Documente Academic
Documente Profesional
Documente Cultură
SEED No 1 - COAG - Principles of Haemostasis
Încărcat de
M Yusuf Ali RDescriere originală:
Titlu original
Drepturi de autor
Formate disponibile
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentDrepturi de autor:
Formate disponibile
SEED No 1 - COAG - Principles of Haemostasis
Încărcat de
M Yusuf Ali RDrepturi de autor:
Formate disponibile
Sysmex Educational Enhancement & Development
SEED-Africa Newsletter | No 1 | October 2010
Introduction to coagulation The purpose of this newsletter is to provide laboratory staff with a simple outline of the principles of haemostasis as the foundation for ensuring that only the best quality results are released and to facilitate logical troubleshooting when problems do occur.
Principles of haemostasis
The primary principle of haemostasis is to minimise blood loss at sites of vessel injury by forming a thrombus (clot) and at the same time maintaining blood flow (figure 1). In order to achieve this, there is a highly regulated, fine-tuned interaction of multiple processes, involving the blood vessel wall, principally the endothelium, platelets and non-cellular blood constituents.
The core elements of haemostasis include blood vessel constriction, platelet activation, coagulation and fibrinolysis. All of these processes are initiated at the same time in response to blood vessel injury. Damage to the endothelial cell lining of the blood vessel results in exposure of collagen which under normal circumstances is not in contact with the blood. When circulating platelets come into contact with collagen they are activated and start to stick to the damaged surface. At the same time a substance called tissue factor is also exposed which activates the coagulation cascade resulting in the formation of a fibrin clot. Platelet adhesion and activation is conventionally known as primary haemostasis and the process of coagulation and fibrin formation as secondary haemostasis. In simplistic terms, the platelet plug can be considered to be the bricks and the fibrin meshwork the cement, both being required for the formation of a stable thrombus.
Normal haemostasis depends on the interaction of the following:
a) b) c) d) Blood vessel Platelets Coagulation system Fibrinolytic system
A defect in one or more of these systems will result in either a bleeding disorder or a tendency to clot.
a) The blood vessel
The blood vessel wall which is lined with endothelial cells is the first line of defence for normal haemostasis. The endothelium is a highly regulated organ in that it is responsible for ensuring that blood remains fluid at all times so that flow continues without hindrance. The surface is therefore regulated to be highly anticoagulant most of the time.
Figure 1 Schematic representation of the principles of haemostasis
SEED-Africa Newsletter | No 1 | October 2010 Sysmex Educational Enhancement & Development
2/4
However, at the first sign of injury where blood loss could be incurred, the endothelium switches to be highly procoagulant.
b) Platelets
Platelets are produced in the bone marrow. They circulate in the blood and have a life span of about 10 days. Platelets have a very complex structure which facilitates the critical role that platelets play in the normal physiology of haemostasis. The major components of the platelet include the surface membrane composed mainly of phospholipids, intracellular granules, surface receptors and the canalicular system. Ordinarily, platelets circulate as inert particles, without interacting with the blood vessel wall or any blood constituent proteins. However, at sites of blood vessel damage, platelets stick to a variety of structures via a multitude of platelet surface receptors. Von Willebrand factor (VWF) is a plasma protein the function of which is to act as an anchor for platelets at sites of injury. VWF and platelets ordinarily do not interact, but once VWF has bound to subendothelial tissues, it changes its own structure which the platelet then recognizes and binds tightly. The bond takes place via one of the major surface receptors, which in turn triggers multiple reactions inside the platelet which lead to activation of the platelet.
