Documente Academic
Documente Profesional
Documente Cultură
Olivia Herliani, DR., M.Si: Darah
Încărcat de
Daniel Victor0 evaluări0% au considerat acest document util (0 voturi)
27 vizualizări82 paginiKHVK
Titlu original
Darah NEW
Drepturi de autor
© © All Rights Reserved
Formate disponibile
PPT, PDF, TXT sau citiți online pe Scribd
Partajați acest document
Partajați sau inserați document
Vi se pare util acest document?
Este necorespunzător acest conținut?
Raportați acest documentKHVK
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PPT, PDF, TXT sau citiți online pe Scribd
0 evaluări0% au considerat acest document util (0 voturi)
27 vizualizări82 paginiOlivia Herliani, DR., M.Si: Darah
Încărcat de
Daniel VictorKHVK
Drepturi de autor:
© All Rights Reserved
Formate disponibile
Descărcați ca PPT, PDF, TXT sau citiți online pe Scribd
Sunteți pe pagina 1din 82
DARAH
Olivia Herliani, dr., M.Si
Aspek Biomedis
• Homeostasis
• Penelitian → perkembangan biokimia +
biokimia klinis
• ↑↓ protein plasma (enzim, Ig) → dx px ttn
Fungsi Darah
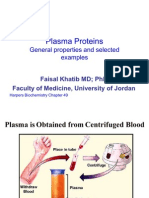
Plasma Darah
• O/ plasma kecuali transport O2 + cell mediated
immunologic defense
• Plasma: air, elektrolit, metabolit, nutrien, protein,
hormon
• Komposisi air & elektrolit: plasma = cairan
ekstraseluler
• Konsentrasi total protein ± 7.0–7.5 g/dL → major
solids part of plasma.
• Protein → complex mixture: simple proteins +
conjugated proteins (glyco-, lipoproteins)
Plasma Darah
• Proteomic techniques → isolasi + karakterisasi →
↑ plasma proteome
• Salting out: pemisahan protein dari kompleks
solven dan/atau elektrolit → sifat kelarutan →
fraksi → 3 kelompok besar: fibrinogen, albumin,
globulin
• Electrophoresis: supporting mediums (lab klinis:
selulose asetat) → stained strip (elektroforetogram)
→ albumin, α1, α2, β, γ globulin → densitometer
(kuantitas)
Protein plasma
Protein Plasma
Starling Forces
• Arteriol: tekanan hidrostatik 37 mmHg –
tekanan interstitial/tissue 1 mmHg - tekanan
osmotik/oncotik protein plasma 25 mmHg =
outward force 11 mm Hg → cairan keluar ke
ruang interstitial
• Venul: tekanan hidrostatik 17 mmHg - tekanan
interstitial/tissue 1 mmHg - tekanan
osmotik/oncotik protein plasma 25 mmHg = 9
mmHg → menarik cairan kembali ke sirkulasi
Edema
• Eg, malnutrition → protein def → plasma
protein concentration ↓↓ → fluid not attracted
back into intravasc → accumulate in extravasc
tissue space
Protein Plasma
• Penelitian manusia & hewan → biosintesis,
turnover, struktur, fungsi, perubahan ∑ &
metabolisme pd px
• Gen → klon & struktur
• Antibodi protein plasma → presipitasi &
isolasi pure protein dari kompleks
• Isotop → jalur biosintesis & laju turnover
dalam plasma
Sintesa Protein Plasma
• Pdu: liver, kec γ-globulin (sel plasma),
beberapa yang lain (tempat lain, misal sel
endothelial)
• Secretory route : rough endoplasmic membrane
→ smooth endoplasmic membrane → golgi
app → secretory vesicles → plasma
• Synthesized as preproteins + initially contain
amino terminal signal peptides → various
posttranslational modifications (proteolysis,
glycosylation, phosphorylation, etc)
Sintesa Protein Plasma
• Transit times (hepatocyte → plasma) : 30 min -
several hours/> for individual proteins
• Most are glycoprotein → N- or O-linked
oligosaccharide chains, or both
• Ex alb → ≠ sugar residues
• Neuraminidase → removal terminal sialic acid
residues (ex ceruloplasmin) → ↓ ↓ T½ in
plasma
Sintesa Protein Plasma
Polimorfism Protein Plasma
• Polymorphism = mendelian or monogenic trait
