Documente Academic
Documente Profesional
Documente Cultură
Activity 2 PH Measurement and Buffer Capacity
Încărcat de
Althea ValenzuelaDescriere originală:
Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Activity 2 PH Measurement and Buffer Capacity
Încărcat de
Althea ValenzuelaDrepturi de autor:
Formate disponibile
Name Group
ACTIVITY pH Measurement and Buffer Section Schedule
2 Capacity
Instructor Date
ACTIVITY 2
pH Measurement and Buffer Capacity
INTRODUCTION
A. Measurement of pH
The measurement of pH is an important laboratory procedure because the pH of a solution
affects the activity of biological molecules. Body fluids have a normal pH range that allows biochemical
activities to occur. A deviation from the normal pH ranges indicates disorder. Following are the normal
pH range of some body fluids:
Fluid pH
Gastric juice 1.0 – 3.0
Urine 5.5 – 7.0
Saliva 6.5 – 7.5
Blood 7.35 – 7.45
Intestinal secretions 7.7
Pancreatic juice 8
The pH scale that we are familiar with today was developed by Soren Sorensen in 1909. pH is
defined as the negative logarithm of the concentration of the hydrogen ions in moles per liter. The values
of pH ranges between 0 and 14. An aqueous solution is acidic if the pH < 7, while at pH > 7 it is basic or
alkaline. PH 7 for water is known to be neutral at room temperatures. Acid solutions have a greater
concentration of hydronium ions than the hydroxide ion, while basic solutions have a greater
concentration of the hydroxide ions than the hydronium ions.
The pH of a solution is measured using a pH meter or a calorimetric indicator. The pH meter is
based on the concept that the voltage of an electric current passing through a solution changes according
to the pH of the solution. A colorimetric indicator is a chemical dye which changes color at certain
hydrogen ion concentrations.
http://jchemed.chem.wisc.edu http://catalog.miniscience.com www.bbc.co.uk
_________________________________________________________________________________________________Activity 2
B. Buffers
Buffers protect the living system, from large changes in pH when an acid or a base is added to
the system. The pH must be regulated because the enzymes that catalyze metabolic reactions work only
within a narrow pH range. An abrupt change in the pH may slow down or stop the action of enzymes. A
buffer solution consists of a weak acid and its conjugate base that resist change in pH upon addition of
small amounts either acid or base.
Standard buffer solutions are prepared from weak acids and the salts of weak acids. The pH of a
buffer can be determined using the following formula:
Since HA H+ + A –
Then Ka =
H A
= H
A
HA HA
We can take the logarithm of both sides pKa = pH – log
A
HA
Thus, pH = pKa + log
A
HA
OBJECTIVES
1. To be able to measure the pH of sample solutions using the pH paper.
2. To differentiate changes in pH in unbuffered and buffered solutions.
MATERIALS
pH paper, wash bottle, stirring rod, petri dish (2), 25 ml graduated cylinder (1), 25 mL pipette (2),
aspirator, 400 ml beaker (1) , 100 ml beaker(3), pH meter, dropper (2)
REAGENTS
aspirin tablet, baking soda, milk of magnesia, urine (infant’s and adult’s), vinegar, albumin, liquid
detergent, 0.1M NaCl, 0.1M H3PO4, 0.1M H2CO3, 0.1M NaC2H3O2, distilled water 1.0M HCl, 1.0M
NaOH, 0.1M HC2H3O2
PROCEDURE
I. pH measurement of common acids, bases and salts
1. Place about 25 mL of the solutions listed in the table in separate, clean, dry beakers.
2. Measure the pH of each solution using a pH meter. Record the pH of each solution in the data
table.
II. pH measurement of common household chemicals
1. Place a half cm length of pH paper strips on a petri dish.
2. Wet each of the pH paper with a solution listed in the table using a stirring rod. Be sure you wash
with distilled water and dry the stirring rod before using in the next solution.
3. Record the pH of each of the solutions listed in the table by comparing the color with the color
chart in the pH paper container.
