Documente Academic
Documente Profesional
Documente Cultură
Lecture 17
Încărcat de
Melo Courabies MacAronoDescriere originală:
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Lecture 17
Încărcat de
Melo Courabies MacAronoDrepturi de autor:
Formate disponibile
EP225 2009 Lecture 17
Energy in Standing Waves
Standing waves are formed by sisuoidal waves propagating in opposite directions and
thus no enenrgy is carried. Rather, energy is conned between nodes (zero displacement
points). We consider a standing wave
:
0
sin (/r) cos (.t) . 0 < r < 1
in a string of length 1. mass desnity j
l
(kg/m) and tension 1 (N). The wave lengths allowed
are \
n
= :12 and /
n
= 4::1 where : is integer. The wave velocity is
c
w
=
s
1
j
l
m/s
The kinetic energy density is
KED =
1
2
j
l
:
t
2
=
1
2
j
l
.
2
:
2
0
sin
2
(/r) sin
2
(.t) J/m
Its spatial average is
1
2
peak =
1
4
j
l
.
2
:
2
0
sin
2
(.t)
and the total kinetic energy is
KE =
1
4
1j
l
.
2
:
2
0
sin
2
(.t)
The potential energy density is
PED =
1
2
1
:
r
2
(N = J/m)
=
1
2
1/
2
:
2
0
cos
2
(/r) cos
2
(.t)
=
1
2
j
l
.
2
:
2
0
cos
2
(/r) cos
2
(.t)
The spatial average is
1
4
j
l
.
2
:
2
0
cos
2
(.t)
and the total potential energy is
PE =
1
4
1j
l
.
2
:
2
0
cos
2
(.t)
The total energy is
1
4
1j
l
.
2
:
2
0
sin
2
(.t) +
1
4
1j
l
.
2
:
2
0
cos
2
(.t) =
1
4
1j
l
.
2
:
2
0
(J)
Beat
1
When two sinusoidla signals with slightly dierent fequencies 1 and 1 + 1 are super-
posed, the amlitude is modulated and a beat with a frequency 1 is detected.
sin(2:1t) + sin [2: (1 + 1) t]
= 2sin
2:
1 + 1
2
t
cos
2:
1
2
t
' 2sin(2:1t) cos
2:
1
2
t
The signal intensity is
4
2
sin
2
(2:1t) cos
2
2:
1
2
t
= 2
2
sin
2
(2:1t) [1 + cos (2:1t)]
where use is made of
cos
2
r =
1 + cos 2r
2
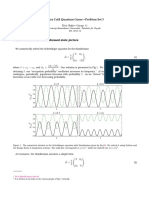
The intensity is modulated at the frequncy dierence 1 . The gure below shows addition
of waves with frequencies 10 Hz and 11 Hz.
sin (2: 10t)
0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0
-1
0
1
x
y
sin (2: 11t)
0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0
-1
0
1
x
y
sin (2: 10t) + sin (2: 11t)
0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0
-2
-1
0
1
2
t
y
2
Phase and Group Velocities
Super position of two waves having slightly dierent frequencies and wavenumbers
sin (/r .t) + sin [(/ + /) r (. + .) t]
' 2(/r .t) cos
/
2
r
.
2
t
Fine ripples propagate at
Phase velocity
.
/
The envelope propagates at
Group velocity
.
/
!
d.
d/
In non-dispersive waves, t/c phase and group velocities are the same. Examples are light
wave in vacuum, sound waves. Examples of dispersive waves:
Water wave . =
p
o/.
.
/
=
r
o
/
.
d.
d/
=
1
2
r
o
/
Electromagnetic waves in plasma .
2
= (c/)
2
+ .
2
p
.
/
= c
1
q
1
.
2
p
.
2
c;
d.
d/
= c
q
1
.
2
p
.
2
< c
Sound Waves in a Gas
When a gas is compressed, the density increases. At the same time, the temperature
increases adiabatically because thermal conduction in a gas is slow. The equation of state is
1\
= const.
where is the ratio of specic heats,
=
1 + 2
1
with 1 the number of degree of freedom. For monoatomic gases (He, Ar, etc.) 1 = 3 and
=
5
3
. For diatomic gases, 1 = 5 and =
7
5
.