(especially phospatidylserine) to the plasma which are normally housed primarily within the inner leaflet of the platelet membrane and hence separated from plasma proteins. This is a critical step in the haemostatic process as phospholipid is an essential element in the support of the coagulation cascade.
n
Fibrinogen receptor
Another vital consequence of platelet activation is the induction of a shape change in the main surface receptor for fibrinogen. As the fibrinogen molecule has two identical arms, one fibrinogen molecule is able to bind to fibrinogen receptors on adjacent platelets thereby linking them together. The overall effect of this is that there is now a physical seal comprising of stretched out platelets which are tethered to collagen in the vessel wall at the site of injury. The primary platelet plug by itself is however only a temporary seal, and the formation of a proper clot is needed in order to seal the vessel wall securely whilst the damaged vessel repairs itself. The exposed phosphatidylserine on the activated platelets surface is ready to support the coagulation cascade which has its substrate, fibrinogen right there, holding the platelets together.
c) Coagulation
Coagulation is the process of converting soluble fibrinogen into insoluble fibrin in order to form a stable blood clot around the platelets that were initially trapped at the site of injury. The plasma contains a large number of proteins called coagulation factors that participate in a series of enzymatic reactions that end in the formation of insoluble fibrin. A very small trigger results in the progressive amplification of each subsequent step which concludes in the production of relatively large amounts of fibrin. This sequential reaction is called the coagulation cascade. The clotting factors involved are factors XI, X, IX, VIII, VII, V and II. Factor II, also called prothrombin, becomes thrombin when activated. For normal haemostasis to occur, a sufficient amount of thrombin needs to be rapidly generated in order for fibrinogen to be converted to fibrin. Fibrin is relatively watertight and therefore further blood loss is prevented. Fibrin is also relatively resistant to fibrinolytic breakdown (see later). When the surface of the endothelium is damaged, coagulation is immediately activated by the exposure of tissue factor (TF), which is present below the endothelial cells in the vessel wall, to the coagulation factors present in plasma.
Platelet activation includes several key end points: n Shape change
Platelets undergo a shape change from disc like to flattened spread out structures with multiple finger-like extensions. This physically assists in plugging the hole in the vessel wall. The platelet is able to do this because it stores an abundance of excess membrane in the form of the canalicular system. This canalicular system is connected to the outside of the platelet and when activated, this excess membrane is pushed to the outside thereby greatly increasing the surface area.
n
Granule release
Platelets contain granules that are secreted when platelets are activated. These granules contain a number of key elements, such as factor V, VWF and calcium which further amplify the haemostatic process.
n
Membrane flip-flop
Platelet activation results in an irreversible flip-flop of its bilayer membrane resulting in the exposure of phospholipids
SEED-Africa Newsletter | No 1 | October 2010 Sysmex Educational Enhancement & Development
3/4
The coagulation cascade
The coagulation cascade as we understand it to exist today is shown in figure 2. The process of coagulation starts with the presence of TF, which binds and activates FVII to FVIIa. The activated clotting factors are enzymes that sequentially cleave each other in a highly regulated fashion. This process happens wherever TF has become exposed. Once the TF:FVIIa complex is formed, it has a potent positive feedback loop on itself thereby increasing the generation of more TF:FVIIa several thousand fold. TF:FVIIa in turn forms a complex with FX and converts it to FXa. It is also able to bind and activate FIX to FIXa although the intensity of this reaction is far less potent. FXa, together with the co-factor FVa, converts prothrombin to thrombin (FIIa). This initial generation of thrombin is very short-lived as TF:FVIIa is very rapidly inactivated by tissue factor pathway inhibitor (TFPI). Furthermore thrombin activates FXIII to FXIIIa which in turn is responsible for irreversibly cross-linking fibrin creating insoluble polymers and a stable clot. It can be said that the primary role of the initial small quantity of thrombin generated is to activate platelets within and close to the growing thrombus via thrombin platelet receptors and the thrombin produced from the secondary positive feedback activation loop of FXI by thrombin is responsible for the sustained burst of thrombin that generates the stable fibrin clot.
Figure 3 The role of calcium in coagulation
n
The importance of vitamin K and calcium
An essential element in the success of coagulation cascade is the assembly of the various proteases in an orderly fashion on the surface of the platelet. The key to this process is the presence of GLA (-carboxyglutamic acid) domains of the vitamin K dependent clotting factors which are factors II, VII, IX and X. These GLA domains of these clotting factors are the key to the assembly of these complexes on platelet surfaces as they provide the critical link. Calcium ions which are positively charged bind to various GLA domains and to the phospholipid of the platelet, both of which are negatively charged, thereby localizing the clotting factors to the platelet surface. The calcium ions essentially form a bridge between the clotting factor and the phospholipid on the activated platelet surface. This is how the clotting process is kept localized at the original site of endothelial injury. An absence of functional vitamin K will result in reduced ability for the coagulation factors to assemble on the platelet surface and compromise clot formation. NB: Calcium is a critical element in the laboratory tests of coagulation!
