Documente Academic
Documente Profesional
Documente Cultură
7.aldehydes and Ketones New
Încărcat de
Mohd Nazri0 evaluări0% au considerat acest document util (0 voturi)
119 vizualizări36 paginiThe document summarizes key information about carbonyl compounds, specifically aldehydes and ketones. It discusses their structures, naming conventions, methods of preparation from alcohols, physical and chemical properties, and common uses. Aldehydes contain a carbonyl group bonded to the first carbon in an alkyl chain, while ketones have the carbonyl group within the chain. Both can be prepared by oxidizing primary or secondary alcohols, respectively. Their properties include solubility in water, boiling points, and participation in reactions like reductions, additions, and condensations. Common aldehydes and ketones are used as solvents, flavors, fragrances, and in chemical synthesis.
Descriere originală:
Titlu original
7.Aldehydes and Ketones New
Drepturi de autor
© © All Rights Reserved
Formate disponibile
PPT, PDF, TXT sau citiți online pe Scribd
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentThe document summarizes key information about carbonyl compounds, specifically aldehydes and ketones. It discusses their structures, naming conventions, methods of preparation from alcohols, physical and chemical properties, and common uses. Aldehydes contain a carbonyl group bonded to the first carbon in an alkyl chain, while ketones have the carbonyl group within the chain. Both can be prepared by oxidizing primary or secondary alcohols, respectively. Their properties include solubility in water, boiling points, and participation in reactions like reductions, additions, and condensations. Common aldehydes and ketones are used as solvents, flavors, fragrances, and in chemical synthesis.
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PPT, PDF, TXT sau citiți online pe Scribd
0 evaluări0% au considerat acest document util (0 voturi)
119 vizualizări36 pagini7.aldehydes and Ketones New
Încărcat de
Mohd NazriThe document summarizes key information about carbonyl compounds, specifically aldehydes and ketones. It discusses their structures, naming conventions, methods of preparation from alcohols, physical and chemical properties, and common uses. Aldehydes contain a carbonyl group bonded to the first carbon in an alkyl chain, while ketones have the carbonyl group within the chain. Both can be prepared by oxidizing primary or secondary alcohols, respectively. Their properties include solubility in water, boiling points, and participation in reactions like reductions, additions, and condensations. Common aldehydes and ketones are used as solvents, flavors, fragrances, and in chemical synthesis.
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PPT, PDF, TXT sau citiți online pe Scribd
Sunteți pe pagina 1din 36
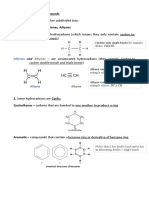
The carbonyl group is formed by joining together the
C atom and the O atom by a double bond.
Compounds having the aldehyde and ketone
functional groups are named carbonyl compounds.
Differences among aldehydes and ketones are due to
the positioning of the carbonyl group in their
molecules.
The C=O group in aldehydes is always located at the
end of the alkyl stem, so aldehydes can be represent
as
R C H
O
Unlike aldehydes, the ketone carbonyl group is situated
away from the ends of the parent carbon chain.
This means that the carbonyl group in ketones is
sandwiched between two alkyl stems and is
represented as
The group stamped a polar region in the aldehyde and
ketone molecules. Most reactions of aldehydes and
ketones will be centred at the carbonyl group.
R C R
O
Nomenclature:
The names of aldehydes and ketones are
simply derived by dropping "-e" from the root
and adding "-al" or "-one" respectively. A
position number is needed for ketones since
the carbonyl group may be on any number of
several carbons in the "middle" of a chain. The
carbonyl on the aldehyde is always on the
number one carbon so no position number is
needed.
Aldehyde -al
Propanal
Ketone -one Propanone
or
acetone
(common
name)
Preparation of Aldehydes and Ketones
Aldehydes can be prepared by the oxidation of
primary alcohols with hot, acidified potassium
manganate (VII) solution or potassium
dichromate(VI) solution.
R-C-OH + (O) R-C=O + H2O
H
H
H
aldehyde
Ketones are prepared by the oxidation of
secondary alcohols by hot acidified KMnO4
solution. Ketones resist oxidation, thus
distillation is not required.
