
0.0 0.5 1.0
Time (hr)
[20]
S-ar putea să vă placă și
- Cat Flash File ReadxrefDocument179 paginiCat Flash File ReadxrefНиколай Белый89% (9)
- O Level Biology Practice Questions And Answers EnzymesDe la EverandO Level Biology Practice Questions And Answers EnzymesEvaluare: 5 din 5 stele5/5 (1)
- Lab Manual (Cre)Document41 paginiLab Manual (Cre)akash100% (1)
- Assignment 1Document6 paginiAssignment 1Yi Hong LowÎncă nu există evaluări
- Exercise 8 Kinetics of Hydrolysis of Ethyl AcetateDocument6 paginiExercise 8 Kinetics of Hydrolysis of Ethyl AcetatePalak BansalÎncă nu există evaluări
- S 1514Document4 paginiS 1514bacuoc.nguyen356Încă nu există evaluări
- Commander's Smartbook Equipment CatalogDocument59 paginiCommander's Smartbook Equipment CatalogWilliam 'Junior' Hof100% (1)
- RMIPA Port Master PlanDocument362 paginiRMIPA Port Master Planjoncpasierb100% (6)
- 2 CreDocument3 pagini2 CreDamien MarleyÎncă nu există evaluări
- Tut1 2016 QDocument5 paginiTut1 2016 QAbhishek SardaÎncă nu există evaluări
- CH3010 2010Document1 paginăCH3010 2010deepakÎncă nu există evaluări
- Cro Tut8Document13 paginiCro Tut8Ernst SmitÎncă nu există evaluări
- DQE January 2001: Additional InformationDocument12 paginiDQE January 2001: Additional InformationryezhuÎncă nu există evaluări
- 2010 ChE 471 HW 8 FinalDocument4 pagini2010 ChE 471 HW 8 FinalSiti HannaÎncă nu există evaluări
- CRE IdocxDocument8 paginiCRE IdocxParth DesaiÎncă nu există evaluări
- Homework 2 - 2020 - 01 - Sent (v4)Document4 paginiHomework 2 - 2020 - 01 - Sent (v4)DeividÎncă nu există evaluări
- Reactor Design IIDocument68 paginiReactor Design IIKORAMA KIEN0% (1)
- Tutorial QuestionsDocument8 paginiTutorial QuestionsMaame Efua Neizer100% (1)
- Chemical Reactors - Problems of Reactor Association 47-60: (Exam Jan'09)Document6 paginiChemical Reactors - Problems of Reactor Association 47-60: (Exam Jan'09)Alfredo ZuñigaÎncă nu există evaluări
- Boon PinDocument13 paginiBoon PinjayaprinaÎncă nu există evaluări
- Assignment 05Document2 paginiAssignment 05Rashmi SahooÎncă nu există evaluări
- Ki KBR H C Ki BR H C: Oducts B ADocument2 paginiKi KBR H C Ki BR H C: Oducts B AnaverfallÎncă nu există evaluări
- Reaactor 3 SheetDocument5 paginiReaactor 3 SheetmohamedÎncă nu există evaluări
- Assignment 2: (Subject: Ch.E. 401 Chemical Reactor Design) Related To CLO1Document4 paginiAssignment 2: (Subject: Ch.E. 401 Chemical Reactor Design) Related To CLO1imtiazÎncă nu există evaluări
- P4E2: Kinetics of Homogeneous Reaction in Batch and Continuous Stirred-Tank Reactor at Two Different TemperatureDocument7 paginiP4E2: Kinetics of Homogeneous Reaction in Batch and Continuous Stirred-Tank Reactor at Two Different TemperaturejayaprinaÎncă nu există evaluări
- Lecture 20: The Fixed Bed Catalytic Reactor: RXN RXNDocument10 paginiLecture 20: The Fixed Bed Catalytic Reactor: RXN RXNreddi ramuÎncă nu există evaluări
- Adiabatic FBR DesignDocument10 paginiAdiabatic FBR DesignRana UzairÎncă nu există evaluări
- Chemical Reaction Engineering II Tutorial Sheet No. VDocument1 paginăChemical Reaction Engineering II Tutorial Sheet No. VShashank_PardhikarÎncă nu există evaluări
- CHE3044F, 2013: Reactor Design 1: TUTORIAL 6Document4 paginiCHE3044F, 2013: Reactor Design 1: TUTORIAL 6nmhatityeÎncă nu există evaluări