Figure 2 The revised coagulation cascade. PL refers to phospholipid which is the phosphatidylserine provided by the activated platelet surface.
SEED-Africa Newsletter | No 1 | October 2010 Sysmex Educational Enhancement & Development
4/4
d) Fibrinolysis
In the absence of adequate quantities of thrombin, a stable clot cannot be formed resulting in bleeding whereas unregulated production of thrombin will result in pathological thrombosis. A variety of mechanisms that collectively inhibit thrombin production have been well described. These include: n Removal of activated clotting factors by blood flow past the clot n Inactivation of clotting factors by circulating natural anticoagulants (protein C, protein S and antithrombin) n Fibrinolysis Fibrinolysis is the process of controlled clot breakdown. As soon as clot formation is triggered, so is the fibrinolytic pathway activated. The primary enzyme involved here is plasmin which degrades fibrin into fibrin breakdown products (FDPs) which includes D-Dimers.
The haemostatic balance
Despite the complexities of the control of haemostasis, ultimately the outcome is quite simply a matter of balance between clot formation and clot breakdown, both of which in turn are influenced by blood flow (or stasis), the vessel wall and the constituents of the blood (figure 4). The next newsletter will focus on the influence of preanalytical factors on coagulation test results.
Figure 4 A schematic representation of the concept of haemostatic balance. Normal haemostasis requires fine balance between clot formation and clot breakdown. Pathological thrombosis occurs when there is excess clotting and/or diminished fibrinolysis and pathological bleeding when clotting is reduced and/or fibrinolysis is enhanced. In this context pathological thrombosis refers to thrombus formation in excess of what is required to seal any damaged blood vessel and pathological bleeding refers to any episode of bleeding which is more severe than one would ordinarily expect based on the nature of the injury.
Sysmex South Africa (Pty) Ltd.
Fernridge Office Park Block 2, 5 Hunter Avenue, Ferndale, Randburg 2194 Phone +27 11 329 9480 Fax +27 11 789 9276 info@sysmex.co.za www.sysmex.co.za
S-ar putea să vă placă și
- Hemostasis, Hemorrhagic Disorders and ThrombosisDocument114 paginiHemostasis, Hemorrhagic Disorders and ThrombosisZeeÎncă nu există evaluări
- Unit IV HemostasisDocument49 paginiUnit IV Hemostasisalshads957Încă nu există evaluări
- Blood Vessels and Endothelial CellsDocument4 paginiBlood Vessels and Endothelial CellsJemmy FandriÎncă nu există evaluări
- What Are The Components That Play A Role in Hemostatis? - Endhotelial CellsDocument7 paginiWhat Are The Components That Play A Role in Hemostatis? - Endhotelial CellsArmella AzzahraÎncă nu există evaluări
- Hemostasis: Presenter-Dr. SonuDocument68 paginiHemostasis: Presenter-Dr. SonukiranÎncă nu există evaluări
- Basic Principles Underlying The Coagulation SystemDocument17 paginiBasic Principles Underlying The Coagulation SystemibntauhidÎncă nu există evaluări
- Bleeding Disorders1Document81 paginiBleeding Disorders1DrMuskan AroraÎncă nu există evaluări
- Schemi 1-2Document7 paginiSchemi 1-2lucaÎncă nu există evaluări