in population in at least two phenotypes
• Eg, ABO blood group substances, α1-AT, Hp,
Tf, ceruloplasmin, Ig → electrophoresis
isoelectric focusing
T½ Protein Plasma
• Labeling isolated pure protein with 131I or
Cr51 → T½
• Alb → 20 d, Hp → 5 d
• Certain disease → altered T½
• Regional ileitis (Crohn disease) : inflamed
intestinal mucosa → >> plasma proteins (inc
alb) lost → protein-losing gastroenteropathy
→ T½ alb ↓ ≈ 1 d
Acute Phase Proteins
• Acute-phase proteins ↑↑ (CRP/C-reactive
protein, α1-AT, Hp, α1-acid glycoprotein,
fibrinogen) in:
• Acute inflammatory state or certain tissue
damage
• Chronic inflammatory state
• Cancer
• Stimulator: cytokine & IL-1 + IL-6, NFkB
Acute Phase Proteins
• CRP: stimulate classic complement pathway;
marker for tissue injury, infection &
inflammation, predictor in cardiovasc
• α1-AT: neutralize certain protease in
inflammation
Protein Plasma
Protein Plasma
Albumin
• 69 kDa
• Major protein of plasma (3.4–4.7 g/dL)
• ± 60% of total plasma
• ± 40% in the plasma, 60% in extracellular
• Liver → 12 g/day → 25% of total hepatic
protein synthesis, 50% of total hepatic secreted
protein
Albumin
• Preproprotein → signal peptide is removed
(cisternae of rough ER) → resulting
hexapeptide amino terminal is cleaved off
farther along the secretory pathway
Albumin
• Mature albumin : 1 polypeptide chain → 585
aa + 17 disulfide bonds
• Proteases → subdivided → 3 domains + f/
• Ellipsoidal shape → ≠ ↑ plasma viscosity (≈
elongated molecule such as fibrinogen)
• Low molecular mass + high concentration →
responsible for 75–80% of osmotic pressure
Albumin
• Liver disease → ↓ albumin-globulin ratio
• Protein malnutrition (eg kwashiorkor) → early
↓ albumin
• Alb prep → tx hemorrhagic shock, burns
Haptoglobin
• Hp binds extracorpuscular Hb in a tight
noncovalent complex (Hb-Hp)
• ∑: 40 – 180 mg of hemoglobin-binding
capacity/deciliter
• ± 10% Hb degraded/day → circulation →
extracorpuscular; 90% in old/damaged rbc →
degraded by cells of the histiocytic system
• 3 polymorphic forms : Hp 1-1, Hp 2-1, Hp 2-2
Haptoglobin
• Hb 65 kDa + simplest polymorphic form of Hp
(Hp 1-1) 90 kDa = Hb-Hp complex 155 kDa
• Free Hb → kidney glomerulus → tubules →
tends to precipitate therein
• Hb-Hp complex → ≠ glomerulus → prevent
loss of free Hb into the kidney → conserves Hb
iron
Transferrin
Transferrin
• 76 kDa β1-globulin
• 20 polymorphic forms
• Transports iron (2 mol of Fe3+ per mole of Tf)
: gut → bone marrow
• 200 billion (± 20 mL) rbc → catabolized/day
→ 25 mg of iron
• Tf binds to receptor (TfR1 & TfR2) on surfaces
of many cells → internalized by receptor-
mediated endocytosis
Transferrin
• Acid pH inside lysosome → iron dissociates
from protein → leaves endosome via DMT1 →
enter the cytoplasm
• ApoTf remains associated with its receptor →
returns to plasma membrane → dissociates
from its receptor → reenters the plasma →
picks up more iron → delivers needy cells
• N :Tf-iron turns over 10–20 x/day
Transferrin
• Concentration ± 300 mg/dL → can bind 300
μg of iron/dl → TIBC
• However, N only ⅓ saturated with iron
• Iron deficiency anemia : <
• Storage of excess iron (eg,hemochromatosis): >
Ceruloplasmin
• α2-globulin
• 160 kDa
• High copper content → blue color
• Carries 90% copper in plasma
• 1 ceruloplasmin binds 6 atoms of copper →
very tightly, copper ≠ exchangeable
• Albumin carries ~10% less tightly → donates
copper more readily → more important in
copper transport
α1-antiproteinase
• 52 kDa
• Single-chain protein : 394 aa