- Ach - 1115 - Chemical Reaction Engineering-I, 29-05-2013, May-2013Document2 paginiAch - 1115 - Chemical Reaction Engineering-I, 29-05-2013, May-2013rahulÎncă nu există evaluări
- (P01, C01, C02, C2, C3) : Confidential EH/JUN 2014/CHE584/594Document11 pagini(P01, C01, C02, C2, C3) : Confidential EH/JUN 2014/CHE584/594Addison JuttieÎncă nu există evaluări
- R (DC /DT) 0.2 Mol L C: ASSIGNMENT #2 - Reaction Kinetics 2Document2 paginiR (DC /DT) 0.2 Mol L C: ASSIGNMENT #2 - Reaction Kinetics 2AndreÎncă nu există evaluări
- Kinetics PretestDocument4 paginiKinetics PretestAngeline SmithÎncă nu există evaluări
- Advanced Chemical Reaction EngineeringDocument1 paginăAdvanced Chemical Reaction EngineeringIbmWasuserÎncă nu există evaluări
- Delhi Public School: Nacharam/ Mahendra Hills/ NadergulDocument3 paginiDelhi Public School: Nacharam/ Mahendra Hills/ Naderguleeshwar saagarÎncă nu există evaluări
- Models - Chem.round Jet BurnerDocument44 paginiModels - Chem.round Jet BurnerLe Nguyen Phuc ThienÎncă nu există evaluări
- DPP 02Document1 paginăDPP 02prathmfedÎncă nu există evaluări
- Kinetics Revision Note-1Document8 paginiKinetics Revision Note-1Madhavilatha LoganathanÎncă nu există evaluări
- Sample Exams Problems CHE 402Document3 paginiSample Exams Problems CHE 402Ricardo VelozÎncă nu există evaluări
- NR-320802 Chemical Reaction Engineering-IDocument8 paginiNR-320802 Chemical Reaction Engineering-ISrinivasa Rao G100% (1)
- 2010 ChE 471 HW 8 FinalDocument4 pagini2010 ChE 471 HW 8 FinalpinhonceteÎncă nu există evaluări
- Soal Chapter 21Document5 paginiSoal Chapter 2121-096 Kharisma Theresia Adelina ButarButarÎncă nu există evaluări
- Tutorial 4Document3 paginiTutorial 4EreenÎncă nu există evaluări
- Chapter 4 - Chemical KineticsDocument7 paginiChapter 4 - Chemical KineticsMADHAVÎncă nu există evaluări
- Practice Problems On Multiple Reactions and Non-Isothermal ReactionsDocument1 paginăPractice Problems On Multiple Reactions and Non-Isothermal ReactionsArunima GhoseÎncă nu există evaluări
- Department of Chemical Engineering, Iit Delhi Reactor Sizing Problems Assignment-2Document2 paginiDepartment of Chemical Engineering, Iit Delhi Reactor Sizing Problems Assignment-2ShubhamGuptaÎncă nu există evaluări
- Compulsory Question: Question (1) : 10 Marks: A B C DDocument4 paginiCompulsory Question: Question (1) : 10 Marks: A B C Dsushant mouleÎncă nu există evaluări
- 1 Mark QuestionsDocument19 pagini1 Mark QuestionsSsÎncă nu există evaluări
- ChEMICAL KINETICS - QUESTIONSDocument3 paginiChEMICAL KINETICS - QUESTIONSChhabi YadavÎncă nu există evaluări
- Microsoft Word - 6 - Prob RTD-Non Id React 11-12 61-78 - EnglishDocument9 paginiMicrosoft Word - 6 - Prob RTD-Non Id React 11-12 61-78 - EnglishPavithra Sivaraja100% (1)
- Chemical KineticsDocument6 paginiChemical KineticsThala SkÎncă nu există evaluări
- CROExam 2013Document4 paginiCROExam 2013KarinaÎncă nu există evaluări
- Cre 1Document52 paginiCre 1Jayakaran PachiyappanÎncă nu există evaluări
- Tutorial CREDocument1 paginăTutorial CREShashank_PardhikarÎncă nu există evaluări
- Ass 1Document2 paginiAss 1poorvig570Încă nu există evaluări
- Che 125: Chemical Reaction Engineering IDocument2 paginiChe 125: Chemical Reaction Engineering IJelor GallegoÎncă nu există evaluări
- CHP 482 - TutorialsDocument4 paginiCHP 482 - Tutorialsgeofrey oburuÎncă nu există evaluări