- Guyton Chapter 36Document6 paginiGuyton Chapter 36g_komolafe100% (1)
- Overview of Hemostasis - UpToDateDocument50 paginiOverview of Hemostasis - UpToDateTrung LêÎncă nu există evaluări
- Platelets-Composition, Function, Hemostasis and Its Individual PhasesDocument14 paginiPlatelets-Composition, Function, Hemostasis and Its Individual PhasesHebsiba PonnayyanÎncă nu există evaluări
- Hemostasis, Clotting, EctDocument33 paginiHemostasis, Clotting, Ectblessing akataÎncă nu există evaluări
- Learning ObjectivesDocument12 paginiLearning ObjectivesjmcvicenteÎncă nu există evaluări
- 11 Coagulation PDFDocument51 pagini11 Coagulation PDFمحمد علي حريج / مسائيÎncă nu există evaluări
- Normal HaemostasisDocument36 paginiNormal HaemostasisReem EshraÎncă nu există evaluări
- Patho 26Document16 paginiPatho 26saddamixoÎncă nu există evaluări
- Platelet Protocols: Research and Clinical Laboratory ProceduresDe la EverandPlatelet Protocols: Research and Clinical Laboratory ProceduresÎncă nu există evaluări
- 1-2 Hemostasis PhysiologyDocument48 pagini1-2 Hemostasis PhysiologyHussein Al Saedi100% (2)
- Laboratory Evaluation of Hemostasis DisordersDocument15 paginiLaboratory Evaluation of Hemostasis Disordersssamuel.garrcia2Încă nu există evaluări
- SGD 1 LBM Ke 3.2Document15 paginiSGD 1 LBM Ke 3.2RaisÎncă nu există evaluări
- Introduction To Blood CoagulationDocument22 paginiIntroduction To Blood Coagulationemman_abzÎncă nu există evaluări
- Coagulation and The Clotting CascadeDocument13 paginiCoagulation and The Clotting CascadeCLAUDIAÎncă nu există evaluări
- ClottingDocument25 paginiClottingAtalabi AdebusolaÎncă nu există evaluări
- 1-4 Hemostasis, Surgical Bleeding and TransfusionDocument17 pagini1-4 Hemostasis, Surgical Bleeding and TransfusionRobin Tolentino100% (3)
- Labmed33 0948Document6 paginiLabmed33 0948VKTNÎncă nu există evaluări
- Hemostasis and Blood CoagulationDocument11 paginiHemostasis and Blood CoagulationRinta MoonÎncă nu există evaluări
- Normal HemostasisDocument34 paginiNormal Hemostasisمصطفي خندقاويÎncă nu există evaluări
- 5 Anticoagulation Medication - Vol 18 Issue 2 May 2006 OmfsDocument9 pagini5 Anticoagulation Medication - Vol 18 Issue 2 May 2006 Omfsapi-265532519Încă nu există evaluări
- Chapter 4 Hemostasis, Surgical Bleeding, and Transfusion 35Document13 paginiChapter 4 Hemostasis, Surgical Bleeding, and Transfusion 35Vladimir OstriaÎncă nu există evaluări
- CoagulationDocument3 paginiCoagulationHerho-nyl CesÎncă nu există evaluări
- Hema 2 AssignmentDocument3 paginiHema 2 AssignmentErnest CañosÎncă nu există evaluări
- ThrombosisDocument15 paginiThrombosisMichelle HutahurukÎncă nu există evaluări
- Primary HemostasisDocument10 paginiPrimary HemostasisMariella DingleÎncă nu există evaluări
- Bleeding DisordersDocument27 paginiBleeding DisordersIliana América AlarcónÎncă nu există evaluări
- Hematology 2011 Kaplan 51 61 PDFDocument11 paginiHematology 2011 Kaplan 51 61 PDFhiran.reisÎncă nu există evaluări
- Biology of HemostasisDocument20 paginiBiology of HemostasisVeronica TomaselloÎncă nu există evaluări
- PHS 221 HaemostasisDocument36 paginiPHS 221 Haemostasismetasynthronos748Încă nu există evaluări
- Coagulation PathwayDocument10 paginiCoagulation Pathwayamonra10Încă nu există evaluări