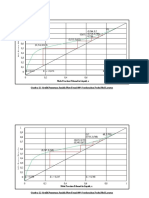
III. Buffer action
A. Preparation of buffer solution and addition of acid
1. Pipet 25.00 mL of 0.1 M sodium acetate, NaC2H3O2, into a clean 100 mL beaker. Pipet 25.00
mL of 0.1 M acetic acid, H2C2H3O2, into the same beaker and mix the solutions. A buffer solution
have just been prepared.
2. Measure the pH of this buffer and record the value in the data table.
_________________________________________________________________________________________________Activity 2
3. Add 5 drops of 1.0 M HCl to the buffer one drop at a time. Swirl the solution after each addition.
Measure the pH after each drop is added, and record the pH on the data table. Wait for several
seconds for the displayed pH value to stabilize.
4. Before proceeding to step B, add 5 drops of 1.0 M NaOH to the buffer solution to compensate for
the 5 drops of 1.0 M HCl added before. Stir the solution well and measure its pH. It should be
the same as that of the original buffer solution.
B. Addition of base to a buffer solution
5. Add 5 drops of 1.0 M NaOH to the buffer solution one drop at a time. Swirl the solution after
each addition. Measure the pH after each drop is added and record the pH on the data table.
C. Addition of acid and base to unbuffered solution
6. Place about 25 mL distilled water into a clean 100 mL beaker. Measure the pH of the water and
record in the data table. Add 5 drops of 1.0 M HCl one drop at a time. Swirl the solution after
each addition. Measure the pH after each drop is added and record the pH on the data table.
7. Place about 25 mL distilled water into another clean 100 mL beaker. Measure the pH of the
water and record in the data table. Add 5 drops of 1.0 M NaOH one drop at a time. Swirl the
solution after each addition. Measure the pH after each drop is added and record the pH on the
data table.
S-ar putea să vă placă și
- Expt.1 BiochemDocument4 paginiExpt.1 BiochemMc de RamosÎncă nu există evaluări
- Alkaline Diet for Beginners: Understand PH, Eat Well and Reclaim Your Health with Easy Alkaline Diet Recipes.De la EverandAlkaline Diet for Beginners: Understand PH, Eat Well and Reclaim Your Health with Easy Alkaline Diet Recipes.Încă nu există evaluări
- Name: Castil, Joyce B. Date Submitted: January 15, 2021 Lab Schedule: TTH, 8:00 - 11:00 RatingDocument31 paginiName: Castil, Joyce B. Date Submitted: January 15, 2021 Lab Schedule: TTH, 8:00 - 11:00 RatingJoyce Castil (Joyceee)Încă nu există evaluări
- Advanced Pharmaceutical analysisDe la EverandAdvanced Pharmaceutical analysisEvaluare: 4.5 din 5 stele4.5/5 (2)
- Lab Experiment 3 Ka Determination Through PH TitrationDocument4 paginiLab Experiment 3 Ka Determination Through PH TitrationxmusiqaÎncă nu există evaluări
- Labexercise 2Document7 paginiLabexercise 2Ma Catherine MalanogÎncă nu există evaluări
- Preparing Buffers and Buffer CapacityDocument7 paginiPreparing Buffers and Buffer CapacityAndreaKatovic100% (1)
- Pka of Salicylic AcidDocument5 paginiPka of Salicylic Acidorganic Aau pharmacyÎncă nu există evaluări
- Exp 1 Prepare Buffer SolnDocument4 paginiExp 1 Prepare Buffer SolnLaila FaeizahÎncă nu există evaluări
- Chem 18.1 Experiment 6 Formal ReportDocument5 paginiChem 18.1 Experiment 6 Formal Reportlouize_1496Încă nu există evaluări
- AP Chemistry Investigation 4 - Judy, Paul, AnthonyDocument13 paginiAP Chemistry Investigation 4 - Judy, Paul, AnthonyAnthony HowerÎncă nu există evaluări