R1-C-OH + (O) R1-C=O + H2O
R2
H
R2
Ketone
Physical Properties of
Aldehydes and Ketones
Physical Properties of Aldehydes and Ketones
a). Solubility in water
The small aldehydes and ketones are freely soluble in
water but solubility falls with chain length. For
example, methanal, ethanal and propanone - the
common small aldehydes and ketones - are miscible
with water in all proportions.
The reason for the solubility is that although
aldehydes and ketones can't hydrogen bond with
themselves, they can hydrogen bond with water
molecules.
As chain lengths increase, the hydrocarbon "tails" of the
molecules start to get in the way.
By forcing themselves between water molecules, they
break the relatively strong hydrogen bonds between
water molecules without replacing them by anything as
good. This makes the process energetically less
profitable, and so solubility decreases.
b). Boiling points
The other aldehydes and the ketones are liquids, with
boiling points rising as the molecules get bigger. The size
of the boiling point is governed by the strengths of the
intermolecular forces.
The other aldehydes and the ketones are liquids, with
boiling points rising as the molecules get bigger. The size
of the boiling point is governed by the strengths of the
intermolecular forces.
c). Density of aldehyde and ketones is lesser than water.
d). Aldehydes and ketones are polar molecules. The bond
angles and hybridization are the same. They also have
dipole-dipole intermolecular bonding. Because of this,
they are held together with greater attraction than you
would find with alkanes of similar size and molecular
weight.
e). They dont have hydrogen bonding but form hydrogen
bonding with the compound which have hydrogen
bonding.
f). They are excellent organic solvents.
Chemical Properties of
Aldehydes and Ketones
Chemical Properties of Aldehydes and
Ketones
Reduction
Variety of agents can reduce aldehydes and ketones to
alcohols
NaBH
4
and H
2
commonly used
Condensation Reactions
Aldehydes and ketones react with derivatives of
ammonia in condensation reactions.
Aldehydes and ketones react with hydroxylamine at
room temperature to form an oxime.
C= O+ H2N- ON C=N-OH+H2O
hydroxylamine oxime
Aldehydes and ketones react with hydrazine at room
temperature to form a hydrazone.
C= O+ H2N- NH C=N-OH+H2O
hydrazine hydrazone
Aldehydes and ketones react with
phenylhydrazine, at room temperature to form
a phenylhydration.
C= O+ H2N- H- C=N-N- + H2O
H H
phenylhydrazine phenyldrazone
The most important condensation reaction is
with 2, 4-dinitrophenyldrazine to form a yellow-
orange precipatate of 2, 4-
dinitrophenylhydrazone.
O2N O2N
C= O+ H2N- H- NO2 C=N-N- NO2+ H2O
H H
A solution of 2, 4- dinitrophenylhydrazine in
methanol and sulphuric acid is called Bradys
reagent.
The formation of a yellow- orange precipitate is
a chemical test for the presence of the carbonyl
group in aldehydes and ketones.
2, 4-dinitrophenyldrazine
2, 4- dinitrophenylhydrazone
Hydration
Formaldehyde dissolves readily in water
Acetaldehyde somewhat also
Form hydrates
Addition Reactions
Aldehydes and ketones react with hydrogen
cyanide in sodium hydroxide solution at 10-20
celcius to form cyanohydrin.
H H
CH3-C=O+H-C= N CH3-C-OH
C=
2- hydroxypropanenitrile
(ethanal cyanohydrin)
The reaction can also be carried put by adding
dilute sulphuric acid to a mixture of carbonyl
compound and potassium cyanide at 10-20
celcius.
The CN group in cyanohydrin can be
hydrolysed by heating with a dilute acid to form
a hydroxyacid. This is a method to prepare 2-
hydroxycarboxylic acid.
H + H
CH3-C=OH+H-H2O CH3-C-OH
C= N COOH
2-hydroxycarboxylic acid
This is a useful way to add a carbon atom to the
carbon chain.
The CN group can also be reduced to an amine
group using hydrogen gas in the presence of hot
nickel catalyst or by using lithium
tetrahydridoaluminate (III)
Oxidation
Aldehydes are oxidised rather easily to
carboxylic acids by heating with acidified
potassium manganate (VII) or potassium
dichromate (VI) solution.