Equation of state is
d1\
+ 1\
1
= 0
1
1
=
\
\
Then the pressure wave is
j = 1
:
r
and the equation of motion is
j
v
2
:
t
2
=
r
j = 1
2
:
r
2
3
The sound speed is given by
c
s
=
s
1
j
v
=
r
11
`
mol
where 1 = 8.3 J/K is the gas constant and `
mol
is the molar mass of the gas. The molar
mass of air is 0.029 kg. Note that the sound speed is independent of the gas density. At
20
C, the sound speed is
c
s
=
r
1.4 8.3 293
0.029
= 343 m/s
Doppler shift
Doppler eect is apparent change in the frequency (or wavelength) due to the motion of
sound source and observer. If the source is moving, the wavelength changes,
\
0
=
c
s
\
s
c
s
\. 1
0
=
c
s
c
s
\
s
1
Waves in front of the source are squeezed (shorter wavelength ! higher frequency).
If the observer is moving, the sound speed changes from c
s
to c
s
+ \
o
. Then
1
00
=
c
s
+ \
o
c
s
1
4
S-ar putea să vă placă și
- Wulfman Lie Group SymmetriesDocument13 paginiWulfman Lie Group SymmetriesMelo Courabies MacAronoÎncă nu există evaluări
- Simple Cond Matter Problem SetDocument7 paginiSimple Cond Matter Problem SetMelo Courabies MacAronoÎncă nu există evaluări
- Solutions To Abstract Algebra - Chapter 1 (Dummit and Foote, 3e)Document25 paginiSolutions To Abstract Algebra - Chapter 1 (Dummit and Foote, 3e)Jason Rosendale82% (17)
- Gauge Invariance and Dof CountDocument20 paginiGauge Invariance and Dof CountMelo Courabies MacAronoÎncă nu există evaluări
- A Note On The Post-Newtonian Limit of Quasi-Local Energy ExpressionsDocument12 paginiA Note On The Post-Newtonian Limit of Quasi-Local Energy ExpressionsMelo Courabies MacAronoÎncă nu există evaluări
- An Introduction To Free Higher Spins PDFDocument25 paginiAn Introduction To Free Higher Spins PDFMelo Courabies MacAronoÎncă nu există evaluări
- A Question of MassDocument7 paginiA Question of MassMelo Courabies MacAronoÎncă nu există evaluări
- Black Holes - Paul K. TownsendDocument145 paginiBlack Holes - Paul K. TownsendtttottoÎncă nu există evaluări
- An Introduction To Black HolesDocument53 paginiAn Introduction To Black HolesMelo Courabies MacAronoÎncă nu există evaluări
- Shoe Dog: A Memoir by the Creator of NikeDe la EverandShoe Dog: A Memoir by the Creator of NikeEvaluare: 4.5 din 5 stele4.5/5 (537)
- Grit: The Power of Passion and PerseveranceDe la EverandGrit: The Power of Passion and PerseveranceEvaluare: 4 din 5 stele4/5 (587)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDe la EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceEvaluare: 4 din 5 stele4/5 (894)
- The Yellow House: A Memoir (2019 National Book Award Winner)De la EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Evaluare: 4 din 5 stele4/5 (98)
- The Little Book of Hygge: Danish Secrets to Happy LivingDe la EverandThe Little Book of Hygge: Danish Secrets to Happy LivingEvaluare: 3.5 din 5 stele3.5/5 (399)
- On Fire: The (Burning) Case for a Green New DealDe la EverandOn Fire: The (Burning) Case for a Green New DealEvaluare: 4 din 5 stele4/5 (73)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDe la EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeEvaluare: 4 din 5 stele4/5 (5794)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDe la EverandNever Split the Difference: Negotiating As If Your Life Depended On ItEvaluare: 4.5 din 5 stele4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDe la EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureEvaluare: 4.5 din 5 stele4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDe la EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryEvaluare: 3.5 din 5 stele3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerDe la EverandThe Emperor of All Maladies: A Biography of CancerEvaluare: 4.5 din 5 stele4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDe la EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreEvaluare: 4 din 5 stele4/5 (1090)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDe la EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyEvaluare: 3.5 din 5 stele3.5/5 (2219)
- Team of Rivals: The Political Genius of Abraham LincolnDe la EverandTeam of Rivals: The Political Genius of Abraham LincolnEvaluare: 4.5 din 5 stele4.5/5 (234)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDe la EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersEvaluare: 4.5 din 5 stele4.5/5 (344)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDe la EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaEvaluare: 4.5 din 5 stele4.5/5 (265)