• > 90% of α1 fraction plasma
• Synthesized by hepatocytes + macrophages
• Principal serine protease inhibitor (serpin, or
Pi) of plasma
• Inhibits trypsin, elastase, and certain other
proteases by forming complexes with them
α1-antiproteinase
• 75 polymorphic forms
• Major genotype MM → phenotypic product
PiM
• Deficiency → ± 5% of emphysema in
genotype ZZ (PiZ) + in PiSZ heterozygotes →
secrete protein < PiMM
α1-antiproteinase
• ∑ α1-at ↓ + polymorphonuclear wbc ↑ in the
lung (eg, pneumonia) → lacks a countercheck
to proteolytic damage of the lung by proteases
(elastase)
α1-antiproteinase
• Methionine(residue 358) of α1-at ≈ binding to
proteases
• Smoking →oxidizes methionine → methionine
sulfoxide → inactivates it → α1-at no longer
neutralize proteases
• Devastating in (eg, PiZZ phenotype) who
already have low levels of α1-at
• Further diminution in α1-at by smoking → ↑
proteolytic destruction of lung tissue →
accelerating emphysema development
α1-antiproteinase
• α1-at iv (augmentation tx) → emphysema due
to α1-at def
• α1-at def liver disease : molecules of ZZ
phenotypeaccumulate + aggregate cisternae ER
of hepatocytes → hepatitis with consequent
cirrhosis → tx: liver transplantation
Immune system
• 3 : B lymphocytes, T lymphocytes, innate
immune system
• B lymphocytes : bone marrow cells →
synthesis of circulating humoral antibodies
(immunoglobulins)
• T lymphocytes : thymic origin → cell-mediated
immunologic processes (graft rejection,
hypersensitivity rx, malignant cells, viruses)
Immune system
• Innate immune system : against infection in a
non-specific manner, not adaptive (unlike B &
T cells), variety of cells (phagocytes,
neutrophils, natural killer cells, others)
Immunoglobulins
Immunoglobulins
Immunoglobulins
• Minimum 2 identical light (L) chains (23 kDa)
+ 2 identical heavy (H) chains (53–75 kDa) →
tetramer (L2H2) by disulfide bonds
• IgG : Y-shaped, binding Ag at both tips of Y
• Each chain → specific domains/regions ≈
structural and functional significance
• ½ L chain toward carboxyl terminal = constant
region (CL), ½ amino terminal = variable
region (VL)
Immunoglobulins
• ¼ H chain at amino terminals = variable region
(VH), ¾ = constant regions (CH1, CH2, CH3)
• Binding of specific Ag @ amino terminal
portions of H & L chains (VH & VL) →
protein chains (2 sheets of antiparallel distinct
stretches of aa)
• Ig digestion by papain → 2 Ag-binding
fragments (Fab) + 1 crystallizable fragment
(Fc) → Ig functions other than Ag direct
binding
Immunoglobulins
• 2 Fab regions → IgG bind 2 Ag molecules →
divalent
• Site on Ag to which an Ab binds = antigenic
determinant (epitope)
• Area in which papain cleaves Ig (between
CH1–CH2) = Hinge region → flexibility +
allows both Fab arms move independently →
helping them to bind to antigenic sites that
variable distances apart (eg, on bacterial
surfaces)
Immunoglobulins
• Fc & Hinge regions differ in the different
classes of Ab, but overall Ab structure is
similar (Y)
Immunoglobulins
• 2 general types of L chains : kappa (κ) &
lambda (λ) → distinguished on basis structural
differences of CL
• Always 2 κ or 2 λ L chains → never mixture of
κ&λ
• Humans : κ → more frequent
Immunoglobulins
• 5 classes of H chain (differences in CH
regions) : λ, α, μ δ, and ε → μ and ε chains
have 4 CH domains
• Type of H chain → class of Ig (IgG, IgA, IgM,
IgD, and IgE) → effector function
Immunoglobulins
Immunoglobulins
Immunoglobulins
Immunoglobulins
• Variable regions (VL & VH) → quite
