Documente Academic
Documente Profesional
Documente Cultură
WHO Classification of Tumours of The Central Nervous
Încărcat de
intan rosli0 evaluări0% au considerat acest document util (0 voturi)
48 vizualizări1 paginăWHO Classification of Tumours of the Central Nervous
Titlu original
WHO Classification of Tumours of the Central Nervous
Drepturi de autor
© © All Rights Reserved
Formate disponibile
PDF, TXT sau citiți online pe Scribd
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentWHO Classification of Tumours of the Central Nervous
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PDF, TXT sau citiți online pe Scribd
0 evaluări0% au considerat acest document util (0 voturi)
48 vizualizări1 paginăWHO Classification of Tumours of The Central Nervous
Încărcat de
intan rosliWHO Classification of Tumours of the Central Nervous
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PDF, TXT sau citiți online pe Scribd
Sunteți pe pagina 1din 1
reveals identical TP53 mutations in both
gemistocytes and non-gemistocytic tu-
mour cells {2094}. Although it has been
reported that the gemistocytic variant
may be particularly prone to progression
to anaplastic astrocytoma and glioblas-
toma {1377,2204,2273}, this does not jus-
tify a general classification as anaplastic
astrocytoma {320,2273}, nor is this im-
pression based on current molecular
Fig. 1.06 Diffuse astrocytoma. Low cellularity and
characterization, in particular knowledge Fig. 1.07 Diffuse astrocytoma with extensive mucoid
nuclear atypia.
of IDH mutation status. degeneration and cobweb-like architecture. This pattern
was previously designated protoplasmic astrocytoma.
Immunophenotype
change may be present, but most cells Diffuse astrocytomas reliably express
look like one another, without the admix- distinction of true neoplasia from reac-
GFAP, although to various degrees and
ture of gemistocytes more often seen in tive gliosis {347,359}. Strong nuclear p53
not in all tumour cells. In particular, small
reactions to injury. Pre-existing cell types expression is also frequently observed,
round cells with scanty cytoplasm and
(e.g. neurons) are often entrapped. consistent with the high incidence of
processes tend not to label avidly for
TP53 mutations in diffuse astrocytoma
Intraoperative diagnosis GFAP. In these cases, immunopositiv-
{915}. However, the use of p53 immuno-
ity may be restricted to a small perinu-
The smear/squash technique is often positivity to reflect TP53 mutation is not
clear rim and to admixed neoplastic cell
used during stereotaxic biopsies and entirely sensitive or specific {1530,1771}.
processes in the fibrillary tumour back-
yields similar findings, although this In contrast, ATRX expression is almost
ground {1292}. Vimentin is typically im-
method is highly unreliable for estimating invariably lost in the setting of ATRX mu-
munopositive as well, with a labelling pat-
cellularity. Many histological features are tations, which also feature prominently in
tern approximating that of GFAP {995}.
exaggerated and amplified (e.g. nuclear diffuse astrocytoma (see Genetic profile)
The signature molecular characteristics
folds, abnormal chromatin pattern, and {361,1160,1215,2105}. ATRX typically,
of diffuse astrocytoma (see Genetic pro-
astrocytic processes). The presence of demonstrates strong nuclear expression
file) can often be demonstrated immuno-
many round to oval nuclei with smooth in normal, unmutated tissue; therefore,
histochemically. For example, expression
chromatin can indicate the presence of retention of immunolabelling in non-
of R132H-mutant IDH1 (the IDH1 R132H
an apparent oligodendroglial component neoplastic vasculature and admixed
mutation accounts for about 90% of all
or (if the nuclei are less prominent) back- neuronal, glial, and microglial elements
glioma-associated IDH mutations) can
ground white matter. Histologically, there serves as a necessary internal control for
be detected using a mutation-specific
may be significant variation between tu- the accurate interpretation of a negative
antibody {360}. In mutant tumours, all
mours and within the same lesion. ATRX immunostaining pattern. Finally,
neoplastic cells typically exhibit some
consistent with its inapparent mitotic ac-
Growthfraction degree of cytoplasmic (stronger) and
tivity, diffuse astrocytoma nearly always
The growth fraction as determined by the nuclear (weaker) labelling, provided the
has a Ki-67 proliferation index of < 4%
Ki-67 proliferation index is usually < 4%. staining preparation used is technically
{492,1137,1223,2059}.
The gemistocytic neoplastic astrocytes adequate {359}. For this reason, R132H-
show a significantly lower rate of prolif- mutant IDH1 immunohistochemistry has Cell of origin
eration than does the intermingled small- become an invaluable diagnostic ad- The available evidence suggests that
cell component {1046,1372,1377,1847, junct, not only in the molecular stratifi- IDH-mutant and 1p/19q-codeleted oli-
2706,2812}. However, microdissection cation of diffuse glioma, but also in the godendrogliomas, IDH-mutant diffuse
Fig. 1.08 Diffuse astrocytoma. A Cytoplasm and cell processes show a variable extent of GFAP immunoreactivity. B The Ki-67 proliferation index is low.