- PH Measurement and Buffer PreparationDocument6 paginiPH Measurement and Buffer PreparationSheena PasionÎncă nu există evaluări
- Buffer SolutionsV3 PDFDocument4 paginiBuffer SolutionsV3 PDFKarunakarÎncă nu există evaluări
- Activity 1 PH Buffers Revised 6.8.20Document5 paginiActivity 1 PH Buffers Revised 6.8.20princessfarah hussinÎncă nu există evaluări
- PH and Buffer Solutions: Name of Student: Mendoza, Jhunlaine Kate GDocument3 paginiPH and Buffer Solutions: Name of Student: Mendoza, Jhunlaine Kate GKate MendozaÎncă nu există evaluări
- Biological Buffer SystemDocument4 paginiBiological Buffer SystemVern NuquiÎncă nu există evaluări
- Biological Buffer SystemDocument6 paginiBiological Buffer SystemJason Raquin RoqueÎncă nu există evaluări
- Experiment 1 PH and Buffers Pre LabDocument12 paginiExperiment 1 PH and Buffers Pre LabMing mingÎncă nu există evaluări
- Experiment 1 Determination of PHDocument5 paginiExperiment 1 Determination of PHRoselle AbrazaldoÎncă nu există evaluări
- BIOCHEM EXP 1 PPT Final 2 PDFDocument8 paginiBIOCHEM EXP 1 PPT Final 2 PDFEUNICE FIGUEROAÎncă nu există evaluări
- Chem 160.1 Ex2 BufferDocument8 paginiChem 160.1 Ex2 BufferAsi JenÎncă nu există evaluări
- Effects of Buffer On PHDocument5 paginiEffects of Buffer On PHAnge OuedraogoÎncă nu există evaluări
- Chem 131A: PH and Buffers ExperimentDocument4 paginiChem 131A: PH and Buffers ExperimentLinh NguyễnÎncă nu există evaluări
- Chapter OneDocument6 paginiChapter OneArjun KatariaÎncă nu există evaluări
- Lab Report 4Document22 paginiLab Report 4wilhelminaanimÎncă nu există evaluări
- PH and BuffersDocument4 paginiPH and BuffersNirmalya Chatterjee100% (1)
- Buffers: IdentificationDocument4 paginiBuffers: IdentificationfaithÎncă nu există evaluări
- PH BuffersDocument5 paginiPH BuffersReem NasserÎncă nu există evaluări
- 3 - Modified Buffers Biochem Lab 1 With WorksheetDocument4 pagini3 - Modified Buffers Biochem Lab 1 With WorksheetkatherinecalmaÎncă nu există evaluări
- PH Dan Larutan PenyanggaDocument38 paginiPH Dan Larutan PenyanggaSri Novita YandaÎncă nu există evaluări
- PH Measurement and Buffer Preparation (Formal Report)Document5 paginiPH Measurement and Buffer Preparation (Formal Report)Paul Benjomin Agregado50% (4)
- S Determination of Phosphoric Acid Content in SoftdrinksDocument5 paginiS Determination of Phosphoric Acid Content in SoftdrinksMike Anderson0% (1)
- FlinnPREP APChem Lab15 TeacherPrintInstructionsDocument15 paginiFlinnPREP APChem Lab15 TeacherPrintInstructionsGalah NasserÎncă nu există evaluări
- Mcnur 2 Biochem Transes+lab QuizDocument6 paginiMcnur 2 Biochem Transes+lab Quizyanna aÎncă nu există evaluări
- Acids Bases and PHDocument4 paginiAcids Bases and PHsatheeshÎncă nu există evaluări
- Lab. 6 Buffer SolutionsDocument4 paginiLab. 6 Buffer Solutionssultan100% (1)
- Plugin bl302 PkavalueDocument3 paginiPlugin bl302 PkavalueSurya KumarÎncă nu există evaluări
- BCHDocument23 paginiBCHClaudette LopezÎncă nu există evaluări
- Experiment 1Document24 paginiExperiment 1Joanna Liezl AbrinaÎncă nu există evaluări
- CWV 35 COMP Phosphoric - Acid PDFDocument4 paginiCWV 35 COMP Phosphoric - Acid PDFNaveen KumarÎncă nu există evaluări
- Buffer 0Document27 paginiBuffer 0Ahmad FarhanÎncă nu există evaluări
- BuffersDocument9 paginiBuffersrajabbasi572Încă nu există evaluări
- Bio Chem Labbb EditDocument37 paginiBio Chem Labbb EditAlbert Azura100% (1)
- PH Measurement and Buffer PreparationDocument7 paginiPH Measurement and Buffer PreparationPatricia Camryne AmbidaÎncă nu există evaluări