heterogeneous → no 2 variable regions from
different humans have identical aa sequences
• Aa analyses : variable regions → relatively
invariable regions + other hypervariable
regions
• VL = 3 hypervariable regions; VH = 4
hypervariable regions
• Hypervariable regions (CDRs) → Ag binding
site ≈ specificity of Ab
Immunoglobulins
• Constant regions, particularly CH2 & CH3 (&
CH4 of IgM and IgE) → Fc → class specific
effector functions of different Ig, eg,
complement fixation or transplacental passage
• IgG only the basic tetrameric structure, while
IgA and IgM can exist as higher order
polymers of 2, 3 (IgA), or 5 (IgM) tetrameric
units
Immunoglobulins
• L & H chains synthesized as separate
molecules → assembled within B cell or
plasma cell → mature Ig
Hemostasis & Thrombosis
• Medical emergencies
• Coronary & cerebral arteries → major cause of
death
• For rational tx
• Hemostasis: cut or severed vessel → cessation
of bleeding
• Thrombosis: damaged or removed blood vessel
endothelium lining (eg ruptur of atherosclerotic
plaque)
Hemostasis & Thrombosis
• Hemostasis: intial vasoconstriction → ↓ blood
flow distal injury
• Hemostasis & thrombosis = 3 phases
• 1. formation of loose & temporary platelet
aggregate: platelets bind collagen →
thromboxane A2 + release ADP → activate
other platelet. Coagulation → thrombin →
further platelet activation. Platelets change
shape + fibrinogen → hemostatic
plug/thrombus
Hemostasis & Thrombosis
• 2. platelet aggregate + fibrin mesh → more
stable hemostatic plug/thrombus
• 3. partial or complete dissolution hemostatic
plug/thrombus by plasmin
Hemostasis & Thrombosis
• 3 types of thrombi/clots:
• White thrombus: platelets + fibrin + poor
erythrocytes; blood flow is rapid (arteries)
• Red thrombus: primarily red cells + fibrin;
morphologically = test tube, retarded blood
flow or stasis (eg, veins) with or without
vascular injury, or @ injury in conjunction with
an initiating platelet plug
• Fibrin deposit: very small blood vessels or
capillaries
Hemostasis & Thrombosis
Hemostasis & Thrombosis
Hemostasis & Thrombosis
• Extrinsic & intrinsic pathway → fibrin clot
formation
• Tissue injury → extrinsic: initiation of fibrin
clot
• Negatively charged surfaces in vitro, eg, glass
→ intrinsic
• Both → prothrombin activation → thrombin →
fibrinogen cleavage → fibrin clot
Hemostasis & Thrombosis
Hemostasis & Thrombosis
Hereditary Bleeding Disorders
• Hemophilia A: deficiency of f.VIII, X
chromosome-linked, history of royal families
of Europe
• Hemophilia B: deficiency of f.IX; clinical
features ≈ A, specific assays → distinguish
• Von Willebrand disease: 1% of population,
von Willebrand factor (endothelial cells →
large multimeric glycoprotein into plasma →
stabilizes f.VIII & promotes platelet adhesion)
deficiency or defect
Fibrinolysis
Fibrinolysis
Fibrinolysis
• Injury: vascular endothelium → inactive tissue
plasminogen activator (t-PA) + fibrin →
active → cleaves plasminogen → plasmin →
digests fibrin → soluble degradation products
→ dissolves clot → released into fluid phase
→ inactivated
• Monocytes, macrophages, fibroblasts,
epithelial cells → urokinase → main action:
degradation of extracellular matrix
Lab Tests
• Platelet count → number of platelet
• Bleeding time → overall test of platelet and
vessel wall function
• Platelet aggregation → responses to specific
aggregating agents
• Activated partial thromboplastin time (aPTT or
PTT) → intrinsic pathway; monitor heparin
therapy
Lab Tests
• Prothrombin time (PT) → extrinsic pathway;