20 Diffuse gliomas
S-ar putea să vă placă și
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
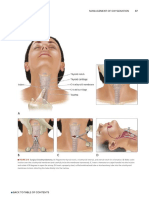
- Surgical CricothyroidotomyDocument1 paginăSurgical Cricothyroidotomyintan rosliÎncă nu există evaluări
- Figure 3-7: PART I Neuroanatomy and Neuroanatomic LocalizationDocument1 paginăFigure 3-7: PART I Neuroanatomy and Neuroanatomic Localizationintan rosliÎncă nu există evaluări
- Orthopaedic AnatomyDocument1 paginăOrthopaedic Anatomyintan rosliÎncă nu există evaluări
- Translocation NeuromaDocument1 paginăTranslocation Neuromaintan rosliÎncă nu există evaluări
- Translocation NeuromaDocument1 paginăTranslocation Neuromaintan rosliÎncă nu există evaluări
- Corticosteroid Injection For de Quervain's Tenosynovitis (Review)Document19 paginiCorticosteroid Injection For de Quervain's Tenosynovitis (Review)intan rosliÎncă nu există evaluări
- 7.2 Non-Absorbable Suture MaterialsDocument1 pagină7.2 Non-Absorbable Suture Materialsintan rosliÎncă nu există evaluări
- Antimicrobial Resistance in LeprosyDocument6 paginiAntimicrobial Resistance in Leprosyintan rosliÎncă nu există evaluări
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (400)
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (74)
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (121)
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)
- Article II Protection Rights of Children: Child Abuse, Exploitation, and DiscriminationDocument12 paginiArticle II Protection Rights of Children: Child Abuse, Exploitation, and DiscriminationMicky MoranteÎncă nu există evaluări
- Hiromi Shinya - The Enzyme Factor 1Document176 paginiHiromi Shinya - The Enzyme Factor 1Trung tâm ngoại ngữ English Zone An ĐồngÎncă nu există evaluări
- Ventricular Tachycardia Bsn3b-Grp1Document35 paginiVentricular Tachycardia Bsn3b-Grp1Jessica RamosÎncă nu există evaluări
- Frederick Classification of HyperlipidemiaDocument2 paginiFrederick Classification of HyperlipidemiaBilly BuswayÎncă nu există evaluări
- 2017 - Fut VirolDocument10 pagini2017 - Fut VirolKashif HaleemÎncă nu există evaluări
- Shear's Cysts of The Oral and Maxillofacial Regions-10-14Document5 paginiShear's Cysts of The Oral and Maxillofacial Regions-10-14muhammad arif100% (1)
- Competency Assessment Tier1-Completed-2021Document20 paginiCompetency Assessment Tier1-Completed-2021api-439027462Încă nu există evaluări
- Vestibular Neuritis HandoutDocument3 paginiVestibular Neuritis HandoutPrisilia QurratuAiniÎncă nu există evaluări
- ITI Decision Tree Immediate PlacementDocument1 paginăITI Decision Tree Immediate Placementxiaoxin zhangÎncă nu există evaluări
- PRELIM CHN LECTURE HandoutsDocument22 paginiPRELIM CHN LECTURE HandoutsJOYCE ANN PEREGRINOÎncă nu există evaluări
- Child Adol Traditional Biopsychosocial TemplateDocument11 paginiChild Adol Traditional Biopsychosocial Templateyeney armenteros100% (1)
- Peptic Ulcer DiseaseDocument75 paginiPeptic Ulcer DiseaseSarah Gaile Tio Inac100% (4)
- Textbook PDFDocument660 paginiTextbook PDFNguyên Tâm Tuệ Nguyễn100% (1)
- Lichterfeld Kottner2019Document23 paginiLichterfeld Kottner2019Nur AnjelinaÎncă nu există evaluări
- HemoroidDocument24 paginiHemoroidfauzieÎncă nu există evaluări
- Jurnal Infeksi Saluran PencernaanDocument22 paginiJurnal Infeksi Saluran PencernaanDini Okta PutriÎncă nu există evaluări
- Ayurveda Can Play A Significant Role in Fight Against CoronavirusDocument5 paginiAyurveda Can Play A Significant Role in Fight Against CoronavirusAzmaFaiqueÎncă nu există evaluări
- GCSE History Britain: Health and The People, C1000-Present DayDocument30 paginiGCSE History Britain: Health and The People, C1000-Present DayAniqah ShujaatÎncă nu există evaluări
- Plantar Fasciitis: What Is The Plantar Fasciae?Document6 paginiPlantar Fasciitis: What Is The Plantar Fasciae?mattamooreÎncă nu există evaluări
- Covid-19 Rat Common-List enDocument27 paginiCovid-19 Rat Common-List enNavneet ValechaÎncă nu există evaluări
- First AidDocument10 paginiFirst AidSanthiya venkadesanÎncă nu există evaluări
- Aneurysmal Bone CystDocument5 paginiAneurysmal Bone CystAustine OsaweÎncă nu există evaluări
- AnthraxDocument14 paginiAnthraxQuenzil LumodÎncă nu există evaluări
- ScabiesDocument18 paginiScabiesRashidul IslamÎncă nu există evaluări
- Standing BaduanjinDocument34 paginiStanding Baduanjinserena100% (1)
- Formulation and in Vitro Evaluation of Bilayer Tablets of Nebivolol Hydrochloride and Nateglinide For The Treatment of Diabetes and HypertensionDocument14 paginiFormulation and in Vitro Evaluation of Bilayer Tablets of Nebivolol Hydrochloride and Nateglinide For The Treatment of Diabetes and HypertensionDIKAÎncă nu există evaluări
- Hazard and RiskDocument13 paginiHazard and RiskMarites BalmasÎncă nu există evaluări
- A Correlational Study of Systemic Blood Pressure and IOP HL SITHOLEDocument6 paginiA Correlational Study of Systemic Blood Pressure and IOP HL SITHOLESurya Nirmala DewiÎncă nu există evaluări
- Resume Xanthomonas Campestris - Fatimah Az Zahra - 2014191006-1Document3 paginiResume Xanthomonas Campestris - Fatimah Az Zahra - 2014191006-1unila satuÎncă nu există evaluări
- Care Plan Worksheet Mental Health 2022Document3 paginiCare Plan Worksheet Mental Health 2022tahani.mah147Încă nu există evaluări