- Buffer ChemistryDocument9 paginiBuffer ChemistrySidra chaudharyÎncă nu există evaluări
- Experiment 1: Preparation and Analysis of Laboratory BuffersDocument16 paginiExperiment 1: Preparation and Analysis of Laboratory BuffersMani ScoopsÎncă nu există evaluări
- PH y CurvasDocument7 paginiPH y CurvasSANTIAGO SANCHEZ ALZATEÎncă nu există evaluări
- Biochem Lab 1Document108 paginiBiochem Lab 1Chem PanganibanÎncă nu există evaluări
- 2512 PH PDF - 4f87e83a9ddeaDocument3 pagini2512 PH PDF - 4f87e83a9ddeachem_dream10Încă nu există evaluări
- 5: PH Measurement and Its Applications (Experiment) : ObjectivesDocument19 pagini5: PH Measurement and Its Applications (Experiment) : ObjectivesNajmi NasirÎncă nu există evaluări
- Buffer 0Document27 paginiBuffer 0Adinda Nur AdilaÎncă nu există evaluări
- Acids & Bases, Titrations & BuffersDocument6 paginiAcids & Bases, Titrations & BuffersAhmed KurdishÎncă nu există evaluări
- Biochemistry LaboratoryDocument13 paginiBiochemistry LaboratoryburgosmjvÎncă nu există evaluări
- Physical Chemistry Sem 2 Exp 1Document9 paginiPhysical Chemistry Sem 2 Exp 1Hasni Abdul Rahim100% (2)
- Laboratory Report 4.2Document4 paginiLaboratory Report 4.2Robelyn CastilloÎncă nu există evaluări
- Exp CHM 1Document8 paginiExp CHM 1qhusnaÎncă nu există evaluări
- Potentiometric Titration Ex17Document10 paginiPotentiometric Titration Ex17Tien HaminhÎncă nu există evaluări
- Preparation of BuffersDocument4 paginiPreparation of Bufferskleyr100% (1)
- BufferDocument6 paginiBufferGladys CastilloÎncă nu există evaluări
- Prelab Activity MPLDocument5 paginiPrelab Activity MPLAlthea Valenzuela100% (1)
- A. Philosophical PerspectiveDocument29 paginiA. Philosophical PerspectiveAlthea ValenzuelaÎncă nu există evaluări
- PLR3 and Procedure For Activity 3 MicparaDocument5 paginiPLR3 and Procedure For Activity 3 MicparaAlthea ValenzuelaÎncă nu există evaluări
- VMVM VDocument2 paginiVMVM VAlthea ValenzuelaÎncă nu există evaluări
- Endocrine System PointersDocument2 paginiEndocrine System PointersAlthea ValenzuelaÎncă nu există evaluări
- DNA Structure and Properties: Laboratory ExerciseDocument4 paginiDNA Structure and Properties: Laboratory ExerciseAlthea ValenzuelaÎncă nu există evaluări
- Module2 NSTP Law 19 20 Final FinalDocument13 paginiModule2 NSTP Law 19 20 Final FinalAlthea ValenzuelaÎncă nu există evaluări
- De La Salle Lipa: Entrepreneurial Behavior Research PaperDocument3 paginiDe La Salle Lipa: Entrepreneurial Behavior Research PaperAlthea ValenzuelaÎncă nu există evaluări
- Generation Z AND Millenials: Section1:Who?Document1 paginăGeneration Z AND Millenials: Section1:Who?Althea ValenzuelaÎncă nu există evaluări
- Name: BAGUI, Andrei Mikhael M. Section: K1ADocument1 paginăName: BAGUI, Andrei Mikhael M. Section: K1AAlthea ValenzuelaÎncă nu există evaluări
- The First Mass in The Philippines OUTLINEDocument2 paginiThe First Mass in The Philippines OUTLINEAlthea Valenzuela100% (7)
- 1920 Module 6 DRRMDocument11 pagini1920 Module 6 DRRMAlthea ValenzuelaÎncă nu există evaluări
- Module # 7:: School Year 2019-2020Document13 paginiModule # 7:: School Year 2019-2020Althea ValenzuelaÎncă nu există evaluări
- Family Background: de La Salle's Mother Nicolle Moët de BrouilletDocument1 paginăFamily Background: de La Salle's Mother Nicolle Moët de BrouilletAlthea ValenzuelaÎncă nu există evaluări
- 30 Secs 30 Secs 30 SecsDocument2 pagini30 Secs 30 Secs 30 SecsAlthea ValenzuelaÎncă nu există evaluări
