Documente Academic
Documente Profesional
Documente Cultură
Handout Chapter 5 Iron Carbon System
Încărcat de
Bikram MuduliDescriere originală:
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
Handout Chapter 5 Iron Carbon System
Încărcat de
Bikram MuduliDrepturi de autor:
Formate disponibile
MSM: Chapter 5, Iron Carbon Systems
Chapter 5: Iron Carbon Systems
Steels and cast irons are among the most extensively used range of engineering materials in the world today. They are essentially iron-carbon alloys and hence of all binary alloys systems, the one that is most important is the Iron-Carbon.
Cooling curve for pure iron:
Liquid 1538 Fe (BCC)
Temperature, oC
1394
Fe (FCC) Non-Magnetic
912 770
Fe (BCC) Non-Magnetic Fe (BCC) Magnetic
Time Fig 5.1 Cooling curve for pure iron
Iron in the pure form, usually known as ingot-iron, basically consists of: Carbon 0.012% Manganese Sulphur 0.017% 0.025%
Phosphorous 0.005% Silicon traces
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 1 of 7
MSM: Chapter 5, Iron Carbon Systems
Typical mechanical properties of Ingot-Iron: Tensile strength Elongation in 2 inches Rockwell B hardness 40000 psi (275 MPa) 40% 30
Iron is allotropic metal, which means that it can exist in more than one type of lattice structure depending upon temperature. A study of cooling curve of pure iron shows that it can exist in more than one allotropic form. When iron solidifies first, at 1538oC, it is in the BCC form called Delta Iron. Further cooling at 1394oC, a phase change occurs and atoms re-arrange themselves into FCC form called as Gamma Iron, which is non-magnetic. When the temperature reaches 912oC, another phase change occurs and atoms begin to re-arrange themselves into form called as Alpha Iron, which is non-magnetic. Finally at 768oC, the -iron becomes magnetic
without a change in the crystal lattice structure. Initially, non-magnetic -iron was called -iron until subsequent x-rays studies showed no change in lattice structure at 768oC. This magnetic transformation does not change the heat treatment of iron-carbon alloys. Hence it will be neglected in the study. All the allotropic changes give off heat (exothermic) when iron is cooled and absorb heat (endothermic) when iron is heated.
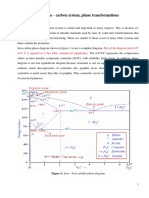
The Iron-Carbon Equilibrium Diagram: The left vertical axis represents 100% iron and shows all the allotropic changes of pure iron with temperature. The temperature at which allotropic changes occur in iron changes when it is alloyed with different amounts of carbon.
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 2 of 7
MSM: Chapter 5, Iron Carbon Systems
1600 1538A 1500 1400 1394 N 1300
+ L B 1495oC
+ L Fe3C + L
Temperature, o C
1200 C 1100 1000 912 900 800 + 700 H 600 500 400 300 1148oC 2%
E 4.33%
+ Fe3C
J 0.8 %
727oC
+ Fe3C
3 Carbon % (by weight)
6.67
STEELS
CAST IRONS Hypo Eutectic Hyper Eutectic
Hypo Eutectoid Steels
Hyper Eutectoid
Cast Irons
Cast Irons
Fig Steels 5.2 Iron Iron Carbide Equilibrium Diagram
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 3 of 7
MSM: Chapter 5, Iron Carbon Systems
The right vertical axis does not represent 100% carbon but instead represents only 6.67% C by weight. This is because only a maximum of 6.67% C can be added to molten iron at which it becomes saturated. Any further addition of carbon will not dissolve in iron but rather floats or gets blown away owing to its very low density. Iron when contains exactly 6.67% C by weight forms an intermediate phase called Cementite or Iron Carbide (Fe3C). Hence, the Iron-Carbon equilibrium diagram is actually called as the Iron Iron Carbide Equilibrium Diagram with pure iron and pure Fe3C (Iron Carbide) forming the extremities. Actually, the phase Fe3C (Iron Carbide) is called meta stable state because it decomposes with passage of time. Here, the equilibrium phase diagram for iron and carbon assumes that Fe3C is stable with respect to time. Hence, it is not a true equilibrium diagram. The diagram shows 3 horizontal lines (isotherms) which represent 3 invariant reactions. (a) The first isotherm at 1493oC indicates the peritectic reaction. This region where the peritectic reaction takes place is called the Delta region. (b) The second isotherm at 1148oC indicates the eutectic reaction. This region where the eutectic reaction takes place is called the Eutectic region. (c) The third isotherm at 727oC indicates the eutectoid reaction. This region where the eutectoid reaction takes place is called the Eutectoid region. The invariant reactions in the Fe-Fe3C diagram:
(a) The Delta Region: The first horizontal line MB at 1495oC indicates the Peritectic reaction. This region is called as the delta region because of the solid solution . The point P is known as the peritectic point at 1495oC & 0.18%C.