- JAB-TALLER 3er PARCIAL IRQ-2020-1Document5 paginiJAB-TALLER 3er PARCIAL IRQ-2020-1JESSICA PAOLA TORO VASCOÎncă nu există evaluări
- Exam I Sem I 2011 12 Cheng 323Document7 paginiExam I Sem I 2011 12 Cheng 323Faisal MumtazÎncă nu există evaluări
- Tutorial 3 QuestionDocument3 paginiTutorial 3 Questionnur hidayatiÎncă nu există evaluări
- Kinetics of Hydrolysis of Ethyl EsterDocument6 paginiKinetics of Hydrolysis of Ethyl EsterJawad AhmadÎncă nu există evaluări
- CHBE 355 Final Examination W23Document7 paginiCHBE 355 Final Examination W23Awesome GeneralÎncă nu există evaluări
- A Modern Course in Statistical PhysicsDe la EverandA Modern Course in Statistical PhysicsEvaluare: 3.5 din 5 stele3.5/5 (2)
- Background of The StudyDocument6 paginiBackground of The StudyNicole Anne BorromeoÎncă nu există evaluări
- 0 - 2019-03-13 17-58 - Page - 1Document2 pagini0 - 2019-03-13 17-58 - Page - 1Nicole Anne BorromeoÎncă nu există evaluări
- Week 7 2014Document21 paginiWeek 7 2014Nicole Anne BorromeoÎncă nu există evaluări
- Fig.4.1 Washer Specification Sheet: Date: 05-22-2018Document9 paginiFig.4.1 Washer Specification Sheet: Date: 05-22-2018Nicole Anne BorromeoÎncă nu există evaluări
- Specific Heat Relations of Ideal GasesDocument8 paginiSpecific Heat Relations of Ideal GasesNicole Anne BorromeoÎncă nu există evaluări
- Trial 2 Natural SoilDocument1 paginăTrial 2 Natural SoilNicole Anne BorromeoÎncă nu există evaluări
- The Aquino Administration Passed Republic Act 1035Document1 paginăThe Aquino Administration Passed Republic Act 1035Nicole Anne BorromeoÎncă nu există evaluări
- APPENDIX C ComputationsDocument9 paginiAPPENDIX C ComputationsNicole Anne BorromeoÎncă nu există evaluări
- ACTIVITY ON ParticulatesDocument7 paginiACTIVITY ON ParticulatesNicole Anne BorromeoÎncă nu există evaluări
- Che 520L: Prelim Exam: Calculations 1Document4 paginiChe 520L: Prelim Exam: Calculations 1Nicole Anne BorromeoÎncă nu există evaluări
- S Announcement 15622Document22 paginiS Announcement 15622Nicole Anne BorromeoÎncă nu există evaluări
- Lecture 5 Block Diagram Representation of Control Systems PDFDocument60 paginiLecture 5 Block Diagram Representation of Control Systems PDFNicole Anne BorromeoÎncă nu există evaluări
- Lecture 5 Block Diagram Representation of Control Systems PDFDocument60 paginiLecture 5 Block Diagram Representation of Control Systems PDFNicole Anne BorromeoÎncă nu există evaluări
- Description Possible RemediesDocument4 paginiDescription Possible RemediesNicole Anne BorromeoÎncă nu există evaluări
- Iii. Proposed Treatment Plant: ScreeningDocument2 paginiIii. Proposed Treatment Plant: ScreeningNicole Anne BorromeoÎncă nu există evaluări
- Lecture 11Document6 paginiLecture 11Nicole Anne Borromeo0% (1)
- LAGUNDIDocument4 paginiLAGUNDINicole Anne BorromeoÎncă nu există evaluări
- Ii. Waste CharacterizationDocument3 paginiIi. Waste CharacterizationNicole Anne BorromeoÎncă nu există evaluări
- Assignment Problems PDFDocument132 paginiAssignment Problems PDFNicole Anne BorromeoÎncă nu există evaluări
- Visag - Part - 1 of 2Document25 paginiVisag - Part - 1 of 2Rajveer SinghÎncă nu există evaluări
- MOI - Riyadh-UL Control Panel With VFD (2 Duty + 1 Standby)Document17 paginiMOI - Riyadh-UL Control Panel With VFD (2 Duty + 1 Standby)Emad AbushrarÎncă nu există evaluări