- CHM 421 Analytical Chemistry Experiment 4: Solubility of Ionic Salts in SeawaterDocument14 paginiCHM 421 Analytical Chemistry Experiment 4: Solubility of Ionic Salts in SeawaterIntan SapuraÎncă nu există evaluări
- USP-NF Atorvastatin Calcium TabletsDocument9 paginiUSP-NF Atorvastatin Calcium TabletsPhạm Đức LộcÎncă nu există evaluări
- Chapter 4: Imperfections in SolidsDocument46 paginiChapter 4: Imperfections in SolidsJesse De LeonÎncă nu există evaluări
- TutorialDocument40 paginiTutorialShamsMohd100% (1)
- Stabilitas Cahaya 2Document12 paginiStabilitas Cahaya 2AdnanÎncă nu există evaluări
- Lab Report 5 PH AnalysisDocument2 paginiLab Report 5 PH Analysiswriter topÎncă nu există evaluări
- Acid Base InvestigationDocument3 paginiAcid Base InvestigationJonathan IrwantoÎncă nu există evaluări
- Thin Layer ChromatographyDocument6 paginiThin Layer ChromatographyVictoria ReaganÎncă nu există evaluări
- Mass SpectrometryDocument30 paginiMass SpectrometryGanesh V GaonkarÎncă nu există evaluări
- Chemistry Homework #2Document2 paginiChemistry Homework #2Own AbbadiÎncă nu există evaluări
- Total Dissolved Solids ProcedureDocument13 paginiTotal Dissolved Solids Procedurehemavathi jayÎncă nu există evaluări
- Data SheetDocument2 paginiData SheetJussier VitorianoÎncă nu există evaluări
- Instrument Assignment 2Document17 paginiInstrument Assignment 2firaol mokonnenÎncă nu există evaluări
- Cht314 Org3 Report ChromatographyDocument38 paginiCht314 Org3 Report ChromatographyBryan bayÎncă nu există evaluări
- Determination of Purity and Acid Dissociation Constant of Potassium Hydrogen Phthalate by Potentiometric TitrationDocument2 paginiDetermination of Purity and Acid Dissociation Constant of Potassium Hydrogen Phthalate by Potentiometric TitrationEXO SVTÎncă nu există evaluări
- Como Funciona ISO GridDocument1 paginăComo Funciona ISO Gridapi-3697331Încă nu există evaluări
- Lecture 15Document33 paginiLecture 15Anas Nasir officalÎncă nu există evaluări
- MCQ - Without Solve-1 (1) - Rotated - RemovedDocument8 paginiMCQ - Without Solve-1 (1) - Rotated - RemovedH.M. SadiÎncă nu există evaluări
- Grafik Bab IDocument3 paginiGrafik Bab IPulbiÎncă nu există evaluări
- Symmetry & Crystal StructuresDocument37 paginiSymmetry & Crystal StructuresNurnajihah Sukimi JiehaÎncă nu există evaluări
- Preparation and Dilution of Solutions: BCH 312 ExperimentDocument12 paginiPreparation and Dilution of Solutions: BCH 312 ExperimentyohannesÎncă nu există evaluări
- Analytical ErrorsDocument39 paginiAnalytical ErrorsSrikrishnaÎncă nu există evaluări
- Técnicas HistoquímicasDocument202 paginiTécnicas HistoquímicasLaura B.Încă nu există evaluări
- Lab Relative WK Acid Strengths Inq Student HandoutDocument3 paginiLab Relative WK Acid Strengths Inq Student HandoutMuhammad HamidÎncă nu există evaluări
- Soxhlet ExtractionDocument1 paginăSoxhlet ExtractionBernadeth VigoÎncă nu există evaluări
- SeaFAST Offline Trace Metals in SeawaterDocument4 paginiSeaFAST Offline Trace Metals in SeawaterJustina Monge ZorrillaÎncă nu există evaluări
- Determining Ni With DimethylglyoximeDocument5 paginiDetermining Ni With Dimethylglyoxime아미르Încă nu există evaluări
- Quetiapine FumarateDocument9 paginiQuetiapine FumarateVinaya SnehalathaÎncă nu există evaluări
- DOC316.53.01173 8ed PDFDocument8 paginiDOC316.53.01173 8ed PDFcarlososorio_184664Încă nu există evaluări
- USP-NF Acebutolol Hydrochloride CapsulesDocument4 paginiUSP-NF Acebutolol Hydrochloride CapsulesStalin VacaÎncă nu există evaluări