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 4 of 7
MSM: Chapter 5, Iron Carbon Systems
The peritectic reaction may be written as:
1538oC cooling
A L+ M P
0.5% C
Liquid +
heatin g
(Austenite)
1495oC
B L+
The maximum solubility of carbon in bcc Fe is 0.1% (point M) while in fcc Fe, the solubility is much greater.
1395oC N
Fig. 5.3 The Delta region
0.18% C 0.1% C
The presence of carbon influences the allotropic change. As carbon is added to iron, the allotropic change increases from 1394oC to 1495oC at 0.1%C. On cooling, the portion NM represents the beginning of the crystal structure change from bcc Fe to fcc Fe for alloys containing less than 0.1%C. The portion MP represents the beginning of this crystal structure change by means of a peritectic reaction for alloys between 0.1 and 0.18% C. For alloys containing less than 0.18% C, on cooling, the end of the crystal structure change is given by the line NP. The portion PB represents the beginning and ending of the crystal structure change by means of peritectic reaction. That is, for alloys between 0.18%C and 0.5% C, the allotropic change begins and ends at constant temperature. For alloys containing more than 0.5%C, the liquid will solidify to austenite ( ) directly, i.e., there is no allotropic change.
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 5 of 7
MSM: Chapter 5, Iron Carbon Systems
(b) The Eutectic Region:
L+ 2%C 1148oC C E 4.33%C
L + Fe3C D
+ Fe3C Fig. 5.4 The Eutectic Region
The Eutectic reaction takes place at 1148oC. The Eutectic point E is at 1148oC and at 4.33% C. whenever an alloy crosses the line line CED, and Eutectic reaction takes place, giving rise to a fine mixture of Austenite + Cementite ( + Fe3C). This eutectic mixture is commonly called a Ledeburite. This reaction can be expressed as:
Liquid (L)
cooling heatin g
Austenite ( ) + Cementite (Fe3C)
Eutectic mixture (Ledeburite)
Since austenite ( ) is not stable at room temperature, its microstructure cannot be seen for slow cooling. However, it has to under go another reaction (Eutectoid reaction).
(c) The Eutectoid Reaction: A small solid solution region to the left of the line GH, which represents -Fe (Ferrite), is a solid solution with small amounts of carbon dissolved in BCC Fe. At 912oC, the FCC -Fe changes to BCC -Fe. Line HJK represents the Eutectoid reaction taking place at 727oC. The Eutectoid point is J which has 0.8%C & 727oC.