effectivenessof oral anticoagulants such as
warfarin
• Thrombin time (TT)
• Concentration of fibrinogen
• Fibrin clot stability
• Fibrin degradation products
Stem Cells
• Stem cell: cell → unaltered daughter cells (ie,
self-renewal) & generate specialized cell types
(potency)
• Totipotent: all cells in organism
• Pluripotent: differentiate cells of 3 germ
layers
• Multipotent: only cells of closely related
family
• Unipotent: only 1 type of cell
Stem Cells
• Stem cells: embryonic & adult
• Adult → more limited capabilities to
differentiate → overcomed by genetic
approaches
Hematopoietic Stem Cells
Hematopoietic Stem Cells
• RBC & platelets → = diff until
megakaryocyte erythroid progenitors
• Lymphoid origin → multipotent progenitors
• WBC → common myeloid progenitors
• Regulator : stem cell factor, thrombopoietin,
various interleukins, erythropoietin, etc) +
transcription factors also involved
Hematopoietic Stem Cells
• Stem cell factor → cytokine important role in
proliferation of hematopoietic stem cells &
progeny
• Thrombopoietin → glycoprotein important
regulating production of platelets by the bone
marrow
• Interleukins → cytokines produced by
leukocytes; regulate hematopoiesis & immune
system
RBC
• F/: ↔ O2 & CO2, protons of tissue metabolism
• Membrane sorrounding Hb, no organelles
• ATP from glycolysis
• Biconcave shape + ion & water transport
• 120 d → ↓ ie hemolytic anemias
• Prod regulation by EPO
RBC
RBC
RBC
RBC
RBC
RBC
Anemia
• WHO: Hb <130 g/L (♂) and <120g/L (♀)
Anemia
THANK YOU
Take Home Exam
• Extrinsic & intrinsic pathways
• G6PD
• Spherocytosis & elliptocytosis
S-ar putea să vă placă și
- Plasma Proteins, Immunoglobulins and Blood CoagulationDocument45 paginiPlasma Proteins, Immunoglobulins and Blood CoagulationKurnia SaptaÎncă nu există evaluări
- University of The Immaculate Conception: ProteinDocument13 paginiUniversity of The Immaculate Conception: ProteinDams ZainalÎncă nu există evaluări
- Biochemistry of Blood 1Document35 paginiBiochemistry of Blood 1AMEER ALSAABRAWIÎncă nu există evaluări
- ABSITE Killer PlusDocument16 paginiABSITE Killer PlusMark Soliman100% (1)
- Metabolism Proteic - LP 1 Electroforeza 11.04.2016Document52 paginiMetabolism Proteic - LP 1 Electroforeza 11.04.2016Andreea ChiochiuÎncă nu există evaluări
- Enzyme Kinetics ProblemsDocument5 paginiEnzyme Kinetics ProblemsBlessy Gabayno100% (1)
- Biochem ABO IncompatibilityDocument62 paginiBiochem ABO IncompatibilityChi Samaniego100% (4)
- Quiz 6Document4 paginiQuiz 6JeevikaGoyal100% (1)
- The Blood: Rubie Maranan-Causaren, MSDocument73 paginiThe Blood: Rubie Maranan-Causaren, MSlady ann jimenez100% (3)
- Clinical Biochemistry ReviewDocument10 paginiClinical Biochemistry Reviewyaykatai100% (2)
- Transformation, Transduction, ConjugationDocument35 paginiTransformation, Transduction, ConjugationMaham ChÎncă nu există evaluări
- Fast Facts: Leukemia: From initial gene mutation to survivorship supportDe la EverandFast Facts: Leukemia: From initial gene mutation to survivorship supportÎncă nu există evaluări
- Omics SciencesDocument20 paginiOmics ScienceslokeshkasaniÎncă nu există evaluări
- MD-5 Plasma Cont. Blood CellssDocument20 paginiMD-5 Plasma Cont. Blood CellssvincenzoÎncă nu există evaluări
- 11 Blood BiochemistryDocument98 pagini11 Blood Biochemistryfeby ary annasÎncă nu există evaluări
- Plasma Proteins ImmunoglobulinsDocument71 paginiPlasma Proteins ImmunoglobulinsIdenyi Daniel EwaÎncă nu există evaluări