- Process Plant Equipment: Operation, Control, and ReliabilityDe la EverandProcess Plant Equipment: Operation, Control, and ReliabilityEvaluare: 5 din 5 stele5/5 (1)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDe la EverandChemistry for Breakfast: The Amazing Science of Everyday LifeEvaluare: 4.5 din 5 stele4.5/5 (14)
- ICH Quality Guidelines: An Implementation GuideDe la EverandICH Quality Guidelines: An Implementation GuideAndrew TeasdaleÎncă nu există evaluări
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincDe la EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincEvaluare: 3.5 din 5 stele3.5/5 (137)
- AP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeDe la EverandAP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeÎncă nu există evaluări
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDe la EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactEvaluare: 5 din 5 stele5/5 (5)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsDe la EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsEvaluare: 5 din 5 stele5/5 (3)
- It's Elemental: The Hidden Chemistry in EverythingDe la EverandIt's Elemental: The Hidden Chemistry in EverythingEvaluare: 4 din 5 stele4/5 (10)
- AP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeDe la EverandAP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeEvaluare: 5 din 5 stele5/5 (1)
- Well Control for Completions and InterventionsDe la EverandWell Control for Completions and InterventionsEvaluare: 4 din 5 stele4/5 (10)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDe la EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactEvaluare: 5 din 5 stele5/5 (1)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeDe la EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeEvaluare: 4 din 5 stele4/5 (1)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeDe la EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeEvaluare: 5 din 5 stele5/5 (4)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideDe la EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideÎncă nu există evaluări
- Guidelines for Defining Process Safety Competency RequirementsDe la EverandGuidelines for Defining Process Safety Competency RequirementsEvaluare: 3 din 5 stele3/5 (1)
- The Production of Volatile Oils and Perfumery Plants in the United StatesDe la EverandThe Production of Volatile Oils and Perfumery Plants in the United StatesÎncă nu există evaluări
- Formulating, Packaging, and Marketing of Natural Cosmetic ProductsDe la EverandFormulating, Packaging, and Marketing of Natural Cosmetic ProductsÎncă nu există evaluări
- Piping Engineering Leadership for Process Plant ProjectsDe la EverandPiping Engineering Leadership for Process Plant ProjectsEvaluare: 5 din 5 stele5/5 (1)
- Guidelines for Chemical Process Quantitative Risk AnalysisDe la EverandGuidelines for Chemical Process Quantitative Risk AnalysisEvaluare: 5 din 5 stele5/5 (1)
- Sodium Bicarbonate: Nature's Unique First Aid RemedyDe la EverandSodium Bicarbonate: Nature's Unique First Aid RemedyEvaluare: 5 din 5 stele5/5 (21)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDe la EverandChemistry for Breakfast: The Amazing Science of Everyday LifeEvaluare: 4.5 din 5 stele4.5/5 (90)
- Taste: Surprising Stories and Science About Why Food Tastes GoodDe la EverandTaste: Surprising Stories and Science About Why Food Tastes GoodEvaluare: 3 din 5 stele3/5 (20)