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 6 of 7
MSM: Chapter 5, Iron Carbon Systems
Whenever an alloy crosses the line HJK, the Eutectoid reaction takes place giving rise to the fine Eutectoid mixture of Ferrite + Cementite ( + Fe3C) commonly known as Pearlite. This reaction can be expressed as:
0.8% C +Fe3 C 912oC + H
+Fe3 C J 727oC K
Fig. 5.5 The Eutectoid Region
Austenite ( )
cooling heatin g
Ferrite ( ) + Cementite (Fe3C)
Eutectoid mixture (Pearlite)
Vijay G. S., Faculty, Dept. of Mech. & Mfg., MIT, Manipal
Page 7 of 7
S-ar putea să vă placă și
- Continuous casting The Ultimate Step-By-Step GuideDe la EverandContinuous casting The Ultimate Step-By-Step GuideÎncă nu există evaluări
- Fundamentals of Electrocatalyst Materials and Interfacial Characterization: Energy Producing Devices and Environmental ProtectionDe la EverandFundamentals of Electrocatalyst Materials and Interfacial Characterization: Energy Producing Devices and Environmental ProtectionÎncă nu există evaluări
- Iron Carbon Phase DiagramDocument4 paginiIron Carbon Phase DiagramMizanur RahmanÎncă nu există evaluări
- Foundations of Materials Science and Engineering 5th Edition Smith Solutions ManualDocument79 paginiFoundations of Materials Science and Engineering 5th Edition Smith Solutions Manualdextrermachete4amgqgÎncă nu există evaluări
- Iron Carbon DiagramDocument10 paginiIron Carbon DiagramsivakumarÎncă nu există evaluări
- Lec3-Stir Casting& Squeeze CastingDocument16 paginiLec3-Stir Casting& Squeeze Castingguru prasad50% (2)
- Physical Metallurgy-18 Heat Treatment of SteelDocument7 paginiPhysical Metallurgy-18 Heat Treatment of SteelDSGÎncă nu există evaluări
- Solid State WeldingDocument61 paginiSolid State Weldingakramakram123Încă nu există evaluări
- Presented By, Uma .B. S Ii Sem Mtech (1MS11MSC013) Department of Mechanical EngineeringDocument18 paginiPresented By, Uma .B. S Ii Sem Mtech (1MS11MSC013) Department of Mechanical EngineeringJyoti Sharma100% (1)
- The Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalDocument38 paginiThe Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalYogesh KumbharÎncă nu există evaluări
- Mf5103 Advances in Casting and Welding: For Syllabus, Question Papers, Notes & Many MoreDocument2 paginiMf5103 Advances in Casting and Welding: For Syllabus, Question Papers, Notes & Many MoreNandha Gopal SenthilnathanÎncă nu există evaluări
- ME 292 - Metallic Materials SessionalDocument39 paginiME 292 - Metallic Materials SessionalMuhammedNayeemÎncă nu există evaluări
- Effect of Basic Chemical Element in Sgi Ductile Iron IJERTV1IS7135Document7 paginiEffect of Basic Chemical Element in Sgi Ductile Iron IJERTV1IS7135Uma KoduriÎncă nu există evaluări
- Meen 360 Cheat SheetDocument1 paginăMeen 360 Cheat SheetSaniya SohailÎncă nu există evaluări
- Solidification, Phase Diagrams and Phase TransformationDocument35 paginiSolidification, Phase Diagrams and Phase TransformationkrishnasaiÎncă nu există evaluări
- Welding of 5083 Aluminum AlloyDocument6 paginiWelding of 5083 Aluminum AlloyMario MirićÎncă nu există evaluări
- wk7 (3) - Fe-C SystemDocument12 paginiwk7 (3) - Fe-C Systemsaeed khaledÎncă nu există evaluări
- Surface EngineeringDocument43 paginiSurface Engineering10rajÎncă nu există evaluări
- Engineering Alloys (Non Ferrous)Document52 paginiEngineering Alloys (Non Ferrous)Sukhwinder Singh GillÎncă nu există evaluări