H OH
R-C=O+[O] R-C=O
Aldehyde carboxylic acid
In alkaline conditions, the aldehyde is oxidised to
the salt of the corresponding carboxylic acid.
The carboxylic acid is formed upon addition of a
dilute acid.
H O-
R-C=O+[O]+OH- R-C=O+H2O
Aldehyde salt of carboxylic acid
O OH
R-C=O+H+ R-C=O
carboxylic acid
Benzaldehyde is oxidised to benzoic acid, which
is soluble in hot water, but when cooled, will
precipitate out as a white solid.
H OH
-C=O+[O] -C=O
Benzaldehyde benzoic acid
Ketones resist oxidation under normal conditions.
However they undergo oxidative cleavage when
boiled for a long time to form a mixture of products.
There will be a loss of carbon atoms in the form of
carbon dioxide.
An important industrial application of the oxidation
of ketones is the oxidation of cyclohexanone by nitric
acid to hexanedioic acid, which is a monomer in the
synthesis of nylon-6,6.
O O O
HO-C-CH2CH2CH2CH2-C-OH
cyclohexanone hexanedioic acid
Tollens reagent
When adding the aldehyde or ketone to Tollens'
reagent, the test tube is put in a warm water bath. If
the reactant under test is an aldehyde, Tollens' test
results in a silver mirror. If the reactant is a ketone, it
will not react because a ketone cannot be oxidized
easily. A ketone has no available hydrogen atom on the
carbonyl carbon that can be oxidized - unlike an
aldehyde, which has this hydrogen atom.
Fehling reagent
An aldehyde is added to the Fehling solution and the
mixture is heated. The aldehyde oxidizes to an acid
and cupric ions, which were complexed with the
tartrate ion, are reduced to cuprous ions, which
precipitate as red cuprous oxide. Ketones (except
alpha hydroxy ketones such as are present in ketoses
and other metabolites) and aromatic aldehydes do
not respond to the Fehling test.
Benedict's test
Benedict's reagent will detect the presence of
aldehydes.
Benedict's reagent contains blue copper(II) ions (Cu
2+
)
which are reduced to copper(I) (Cu
+
). These are
precipitated as red copper(I) oxide which is insoluble in
water.
Uses of Aldehydes and
Ketones
Hundreds of aldehydes and ketones are used as
flavoring, odouring and preserving agents e.g.,
vanillin and cinnamonaldehyde (flavors).
Glyceraldehyde is an intermediate in carbohydrate
metabolism and testosterone and progesterone are
hormones that relate to the biological role of these
compounds.
Aldehydes and ketones are being used by chemists
daily basically to synthesize other compounds.
In industry these compounds are used as solvents.
Uses of Aldehydes
A 40% formaldehyde known as formalin solution
can be used to preserve dead animals.
It is also used in the manufacture of polymeric
plastics like bakelite, melamine and urea-
formaldehyde glues.
Formaldehyde is used in the tanning of leather,
decolouring of vat dyes and as a reducing agent in
silvering of mirrors.
It is also used in manufacture of plywood.
Paraldehyde is a sweet smelling liquid used in
medicine as a hypnotic.
Acetaldehyde is used primarily as a starting
material in the manufacture of acetic acid, ethyl
acetate, vinyl acetate, polymers and drugs.
It is also used in silvering of mirrors.
Mataldehyde a polymer of acetaldehyde is used
as a solid fuel and for killing slugs when mixed
with bran.
Benzaldehyde is an almond extract used as a
flavoring agent as well as in perfumery.
It is used in the manufacture of dyes like
malachite green.
In the chemical industry it is also used as a
starting material for making a number of
organic compounds such as cinnamic acid,
benzoic acid, benzoyl chloride etc.
Uses of ketones
Acetone is a common fingernail polish remover and
is a solvent for lacquers and varnishes.
Acetone is used as a cleaning fluid and is very
flammable.
It is also used to prepare a number of compounds
such as chloroform, ketene acetic anhydride etc.
It finds further use in the manufacture of smokeless
powders cordite, celluloid.