- Plasma Proteins 1Document46 paginiPlasma Proteins 1Faisal_Khatib_juÎncă nu există evaluări
- 1 Faisal Plasma ProteinsDocument23 pagini1 Faisal Plasma Proteinsraanja2Încă nu există evaluări
- Komposisi Darah: Oleh: Dr. Husnil Kadri, M.Kes Bagian Biokimia Fakultas Kedokteran Universitas Andalas PadangDocument32 paginiKomposisi Darah: Oleh: Dr. Husnil Kadri, M.Kes Bagian Biokimia Fakultas Kedokteran Universitas Andalas PadangkhalishaÎncă nu există evaluări
- 2962 Plasma Proteins PPT 53b668dea4a12Document35 pagini2962 Plasma Proteins PPT 53b668dea4a12mahmoud fuqahaÎncă nu există evaluări
- Plasma ProteinDocument79 paginiPlasma ProteinnadieÎncă nu există evaluări
- Physiology of Blood: H. Khorrami PH.DDocument81 paginiPhysiology of Blood: H. Khorrami PH.DHossein KhorramiÎncă nu există evaluări
- Physiology of The BloodDocument16 paginiPhysiology of The BlooddiytÎncă nu există evaluări
- PROTEINS Cert 2Document39 paginiPROTEINS Cert 2johnnymills606Încă nu există evaluări
- BiochemistryDocument81 paginiBiochemistryGlorivy E. Mora GonzalezÎncă nu există evaluări
- Congenital and Acquired Hemolytic Anemias: Ellis J. Neufeld MD, PHDDocument63 paginiCongenital and Acquired Hemolytic Anemias: Ellis J. Neufeld MD, PHDRabi DhakalÎncă nu există evaluări
- BloodDocument32 paginiBloodPushpa AdhikariÎncă nu există evaluări
- BiochemistryDocument65 paginiBiochemistryjosue gomezÎncă nu există evaluări
- BLOOD Dr. AshishDocument4 paginiBLOOD Dr. AshishIsmath JahangirÎncă nu există evaluări
- Pemeriksaan Fungsi HatiDocument46 paginiPemeriksaan Fungsi HatiNadyaWevtÎncă nu există evaluări
- Haematology: 1. Cellular Component 2. Fluid Component (Plasma)Document4 paginiHaematology: 1. Cellular Component 2. Fluid Component (Plasma)Khant Si ThuÎncă nu există evaluări
- 4 BloodDocument43 pagini4 BloodAn ThịnhÎncă nu există evaluări
- Enzymes Patterns in Different DiseasesDocument47 paginiEnzymes Patterns in Different DiseasesMustafa KhandgawiÎncă nu există evaluări
- Plasma ProteinDocument13 paginiPlasma ProteinShahroze AlamÎncă nu există evaluări
- Plasma ProteinsDocument44 paginiPlasma ProteinsTinta Jisha AnaswaraÎncă nu există evaluări
- Physiology of Plasma ProteinsDocument21 paginiPhysiology of Plasma Proteinsdr.a.k.gupta6924100% (4)
- Week 10 Discussion SectionDocument14 paginiWeek 10 Discussion Sectionwriter topÎncă nu există evaluări
- Plasma Proteins in Disease DiagnosisDocument81 paginiPlasma Proteins in Disease DiagnosisSaaqo Qasim100% (1)
- ProteinsDocument35 paginiProteinsFiolta Ivar Del CastilloÎncă nu există evaluări
- Lec 1 Plasma Proteins 2021Document35 paginiLec 1 Plasma Proteins 2021maimona abdoÎncă nu există evaluări
- Ch6 Electrolytes and Blood GasesDocument53 paginiCh6 Electrolytes and Blood GasesLara MasriÎncă nu există evaluări
- Lymphocytic Leukemia in A Golden Retriever Dog - A Case ReportDocument14 paginiLymphocytic Leukemia in A Golden Retriever Dog - A Case ReportGanesh DasaraÎncă nu există evaluări
- Ch4 Hemodyn PDFDocument113 paginiCh4 Hemodyn PDFMarcusÎncă nu există evaluări
- Jayanti Tokkas, Shalini Jain and Hariom Yadav: Corresponding AuthorDocument20 paginiJayanti Tokkas, Shalini Jain and Hariom Yadav: Corresponding AuthorgereseÎncă nu există evaluări
- EnzymesDocument18 paginiEnzymesRichard PoonÎncă nu există evaluări
- Ap Exam Supplement: Specifically Developed For This Year's AP ExamDocument10 paginiAp Exam Supplement: Specifically Developed For This Year's AP ExamGan PentonÎncă nu există evaluări