- Tugas 5 Weld Metal Solidification & MicrostructureDocument16 paginiTugas 5 Weld Metal Solidification & MicrostructureDeepakÎncă nu există evaluări
- Chapter 11 (Foundry Modernization and Mechanization) 2003Document10 paginiChapter 11 (Foundry Modernization and Mechanization) 2003gere33% (3)
- 5-Hot Tear PDFDocument9 pagini5-Hot Tear PDFPeeka Prabhakara RaoÎncă nu există evaluări
- Chap 6 TTT Diagram (New)Document26 paginiChap 6 TTT Diagram (New)eeit_nizamÎncă nu există evaluări
- Deformation Behavior of The Surface Defects of Low Carbon Steel in Wire Rod RollingDocument6 paginiDeformation Behavior of The Surface Defects of Low Carbon Steel in Wire Rod RollingAvinash KumarÎncă nu există evaluări
- 4 Unit - Heat Treatment of SteelsDocument72 pagini4 Unit - Heat Treatment of SteelsAnway WalkeÎncă nu există evaluări
- Lecture 04-CCT and TemperingDocument17 paginiLecture 04-CCT and TemperingRudy Dwi PrasetyoÎncă nu există evaluări
- Practise Question (Calculations) AnswerDocument26 paginiPractise Question (Calculations) AnswerYeo JosephÎncă nu există evaluări
- The TEMPCORE ProcessDocument15 paginiThe TEMPCORE ProcessShanna Lee100% (1)
- 86 - The Formation of Panel Cracks in Steel Ingots - A State-Of-The-Art Review - II. Mid-Face and Off-Corner CracksDocument9 pagini86 - The Formation of Panel Cracks in Steel Ingots - A State-Of-The-Art Review - II. Mid-Face and Off-Corner CracksdzizicÎncă nu există evaluări
- Tempcore ProcessDocument23 paginiTempcore ProcessKumaran 1987Încă nu există evaluări
- Problems 13.1: Difficulties Associated With The Partially Melted ZoneDocument2 paginiProblems 13.1: Difficulties Associated With The Partially Melted ZoneSayyidah FarhanaÎncă nu există evaluări
- Reducing of Line StopaggesDocument39 paginiReducing of Line StopaggesSuvro ChakrabortyÎncă nu există evaluări
- On Plasma Arc WeldingDocument12 paginiOn Plasma Arc WeldingSandeep MauryaÎncă nu există evaluări
- Electrode Converted Into TextDocument14 paginiElectrode Converted Into TextNeha NimbarkÎncă nu există evaluări
- 10 Powder Metalurgy IDocument21 pagini10 Powder Metalurgy IilhamÎncă nu există evaluări
- Universitas Indonesia: Mata Kuliah (Kode) Dosen Pengampu Tugas Nama MahasiswaDocument10 paginiUniversitas Indonesia: Mata Kuliah (Kode) Dosen Pengampu Tugas Nama MahasiswaHari AntoÎncă nu există evaluări
- Bhushan Power and Steel LTDDocument8 paginiBhushan Power and Steel LTDAkshay AgarwalÎncă nu există evaluări
- Tugas 5 Weld Metal Solidification & MicrostructureDocument16 paginiTugas 5 Weld Metal Solidification & MicrostructureDeepakÎncă nu există evaluări
- Ductile Iron ReviewDocument36 paginiDuctile Iron ReviewDenis Yasmin AlineÎncă nu există evaluări
- Evaluation of MIG Welding Process Parameter Using Activated Flux On SS316L by AHP-MOORA MethodDocument13 paginiEvaluation of MIG Welding Process Parameter Using Activated Flux On SS316L by AHP-MOORA MethodAnonymous uwTzeJnoKÎncă nu există evaluări
- Heat Treatment by Quenching - DiagramsDocument20 paginiHeat Treatment by Quenching - Diagramssunilmathew4477Încă nu există evaluări
- Project ReportDocument54 paginiProject ReportRakesh Kumar Munda100% (1)
- Corrosion Resistance of Cerium-Doped Zinc Calcium Phosphate Chemical Conversion Coatings On AZ31 Magnesium AlloyDocument15 paginiCorrosion Resistance of Cerium-Doped Zinc Calcium Phosphate Chemical Conversion Coatings On AZ31 Magnesium AlloyMichael Merino CrispinÎncă nu există evaluări