2-Butanone (MEK, methyl ethyl ketone) finds use as
a solvent and paint stripper.
Carvone is used as spearmint flavoring. It is used as
caraway seed flavoring.
S-ar putea să vă placă și
- Carbonyl Compounds Aldehydes KetonesDocument58 paginiCarbonyl Compounds Aldehydes KetonesNur Aliyah Abdul RazakÎncă nu există evaluări
- Newer Methods of Preparative Organic Chemistry V2De la EverandNewer Methods of Preparative Organic Chemistry V2Wilhelm FoerstÎncă nu există evaluări
- Chemistry of Carbonyl CompoundsDocument28 paginiChemistry of Carbonyl CompoundsRhondene WintÎncă nu există evaluări
- Acids, Bases and Salts Chapter-4: Answer To The Short QuestionsDocument17 paginiAcids, Bases and Salts Chapter-4: Answer To The Short Questionskawsar22Încă nu există evaluări
- Chem 31: Reviewer For 2 DepexDocument27 paginiChem 31: Reviewer For 2 DepexAlma PabilaneÎncă nu există evaluări
- Class XII: Chemistry Chapter 11: Alcohols, Phenols and Ethers Top Concepts 1. Structure of Alcohols, Phenols and EthersDocument10 paginiClass XII: Chemistry Chapter 11: Alcohols, Phenols and Ethers Top Concepts 1. Structure of Alcohols, Phenols and EthersAshaÎncă nu există evaluări
- Experiment 3 Synthesis, Characterization and Ligand Exchange Equilibria of Potassium Tris (Oxalato) Ferrate (Iii)Document7 paginiExperiment 3 Synthesis, Characterization and Ligand Exchange Equilibria of Potassium Tris (Oxalato) Ferrate (Iii)skrim240100% (1)
- Pharmaceutical Organic Chemistry LecDocument51 paginiPharmaceutical Organic Chemistry Lecبن آجرومÎncă nu există evaluări
- Synthesis and Characterization of TrisDocument1 paginăSynthesis and Characterization of TrisforfunÎncă nu există evaluări
- Penicillium Grew, The Streptomyces Were UnableDocument3 paginiPenicillium Grew, The Streptomyces Were UnableCamille Andrea RositaÎncă nu există evaluări
- Synthesis of Acetophenone DerivativesDocument6 paginiSynthesis of Acetophenone DerivativesAwad SaidÎncă nu există evaluări
- An Efficient Method For The Synthesis of 1,5-Benzodiazepine Derivatives Under Microwave Irradiation Without SolventDocument4 paginiAn Efficient Method For The Synthesis of 1,5-Benzodiazepine Derivatives Under Microwave Irradiation Without SolventHaouassi LakhdarÎncă nu există evaluări
- Organic ChemistryDocument8 paginiOrganic ChemistryAndré Brincat100% (1)
- A-Bromination Using HBR H2O2 APKDocument7 paginiA-Bromination Using HBR H2O2 APKAshutosh BhaveÎncă nu există evaluări
- Lab Activity 5Document5 paginiLab Activity 5Jasmin CeciliaÎncă nu există evaluări
- Oppenauer OxidationDocument38 paginiOppenauer OxidationHarumi Nabila RidzkiÎncă nu există evaluări
- BCHCT 131 Block-4eDocument100 paginiBCHCT 131 Block-4esarath chandranÎncă nu există evaluări
- Carbonyl Compounds Aldehydes and Ketones3Document4 paginiCarbonyl Compounds Aldehydes and Ketones3Sachitra WijethungaÎncă nu există evaluări
- PhenolDocument3 paginiPhenolShi Han75% (4)
- Chapter 18 - Carbonyl CompoundsDocument9 paginiChapter 18 - Carbonyl CompoundsNabindra RuwaliÎncă nu există evaluări
- Alcohol, Aldehyde and KetonesDocument12 paginiAlcohol, Aldehyde and KetonesFranky TeeÎncă nu există evaluări
- Enhancing Nabh4 Reactivity and Selectivity PDFDocument15 paginiEnhancing Nabh4 Reactivity and Selectivity PDFjohannes karcherÎncă nu există evaluări