- Basic Concepts of Hematology (Hemostasis)Document53 paginiBasic Concepts of Hematology (Hemostasis)Hendra SshÎncă nu există evaluări
- Hemostasis: Primary and Secondary HemostasisDocument52 paginiHemostasis: Primary and Secondary HemostasisluckyÎncă nu există evaluări
- Primary and Secondary Hemostasis PDFDocument16 paginiPrimary and Secondary Hemostasis PDFMae AnnÎncă nu există evaluări
- HemostasisDocument17 paginiHemostasisHadi AdamÎncă nu există evaluări
- Hemostasis ReviewerDocument14 paginiHemostasis ReviewerDayledaniel SorvetoÎncă nu există evaluări
- Aula Af 2 PDFDocument49 paginiAula Af 2 PDFtobiasmanuel179Încă nu există evaluări
- Clinical Anesthesiolog1 BLOODDocument14 paginiClinical Anesthesiolog1 BLOODIsra JavedÎncă nu există evaluări
- 1) VasoconstrictionDocument7 pagini1) VasoconstrictionDwi Wahyu WulandariÎncă nu există evaluări
- Haemostasis: Dr. Ali ElhassanDocument45 paginiHaemostasis: Dr. Ali ElhassanMohja JaberÎncă nu există evaluări
- Essentials of Anticoagulation in Hemodialysis: Core CurriculumDocument12 paginiEssentials of Anticoagulation in Hemodialysis: Core CurriculumKavidu KeshanÎncă nu există evaluări
- Haemocompatibility of Blood Contacting Devices Repaired)Document16 paginiHaemocompatibility of Blood Contacting Devices Repaired)Sinéad LyonsÎncă nu există evaluări
- LBM 3 Hematopoietin Master - AndriDocument8 paginiLBM 3 Hematopoietin Master - AndriAndri Tri AtmojoÎncă nu există evaluări
- HemostasisDocument21 paginiHemostasisilyasÎncă nu există evaluări
- Admin, 32551Document14 paginiAdmin, 32551Buthaina HusseinÎncă nu există evaluări
- Physiology of CoagulationDocument16 paginiPhysiology of CoagulationIlyas HalimÎncă nu există evaluări
- Coagulation CascadeDocument41 paginiCoagulation CascadeJae TÎncă nu există evaluări
- Platelets InformationDocument15 paginiPlatelets Informationmuthurajan_hÎncă nu există evaluări
- Lecture On Hemostasis by Dr. RoomiDocument43 paginiLecture On Hemostasis by Dr. RoomiMudassar Roomi100% (1)
- HaematologyDocument68 paginiHaematologytapoolnoÎncă nu există evaluări
- Hemostasis: /coagulationDocument80 paginiHemostasis: /coagulationLeonardo Adi BestÎncă nu există evaluări
- Hema OmgDocument82 paginiHema OmgBernadeth BaduaÎncă nu există evaluări
- 3 - Toronto Notes 2011 - Anesthesia - and - Peri-Operative - MedicineDocument28 pagini3 - Toronto Notes 2011 - Anesthesia - and - Peri-Operative - MedicineHisham Qassrawi100% (1)
- Seed No 3 Coag Prothrombin TimeDocument4 paginiSeed No 3 Coag Prothrombin TimeM Yusuf Ali RÎncă nu există evaluări
- The Diagnostic Challenge of Joint Pain - Part2 PDFDocument5 paginiThe Diagnostic Challenge of Joint Pain - Part2 PDFM Yusuf Ali RÎncă nu există evaluări
- The Diagnostic Challenge of Joint Pain - Part1 PDFDocument5 paginiThe Diagnostic Challenge of Joint Pain - Part1 PDFM Yusuf Ali RÎncă nu există evaluări
- Homozygous Familial HypercholesterolaemiaDocument1 paginăHomozygous Familial HypercholesterolaemiaM Yusuf Ali RÎncă nu există evaluări
- The Essential Guide To Brain TumoursDocument43 paginiThe Essential Guide To Brain TumoursM Yusuf Ali RÎncă nu există evaluări