- An Investigation On The Roll Force and Torque Fluctuations During Hot Strip Rolling ProcessDocument16 paginiAn Investigation On The Roll Force and Torque Fluctuations During Hot Strip Rolling ProcessedpsousaÎncă nu există evaluări
- Martensitic Stainless SteelsDocument8 paginiMartensitic Stainless SteelsAdilmar E. NatãnyÎncă nu există evaluări
- Rr411802 Steel MakingDocument4 paginiRr411802 Steel MakingSrinivasa Rao GÎncă nu există evaluări
- 1993-1.11 The Effect of Pour TimeDocument9 pagini1993-1.11 The Effect of Pour TimeKhin Aung ShweÎncă nu există evaluări
- APM PapersDocument49 paginiAPM Papershosseinidokht86Încă nu există evaluări
- Cast Iron BMEDocument2 paginiCast Iron BMEsahitya karaheÎncă nu există evaluări
- Indian Standard: Specification FOR Foundry Moulding Boxes of Steel Construction (Document13 paginiIndian Standard: Specification FOR Foundry Moulding Boxes of Steel Construction (Muthusamy Arumugam100% (1)
- Welding Chrome-Moly Steel: What You Should Know About T-1 WiresDocument22 paginiWelding Chrome-Moly Steel: What You Should Know About T-1 WiresKentDemeterioÎncă nu există evaluări
- Alloys: I) IntroductionDocument12 paginiAlloys: I) IntroductionNikhil ShelarÎncă nu există evaluări
- O HC HCDocument101 paginiO HC HCIndustrial Infra Jobs100% (1)
- Grains Structure of IngotsDocument6 paginiGrains Structure of Ingotshayder1920Încă nu există evaluări
- Tugas 04 - AndriyansaDocument7 paginiTugas 04 - AndriyansaAndriÎncă nu există evaluări
- Aisi 305Document3 paginiAisi 305Aditya PratapÎncă nu există evaluări
- Ass 1 Mech 6511 Mechanical Shaping of Metals and PlasticsDocument2 paginiAss 1 Mech 6511 Mechanical Shaping of Metals and PlasticsVarinder ThandiÎncă nu există evaluări
- Chapter 6 Phase DiagramsDocument73 paginiChapter 6 Phase DiagramsSaiful AzrieÎncă nu există evaluări
- MSM GTU Study Material E-Notes Unit-5 23112020052908AMDocument14 paginiMSM GTU Study Material E-Notes Unit-5 23112020052908AMVijayÎncă nu există evaluări
- Iron-Iron Carbide Phase Diagram: Effect of Pressure On Allotropy of IronDocument5 paginiIron-Iron Carbide Phase Diagram: Effect of Pressure On Allotropy of IronnareshÎncă nu există evaluări
- Office 2013 KeysDocument1 paginăOffice 2013 KeysBikram MuduliÎncă nu există evaluări
- Mech Eng Handbook ONGCDocument220 paginiMech Eng Handbook ONGCBikram MuduliÎncă nu există evaluări
- Plate Heat ExchangerDocument6 paginiPlate Heat ExchangerBikram MuduliÎncă nu există evaluări
- Plate Heat ExchangerDocument6 paginiPlate Heat ExchangerBikram MuduliÎncă nu există evaluări
- Jigs&FixtureDocument14 paginiJigs&FixtureBikram MuduliÎncă nu există evaluări
- Unconventional Machining ProcessDocument15 paginiUnconventional Machining ProcessShashwatAgÎncă nu există evaluări
- Emerald Walk ThroughDocument192 paginiEmerald Walk ThroughBikram MuduliÎncă nu există evaluări
- Rapid Prototyping AbstractDocument1 paginăRapid Prototyping AbstractBikram Muduli100% (1)
- Kinematics of MachinesDocument227 paginiKinematics of Machinesborchec100% (2)
- Leafgreen WalkthroughDocument139 paginiLeafgreen WalkthroughBikram MuduliÎncă nu există evaluări
- Higher Algebra - Hall & KnightDocument593 paginiHigher Algebra - Hall & KnightRam Gollamudi100% (2)
- Spring Dowel SpiralDocument24 paginiSpring Dowel SpiraldineshkshirsagarÎncă nu există evaluări
- Strength of Material Lab: Abdul Razzak Kalsekar Polytechnic, New PanvelDocument15 paginiStrength of Material Lab: Abdul Razzak Kalsekar Polytechnic, New PanvelAamirqadriÎncă nu există evaluări