- CHEM 2425. Chapter 28. Biomolecules - Nucleic Acids (Homework) WDocument15 paginiCHEM 2425. Chapter 28. Biomolecules - Nucleic Acids (Homework) WPhú NguyễnÎncă nu există evaluări
- Cannizarorxn 120207190937 Phpapp01Document71 paginiCannizarorxn 120207190937 Phpapp01Adrian PIÎncă nu există evaluări
- 06 Chapter 1Document63 pagini06 Chapter 1Kautsar NurfalaqÎncă nu există evaluări
- Biochemistry and Biophysics ReportsDocument7 paginiBiochemistry and Biophysics ReportsLesly LópezÎncă nu există evaluări
- Preparation and Reaction of Carboxylic AcidsDocument6 paginiPreparation and Reaction of Carboxylic AcidsIndhumathiÎncă nu există evaluări
- Reactions of Alcohols, Phenols, Aldehydes and KetonesDocument44 paginiReactions of Alcohols, Phenols, Aldehydes and KetonesGlen Mangali100% (4)
- Aldol Notes PDFDocument8 paginiAldol Notes PDFAna100% (1)
- SolubilityDocument9 paginiSolubilityEric AllanigueÎncă nu există evaluări
- Aldol Condensation and Synthesis of DibenzalacetoneDocument8 paginiAldol Condensation and Synthesis of DibenzalacetoneArturo CamañoÎncă nu există evaluări
- RXN of Sulphuric Acid With SaltsDocument16 paginiRXN of Sulphuric Acid With SaltsSteve ClowesÎncă nu există evaluări
- Jam - 3 - 3 - 1 A Developed Process For The Synthesis of 2-Ethyl Phenyl Hydrazine Hydrochloride, A Key Starting Material For 7-Ethyl Tryptophol.Document4 paginiJam - 3 - 3 - 1 A Developed Process For The Synthesis of 2-Ethyl Phenyl Hydrazine Hydrochloride, A Key Starting Material For 7-Ethyl Tryptophol.jamonline100% (3)
- Solubility Behavior of Organic CompoundsDocument2 paginiSolubility Behavior of Organic CompoundsIlac CapangpanganÎncă nu există evaluări
- Oxidation of Cyclohexanol To Adipic AcidDocument29 paginiOxidation of Cyclohexanol To Adipic AcidDavid Joram Mendoza67% (6)
- Hydrazine Formate ReductionsDocument3 paginiHydrazine Formate ReductionsPedro Mendonca100% (1)
- Ninety Years of Using Azo Compounds of The Pyridine SeriesDocument5 paginiNinety Years of Using Azo Compounds of The Pyridine SeriesrajdewaanÎncă nu există evaluări
- Alexandra Doddridge, Michael Collins and Helen SalourosDocument26 paginiAlexandra Doddridge, Michael Collins and Helen SalourosDoc MartenzÎncă nu există evaluări
- Preparation and Purification of An Alkyl Halide FRDocument6 paginiPreparation and Purification of An Alkyl Halide FRCamille GrefaldiaÎncă nu există evaluări
- Organic Chemistry - Study of Hydrocarbons NotesDocument17 paginiOrganic Chemistry - Study of Hydrocarbons Noteszarna nirmal rawalÎncă nu există evaluări
- Name Reactions in Organic ChemistryDocument16 paginiName Reactions in Organic Chemistrysatyamd1979Încă nu există evaluări
- Anthranilic Acid PDFDocument20 paginiAnthranilic Acid PDFGlibÎncă nu există evaluări
- Families of Organic CompoundsDocument8 paginiFamilies of Organic CompoundsJessa Mae LangcuyanÎncă nu există evaluări
- 06 Chapter 1Document63 pagini06 Chapter 1Dintakurthi PrasanthÎncă nu există evaluări
- Bouveault-Blanc Ester ReductionDocument3 paginiBouveault-Blanc Ester ReductionAriel GarciaÎncă nu există evaluări
- Classification and Identification of AldehydesDocument9 paginiClassification and Identification of AldehydesMeh Neff100% (1)