- The Unwinding: An Inner History of the New AmericaDe la EverandThe Unwinding: An Inner History of the New AmericaEvaluare: 4 din 5 stele4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)De la EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Evaluare: 4.5 din 5 stele4.5/5 (119)
- Her Body and Other Parties: StoriesDe la EverandHer Body and Other Parties: StoriesEvaluare: 4 din 5 stele4/5 (821)
- Wave MCQDocument24 paginiWave MCQMohammed El-kattanÎncă nu există evaluări
- Time and Frequency Users ManualDocument273 paginiTime and Frequency Users ManualSavuDanielFÎncă nu există evaluări
- Norma Is - Iso.7919.5.2005 PDFDocument22 paginiNorma Is - Iso.7919.5.2005 PDFRafael García100% (1)
- Module 2 Measurement of Horizontal DistancesDocument16 paginiModule 2 Measurement of Horizontal DistancesSkyÎncă nu există evaluări
- Using Spooky2 As A Frequency MeterDocument1 paginăUsing Spooky2 As A Frequency MeterYoko SumartoÎncă nu există evaluări
- A Primer For Vibration IsolationDocument9 paginiA Primer For Vibration IsolationFadooollÎncă nu există evaluări
- A Practical Handbook of Speech CodersDocument13 paginiA Practical Handbook of Speech CodersVany BraunÎncă nu există evaluări
- Vibration Diagnostic Guide 2Document2 paginiVibration Diagnostic Guide 2Praveen SreedharanÎncă nu există evaluări
- CH1Document34 paginiCH1none5402Încă nu există evaluări
- Wave PDFDocument10 paginiWave PDFAswin RangkutiÎncă nu există evaluări
- Natural Frequency and Resonance ExperimentsDocument4 paginiNatural Frequency and Resonance ExperimentsPRASANTH G KRISHNANÎncă nu există evaluări
- Acoustic GratingDocument1 paginăAcoustic GratingShwetha PriyadharshiniÎncă nu există evaluări
- LEP1101 - 00 Measurement of Basic Constants Length, Weight and Time PDFDocument5 paginiLEP1101 - 00 Measurement of Basic Constants Length, Weight and Time PDFJose GalvanÎncă nu există evaluări
- Basic Administration ProceduresDocument252 paginiBasic Administration Proceduresaxleugenio100% (1)
- UT Level 1 Trial QuestionsDocument6 paginiUT Level 1 Trial QuestionsKhepa BabaÎncă nu există evaluări
- White Paper Understanding Data Averaging Modes en 5545142 PDFDocument3 paginiWhite Paper Understanding Data Averaging Modes en 5545142 PDFMuhammad Fahmi MahmudÎncă nu există evaluări
- Holzland Stoellger GesamtpreislisteDocument11 paginiHolzland Stoellger GesamtpreislisteBerndÎncă nu există evaluări
- And Application Reliable Ultrasonic Atomizer: DesignDocument5 paginiAnd Application Reliable Ultrasonic Atomizer: DesignSteve WanÎncă nu există evaluări
- Bmom&Yftajcch Ar Cgef WDKRSM : Chapter 1: Work Done and Power DfnsDocument62 paginiBmom&Yftajcch Ar Cgef WDKRSM : Chapter 1: Work Done and Power DfnsFrock Johnson100% (1)
- Noise source identification techniques: simple to advanced applicationsDocument6 paginiNoise source identification techniques: simple to advanced applicationscowokÎncă nu există evaluări
- Hy Series InverterDocument60 paginiHy Series Invertersea13Încă nu există evaluări
- Acoustic Simulation with Actran: Acoustic Field Maps and Transmission LossDocument45 paginiAcoustic Simulation with Actran: Acoustic Field Maps and Transmission Lossvivashwanth paiÎncă nu există evaluări
- Wave GuideDocument16 paginiWave Guideefren6716Încă nu există evaluări
- Analysis of Automated Flow Line Line BalancingDocument23 paginiAnalysis of Automated Flow Line Line BalancingMuzammil MuhammedÎncă nu există evaluări
- Subliminal Mind Control Hemi Sync Basics What It Does and How Amp Why It WorksDocument5 paginiSubliminal Mind Control Hemi Sync Basics What It Does and How Amp Why It Worksdruidess3Încă nu există evaluări
- Catalouge Clarion 2008Document8 paginiCatalouge Clarion 2008Andrew BondtskovichÎncă nu există evaluări
- Priminary of Spar Truss PlatformDocument15 paginiPriminary of Spar Truss PlatformCROSSFIELD ENGINEERING ENERGYÎncă nu există evaluări
- COMS2Document490 paginiCOMS2Mora JoramÎncă nu există evaluări
- Configure F-1000 Turbine Flow MeterDocument37 paginiConfigure F-1000 Turbine Flow MeterĐăng LêÎncă nu există evaluări
- Agilent 8902A Measuring Receiver: Product NoteDocument8 paginiAgilent 8902A Measuring Receiver: Product Notejcfermosell100% (1)