- WQT Enrolment Form TWIT-WQ-F1 Rev 1Document1 paginăWQT Enrolment Form TWIT-WQ-F1 Rev 1reezmanÎncă nu există evaluări
- NLMK Strip Product Catalogue WebDocument54 paginiNLMK Strip Product Catalogue WebhdierkeÎncă nu există evaluări
- GVP College of Engineering (A) 2015Document3 paginiGVP College of Engineering (A) 2015Vasanth KumarÎncă nu există evaluări
- Machining: ME 338: Manufacturing Processes II Instructor: Ramesh Singh Notes: Profs. Singh/Melkote/ColtonDocument59 paginiMachining: ME 338: Manufacturing Processes II Instructor: Ramesh Singh Notes: Profs. Singh/Melkote/ColtonGulshan Kumar SinhaÎncă nu există evaluări
- QCC GRESSMEN (Magnesium)Document43 paginiQCC GRESSMEN (Magnesium)Choirun Ni'amÎncă nu există evaluări
- BS Iso 4991Document27 paginiBS Iso 4991Thomas ValoÎncă nu există evaluări
- Astm-A 992-A992mDocument3 paginiAstm-A 992-A992medwinbadajosÎncă nu există evaluări
- R11 MTSR Mar 2019Document42 paginiR11 MTSR Mar 2019Jaypee DebandinaÎncă nu există evaluări
- Bhushan Traders - Delhi - Steel & Stainless Steel Manufacturers and SuppliersDocument3 paginiBhushan Traders - Delhi - Steel & Stainless Steel Manufacturers and Suppliersbiswasdipankar05Încă nu există evaluări
- Toilet and Bath Accessories - MSTDocument7 paginiToilet and Bath Accessories - MSTwafikmh4Încă nu există evaluări
- Higt Temp Failure Case LanceDocument2 paginiHigt Temp Failure Case LanceDevin Trianda PutraÎncă nu există evaluări
- Sa268 2019Document8 paginiSa268 2019CK CkkouÎncă nu există evaluări
- Common Fastener Related Abbreviations: Abbreviation MeaningDocument3 paginiCommon Fastener Related Abbreviations: Abbreviation MeaningezhiljananiÎncă nu există evaluări
- 5.9 Backing: AWS D1.1/D1.1M:2015 Clause 5. FabricationDocument1 pagină5.9 Backing: AWS D1.1/D1.1M:2015 Clause 5. FabricationRohit KambleÎncă nu există evaluări
- Astm A668mDocument7 paginiAstm A668mcacalinoÎncă nu există evaluări
- Riveted Joints Types and UsesDocument11 paginiRiveted Joints Types and UsesAbdurrahman shuaibÎncă nu există evaluări
- LPPM Filling ChiesaDocument8 paginiLPPM Filling ChiesaKmilo GiraldoÎncă nu există evaluări
- MAKALAH Pengolahan LogamDocument21 paginiMAKALAH Pengolahan Logamrahmat taufikÎncă nu există evaluări
- Unit III - Introduction To Manufacturing EngineeringDocument281 paginiUnit III - Introduction To Manufacturing EngineeringJohnÎncă nu există evaluări
- IS 4049-2 (1996) Formed Ends For Tanks and Pressure (Inside Diameter Basis) PDFDocument14 paginiIS 4049-2 (1996) Formed Ends For Tanks and Pressure (Inside Diameter Basis) PDFShivprasadÎncă nu există evaluări
- MME 291 Final QuestionDocument2 paginiMME 291 Final QuestionTahmeed HossainÎncă nu există evaluări
- RT Acceptance CriteriaDocument4 paginiRT Acceptance CriteriaSandipÎncă nu există evaluări
- Npi Vs FerronickelDocument4 paginiNpi Vs Ferronickelarkent98100% (1)
- Copper and Copper Alloy Clad Steel Plate: Standard Specification ForDocument6 paginiCopper and Copper Alloy Clad Steel Plate: Standard Specification ForAdnan KhanÎncă nu există evaluări
- Buehler Technotes - Indentation Testing MappingDocument3 paginiBuehler Technotes - Indentation Testing Mappingdev-nullÎncă nu există evaluări
- Cement Steel Rates of January 2021Document1 paginăCement Steel Rates of January 2021excutiveengineer rbnhwglÎncă nu există evaluări
- Corrosion (Study Material)Document8 paginiCorrosion (Study Material)SANDEEP NAYAKÎncă nu există evaluări
- WPS Api 1104Document85 paginiWPS Api 1104Jose nuñez100% (1)