- SEED No 2 - Pre Analytical Factors That Influence Coagulation Test ResultsDocument4 paginiSEED No 2 - Pre Analytical Factors That Influence Coagulation Test ResultsM Yusuf Ali RÎncă nu există evaluări
- Modele de ParentsDocument4 paginiModele de ParentsM Yusuf Ali RÎncă nu există evaluări
- Noor E Muhammad Masjid DocumentsDocument1 paginăNoor E Muhammad Masjid DocumentsM Yusuf Ali RÎncă nu există evaluări
- Aupres Du Prophete Mouhammad SAWDocument4 paginiAupres Du Prophete Mouhammad SAWM Yusuf Ali RÎncă nu există evaluări
- Eye Can See A Nest of Worms!Document1 paginăEye Can See A Nest of Worms!M Yusuf Ali RÎncă nu există evaluări
- Sturge-Weber AngiomatosisDocument1 paginăSturge-Weber AngiomatosisM Yusuf Ali RÎncă nu există evaluări
- Nearly An Eye-CatcherDocument1 paginăNearly An Eye-CatcherM Yusuf Ali RÎncă nu există evaluări
- Bilateral Spontaneous Anterior Dislocation of Crystalline LensDocument1 paginăBilateral Spontaneous Anterior Dislocation of Crystalline LensM Yusuf Ali RÎncă nu există evaluări
- A Painful Cranial BulgeDocument1 paginăA Painful Cranial BulgeM Yusuf Ali RÎncă nu există evaluări
- Corkscrew Oesophagus On Virtual EndosDocument1 paginăCorkscrew Oesophagus On Virtual EndosM Yusuf Ali RÎncă nu există evaluări
- Retro Peritoneal Liposarcoma With Lamellar Bone InsideDocument1 paginăRetro Peritoneal Liposarcoma With Lamellar Bone InsideM Yusuf Ali RÎncă nu există evaluări
- Placental Vasa PreviaDocument1 paginăPlacental Vasa PreviaM Yusuf Ali RÎncă nu există evaluări
- Tic Choroidal MelanomaDocument1 paginăTic Choroidal MelanomaM Yusuf Ali RÎncă nu există evaluări
- Al Hidaya 161Document2 paginiAl Hidaya 161M Yusuf Ali RÎncă nu există evaluări
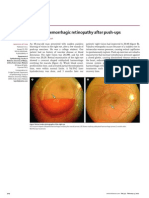
- Valsalva Haemorrhagic Thy After Push-UpsDocument1 paginăValsalva Haemorrhagic Thy After Push-UpsM Yusuf Ali RÎncă nu există evaluări
- ALHIDAYA162Document2 paginiALHIDAYA162M Yusuf Ali RÎncă nu există evaluări
- AlcoholDocument10 paginiAlcoholJean Paul GottretÎncă nu există evaluări
- Tuberculosis Sem LancetDocument16 paginiTuberculosis Sem LancetM Yusuf Ali RÎncă nu există evaluări
- Acute Liver FailureDocument12 paginiAcute Liver FailureM Yusuf Ali RÎncă nu există evaluări
- DilatedcardiomyopathyDocument11 paginiDilatedcardiomyopathyM Yusuf Ali RÎncă nu există evaluări
- JAMA 2011 Chang 767 8.... Skin ChangesDocument2 paginiJAMA 2011 Chang 767 8.... Skin ChangesM Yusuf Ali RÎncă nu există evaluări
- Unexpected Life Lessons From Cardiac Anatomy and PhysiologyDocument2 paginiUnexpected Life Lessons From Cardiac Anatomy and PhysiologyM Yusuf Ali RÎncă nu există evaluări
- MGT403 Slide All ChaptersDocument511 paginiMGT403 Slide All Chaptersfarah aqeelÎncă nu există evaluări
- Promoting Services and Educating CustomersDocument28 paginiPromoting Services and Educating Customershassan mehmoodÎncă nu există evaluări
- Project Report On HeritageDocument39 paginiProject Report On HeritageBALA YOGESH YANDAMURIÎncă nu există evaluări
- Sip Poblacion 2019 2021 Revised Latest UpdatedDocument17 paginiSip Poblacion 2019 2021 Revised Latest UpdatedANNALLENE MARIELLE FARISCALÎncă nu există evaluări
- Vietnamese Alphabet and PronounDocument10 paginiVietnamese Alphabet and Pronounhati92Încă nu există evaluări
- Thesis Statement On Corporate Social ResponsibilityDocument5 paginiThesis Statement On Corporate Social Responsibilitypjrozhiig100% (2)