- Kashimali Aluminium - Karnataka - Anodizing Plant - 18.11.2023Document7 paginiKashimali Aluminium - Karnataka - Anodizing Plant - 18.11.2023Manjunath NemagoudarÎncă nu există evaluări
- Quick Exhaust Solenoid Valves: NC NODocument4 paginiQuick Exhaust Solenoid Valves: NC NOMuhammad AsadÎncă nu există evaluări
- Radial Turbine Design ProcessDocument15 paginiRadial Turbine Design ProcessKamaldeep GuptaÎncă nu există evaluări
- France en Dec. 2022 v3Document37 paginiFrance en Dec. 2022 v3Tarek OsmanÎncă nu există evaluări
- Wyser - Manutentore Meccanico LFDocument6 paginiWyser - Manutentore Meccanico LFMorfferroÎncă nu există evaluări
- Carbon Adsorption VaporDocument12 paginiCarbon Adsorption VaporMehdi Hajd KacemÎncă nu există evaluări
- Penggantian Relay Dist Ke LCD & Progress Koordinasi Penghantar Dan PembangkitDocument11 paginiPenggantian Relay Dist Ke LCD & Progress Koordinasi Penghantar Dan PembangkitCahyo Tri HartoyoÎncă nu există evaluări
- Module No 3: Liquefied Natural Gas Markets, Consumption, Measurement & CalculationDocument30 paginiModule No 3: Liquefied Natural Gas Markets, Consumption, Measurement & CalculationAndreiÎncă nu există evaluări
- High Temperature Primary Batteries For Venus Surface MissionsDocument24 paginiHigh Temperature Primary Batteries For Venus Surface MissionsEsau Hernandez CalosÎncă nu există evaluări
- MM435 - CDP-2 - Corrosion CellsDocument24 paginiMM435 - CDP-2 - Corrosion CellsAbdul Ahad ShamsÎncă nu există evaluări
- Electrical Disturbances in LV Networks Due To Energization of MV NetworksDocument5 paginiElectrical Disturbances in LV Networks Due To Energization of MV NetworksAlejandro Solis GomezÎncă nu există evaluări
- ME8512 - TE - AU Practical Students Individual QuestionDocument4 paginiME8512 - TE - AU Practical Students Individual Questionsikkandar faizÎncă nu există evaluări
- All AcDocument11 paginiAll Acfb_menotifyÎncă nu există evaluări
- KKT Chillers PI ECO EngDocument2 paginiKKT Chillers PI ECO EngjltenorioÎncă nu există evaluări
- Blacksmithing 101 - How To Make A Forge and Start Hammering Metal - Popular MechanicsDocument6 paginiBlacksmithing 101 - How To Make A Forge and Start Hammering Metal - Popular MechanicsByram JÎncă nu există evaluări
- Temperature MeasuremenntDocument25 paginiTemperature MeasuremenntJan Edward L. SuarezÎncă nu există evaluări
- Alpha, Beta and Gamma RadiationDocument8 paginiAlpha, Beta and Gamma RadiationFarhan SadiqueÎncă nu există evaluări
- Size CalculationDocument13 paginiSize CalculationGopal DasiriÎncă nu există evaluări
- EN13480-3 Chap12 ModificationsDocument37 paginiEN13480-3 Chap12 ModificationsPrashant ChauhanÎncă nu există evaluări
- OIDA Improved CarbonizerDocument14 paginiOIDA Improved CarbonizerRicardo F. OrgeÎncă nu există evaluări
- Full ReportDocument44 paginiFull ReportCes ShengÎncă nu există evaluări
- Fallsem2016-17 1286 RM001 27-Jul-2016 Mee1003 THDocument74 paginiFallsem2016-17 1286 RM001 27-Jul-2016 Mee1003 THaman agarwalÎncă nu există evaluări
- ATP Production Through Lipid CatabolismDocument1 paginăATP Production Through Lipid CatabolismZaivie VillanobosÎncă nu există evaluări
- Position On Strategic GraphiteDocument26 paginiPosition On Strategic GraphitemcnpopaÎncă nu există evaluări