- Leuckart ReactionDocument4 paginiLeuckart ReactionangelofgloryÎncă nu există evaluări
- CHE143 Organic Chemistry Lab Report CompDocument8 paginiCHE143 Organic Chemistry Lab Report CompSN2-0618 Nur Anith Aqilah Binti Mohd RaniÎncă nu există evaluări
- Solvent Free Reduction of Aromatic Nitro Compounds With Alumina Supported Iron Powder and Acetic Acid Under Microwave IrradiationDocument5 paginiSolvent Free Reduction of Aromatic Nitro Compounds With Alumina Supported Iron Powder and Acetic Acid Under Microwave IrradiationKybernetikumÎncă nu există evaluări
- Experiment 12 Identification of Alcohols and PhenolsDocument6 paginiExperiment 12 Identification of Alcohols and Phenolsh1iraqÎncă nu există evaluări
- Synthesis of Mandelic AcidDocument6 paginiSynthesis of Mandelic AcidBea A.Încă nu există evaluări
- Organic Chemistry 2Document298 paginiOrganic Chemistry 2arielÎncă nu există evaluări
- Experiment 9 Organic Chemistry LabDocument7 paginiExperiment 9 Organic Chemistry LabRhodelyn TolentinoÎncă nu există evaluări
- Lab+Manual+2014 Cbe 2207Document63 paginiLab+Manual+2014 Cbe 2207Krishnan Mohan100% (1)
- A Developed Process For The Synthesis of Tryptophol A Key Starting Material For Indoramin PDFDocument4 paginiA Developed Process For The Synthesis of Tryptophol A Key Starting Material For Indoramin PDFAntares1973Încă nu există evaluări
- Aminative Reduction of KetonesDocument2 paginiAminative Reduction of Ketonesgeovani2100% (1)
- Pinacol RearrangementDocument2 paginiPinacol RearrangementkarinadegomaÎncă nu există evaluări
- 15 - Aldehyde and KetonesDocument66 pagini15 - Aldehyde and KetonesIrfan Raza100% (1)
- Alcohols From Carbonyl Compounds: Oxidation-Reduction: Central Linking Role of Alcohols and CarbonylsDocument12 paginiAlcohols From Carbonyl Compounds: Oxidation-Reduction: Central Linking Role of Alcohols and CarbonylsAmihanÎncă nu există evaluări
- 14 PPGWAJ3102 Topic 7 Lreading SkillsDocument37 pagini14 PPGWAJ3102 Topic 7 Lreading SkillsMohd NazriÎncă nu există evaluări
- 09 PPGWAJ3102 Topic 2 TensesDocument5 pagini09 PPGWAJ3102 Topic 2 TensesMohd NazriÎncă nu există evaluări
- 13 PPGWAJ3102 Topic 6 Listening & Speaking SkillsDocument6 pagini13 PPGWAJ3102 Topic 6 Listening & Speaking SkillsMohd NazriÎncă nu există evaluări
- 08 PPGWAJ3102 Topic1 Parts of SpeechDocument23 pagini08 PPGWAJ3102 Topic1 Parts of SpeechMohd NazriÎncă nu există evaluări
- Conservation and Human Societies: - Preservation of Biological Diversity Is Often in - What IsDocument29 paginiConservation and Human Societies: - Preservation of Biological Diversity Is Often in - What IsMohd NazriÎncă nu există evaluări
- L07 BiodiversityDocument35 paginiL07 BiodiversityMohd NazriÎncă nu există evaluări
- L05perkihdmatan EkosistemDocument18 paginiL05perkihdmatan EkosistemMohd NazriÎncă nu există evaluări
- Ester & AmidesDocument13 paginiEster & AmidesMohd NazriÎncă nu există evaluări
- Chapter 11Document53 paginiChapter 11Vijendhar ReddyÎncă nu există evaluări
- GGGB6023 Tugasan Tutorial 3 - P69060 Mior SyazrilDocument5 paginiGGGB6023 Tugasan Tutorial 3 - P69060 Mior SyazrilAmizan AbdullahÎncă nu există evaluări