- Ingres in ReproductionDocument20 paginiIngres in ReproductionKarlÎncă nu există evaluări
- PC300-8 New ModelDocument22 paginiPC300-8 New Modeljacklyn ade putra100% (2)
- 1 s2.0 S0378432004002465 MainDocument20 pagini1 s2.0 S0378432004002465 MainMuhammad JameelÎncă nu există evaluări
- Security Policy 6 E CommerceDocument6 paginiSecurity Policy 6 E CommerceShikha MehtaÎncă nu există evaluări
- Interceptor Specifications FinalDocument7 paginiInterceptor Specifications FinalAchint VermaÎncă nu există evaluări
- Digital Signatures: Homework 6Document10 paginiDigital Signatures: Homework 6leishÎncă nu există evaluări
- Alan Freeman - Ernest - Mandels - Contribution - To - Economic PDFDocument34 paginiAlan Freeman - Ernest - Mandels - Contribution - To - Economic PDFhajimenozakiÎncă nu există evaluări
- Form PersonalizationDocument5 paginiForm PersonalizationSuneelTejÎncă nu există evaluări
- Jail Versus Substance Abuse TreatmentDocument5 paginiJail Versus Substance Abuse Treatmentapi-240257564Încă nu există evaluări
- (Biophysical Techniques Series) Iain D. Campbell, Raymond A. Dwek-Biological Spectroscopy - Benjamin-Cummings Publishing Company (1984)Document192 pagini(Biophysical Techniques Series) Iain D. Campbell, Raymond A. Dwek-Biological Spectroscopy - Benjamin-Cummings Publishing Company (1984)BrunoRamosdeLima100% (1)
- Supreme Court Case Analysis-Team ProjectDocument5 paginiSupreme Court Case Analysis-Team ProjectJasmineA.RomeroÎncă nu există evaluări
- Using MonteCarlo Simulation To Mitigate The Risk of Project Cost OverrunsDocument8 paginiUsing MonteCarlo Simulation To Mitigate The Risk of Project Cost OverrunsJancarlo Mendoza MartínezÎncă nu există evaluări
- YS1700 Drum Level ControlDocument2 paginiYS1700 Drum Level ControlIdriss BarçaÎncă nu există evaluări
- Mystakidis2022 MetaverseDocument13 paginiMystakidis2022 MetaverseVennela NandikondaÎncă nu există evaluări
- D&D 3.5 Edition - Fiendish Codex I - Hordes of The Abyss PDFDocument191 paginiD&D 3.5 Edition - Fiendish Codex I - Hordes of The Abyss PDFIgnacio Peralta93% (15)
- Integrated Management System 2016Document16 paginiIntegrated Management System 2016Mohamed HamedÎncă nu există evaluări
- TSR 9440 - Ruined KingdomsDocument128 paginiTSR 9440 - Ruined KingdomsJulien Noblet100% (15)
- 3-CHAPTER-1 - Edited v1Document32 pagini3-CHAPTER-1 - Edited v1Michael Jaye RiblezaÎncă nu există evaluări
- Color Coding Chart - AHGDocument3 paginiColor Coding Chart - AHGahmedÎncă nu există evaluări
- Dreizler EDocument265 paginiDreizler ERobis OliveiraÎncă nu există evaluări
- CIVE2304 C1-1 Hydrological CycleDocument54 paginiCIVE2304 C1-1 Hydrological CyclenaserÎncă nu există evaluări
- CBSE Class 12 Informatics Practices Marking Scheme Term 2 For 2021 22Document6 paginiCBSE Class 12 Informatics Practices Marking Scheme Term 2 For 2021 22Aryan BhardwajÎncă nu există evaluări
- F. Moyra Allen: A Life in Nursing, 1921-1996: Meryn Stuart, R.N., PH.DDocument9 paginiF. Moyra Allen: A Life in Nursing, 1921-1996: Meryn Stuart, R.N., PH.DRose Nirwana HandayaniÎncă nu există evaluări
- Product 97 File1Document2 paginiProduct 97 File1Stefan StefanÎncă nu există evaluări