- Camcor Coriolis Meter User ManualDocument140 paginiCamcor Coriolis Meter User ManualGerardo OrtigozaÎncă nu există evaluări
- FB-150 FQ-150 Basket StrainerDocument1 paginăFB-150 FQ-150 Basket Strainerklich77Încă nu există evaluări
- IECEx INE 12.0010X 002Document9 paginiIECEx INE 12.0010X 002Marcos SiqueiraÎncă nu există evaluări
- Coding 3 D ShapesDocument10 paginiCoding 3 D ShapesgmconÎncă nu există evaluări
- Electric Power Applications, Engine & Generator SizingDocument120 paginiElectric Power Applications, Engine & Generator SizingDIPPOSÎncă nu există evaluări
- Unit Iv Ce 6405Document13 paginiUnit Iv Ce 6405HanafiahHamzahÎncă nu există evaluări
- BMG Idle CatlogueDocument20 paginiBMG Idle Catloguechintan mistryÎncă nu există evaluări
- DCS800 Firmware Manual EnglishDocument298 paginiDCS800 Firmware Manual EnglishMadson FernandesÎncă nu există evaluări
- Alarm ListDocument4 paginiAlarm ListMirela Perenda ŠabićÎncă nu există evaluări
- Direct Synthesis of Sodalite From Kaolin: The Influence of AlkalinityDocument7 paginiDirect Synthesis of Sodalite From Kaolin: The Influence of AlkalinityIsye RahmaenaÎncă nu există evaluări
- Clipping Clamping CircuitsDocument7 paginiClipping Clamping CircuitsPavan SaiÎncă nu există evaluări
- Reflexive Pronoun: Object SubjectDocument5 paginiReflexive Pronoun: Object SubjectSiti Sarah Abdullah100% (1)
- 351 DatasheetDocument14 pagini351 DatasheetRafael NavarroÎncă nu există evaluări
- Diagbootx: // Public Release 1Document4 paginiDiagbootx: // Public Release 1Tedy AdhinegoroÎncă nu există evaluări
- LS Retail Data Director User GuideDocument28 paginiLS Retail Data Director User GuideJad EL HittiÎncă nu există evaluări
- Applied Physics: Ayesha Anwer Ul Haq Qureshi MCS Karachi University (UBIT) E-Mail: Ayesha - Anwer@indus - Edu.pkDocument37 paginiApplied Physics: Ayesha Anwer Ul Haq Qureshi MCS Karachi University (UBIT) E-Mail: Ayesha - Anwer@indus - Edu.pkEhsan JavedÎncă nu există evaluări
- Omega PTFE Needle Valve - FVLT100Document1 paginăOmega PTFE Needle Valve - FVLT100XavierÎncă nu există evaluări
- TB-2100 Operational and Maintenance Manual Rev C OptimiseDocument224 paginiTB-2100 Operational and Maintenance Manual Rev C OptimisearnaudmarielÎncă nu există evaluări
- Cobol Language Fundamentals: Structured COBOL Programming, Stern & Stern, 9th Ed ItionDocument53 paginiCobol Language Fundamentals: Structured COBOL Programming, Stern & Stern, 9th Ed ItionAsk NameÎncă nu există evaluări
- MAJOR14Document14 paginiMAJOR14swathi100% (1)
- TM4320 PDFDocument85 paginiTM4320 PDFandres cortezÎncă nu există evaluări
- IIEE La Union Harmonics AnalysisDocument128 paginiIIEE La Union Harmonics AnalysisCarlnagum 123456789Încă nu există evaluări
- V-Ray For SketchUp Rendering An Exterior Scene PDFDocument7 paginiV-Ray For SketchUp Rendering An Exterior Scene PDFDevohÎncă nu există evaluări
- What Is A Stress Intensification FactorDocument7 paginiWhat Is A Stress Intensification FactorMahendra RathoreÎncă nu există evaluări
- Microsoft WordDocument79 paginiMicrosoft Wordthamel_09Încă nu există evaluări
- Shared Memory ArchitectureDocument2 paginiShared Memory ArchitectureNeethu RajeshÎncă nu există evaluări
- JAVA NotesDocument12 paginiJAVA NotesVarun BawaÎncă nu există evaluări