- Major and Mainor Blood GroupDocument42 paginiMajor and Mainor Blood GroupMichelle San Miguel FeguroÎncă nu există evaluări
- Law of Thermodynamics: - 1 - Energy Is Conserved - 2 - Increased Disorder - 3 - at Absolute Zero, Entropy Is ZeroDocument30 paginiLaw of Thermodynamics: - 1 - Energy Is Conserved - 2 - Increased Disorder - 3 - at Absolute Zero, Entropy Is Zerolitzzo8Încă nu există evaluări
- Introduction of Blood and Plasma Protein: Amit Sharma Roll No: 09 Mbbs 2020 BatchDocument41 paginiIntroduction of Blood and Plasma Protein: Amit Sharma Roll No: 09 Mbbs 2020 BatchAmit SharmaÎncă nu există evaluări
- Aspek Laboratorium Pemeriksaan ProteinDocument28 paginiAspek Laboratorium Pemeriksaan ProteinVania Nazhara FitrianaÎncă nu există evaluări
- Cardiovascular SystemDocument127 paginiCardiovascular Systembreanna coyneÎncă nu există evaluări
- DarahDocument33 paginiDarahmemeyÎncă nu există evaluări
- Endocrine System Module: Biochemistry and Molecular BiologyDocument179 paginiEndocrine System Module: Biochemistry and Molecular BiologyƯớc Của BếttingÎncă nu există evaluări
- 6 EnzymesDocument89 pagini6 EnzymesFathi MesoÎncă nu există evaluări
- Biochemistry EnzymesDocument18 paginiBiochemistry EnzymesGiorgi TamazashviliÎncă nu există evaluări
- All Clinical PathologyDocument100 paginiAll Clinical Pathologyمروة خليل حسن حسينÎncă nu există evaluări
- Blood Physio Formed Tech 2024Document80 paginiBlood Physio Formed Tech 2024Joshua BucioÎncă nu există evaluări
- Phys Offluidselectrolytes7 121107201529 Phpapp02Document38 paginiPhys Offluidselectrolytes7 121107201529 Phpapp02Rehab weridaÎncă nu există evaluări
- Plasma Proteins: Dr. NG Chin ThengDocument20 paginiPlasma Proteins: Dr. NG Chin ThengYaashinii PeriathambyÎncă nu există evaluări
- Thyroid Metabolism UwksDocument54 paginiThyroid Metabolism UwksDaniel VictorÎncă nu există evaluări
- ELEKTROFORESISDocument1 paginăELEKTROFORESISDaniel VictorÎncă nu există evaluări
- Imunologi TransplantasiDocument42 paginiImunologi TransplantasiDaniel VictorÎncă nu există evaluări
- Imunologi TransplantasiDocument42 paginiImunologi TransplantasiDaniel VictorÎncă nu există evaluări
- Loo Hariyanto Raharjo, Dr.,MsiDocument43 paginiLoo Hariyanto Raharjo, Dr.,MsiDaniel Victor100% (1)
- Penentuan Protein Menurut Kingsley / Metoda BiuretDocument25 paginiPenentuan Protein Menurut Kingsley / Metoda BiuretDaniel VictorÎncă nu există evaluări
- Porphyrins: & Bile PigmentsDocument31 paginiPorphyrins: & Bile PigmentsDaniel VictorÎncă nu există evaluări
- An Tibi OtikDocument8 paginiAn Tibi OtikDaniel VictorÎncă nu există evaluări
- Acs - Sindroma Koroner Akut (Ska)Document55 paginiAcs - Sindroma Koroner Akut (Ska)Daniel VictorÎncă nu există evaluări
- CT Scan Hernia TranscalvariaDocument1 paginăCT Scan Hernia TranscalvariaDaniel VictorÎncă nu există evaluări
- RespirationDocument26 paginiRespirationSarang2426Încă nu există evaluări
- Neuronal CommunicationDocument9 paginiNeuronal CommunicationNatalia RamirezÎncă nu există evaluări
- Mutation of MyostatinDocument8 paginiMutation of MyostatinAndres Amador FernandezÎncă nu există evaluări
- Lecture Virus 2022Document40 paginiLecture Virus 2022JenniferÎncă nu există evaluări
- En Ni NTA Spin Kit HandbookDocument36 paginiEn Ni NTA Spin Kit HandbookHu VoÎncă nu există evaluări
- Service Jobcards Details 2018181Document30 paginiService Jobcards Details 2018181Rohit Om TiwariÎncă nu există evaluări
- Cam PathwayDocument7 paginiCam PathwayMelanie CabforoÎncă nu există evaluări
- Cellular ReproductionDocument24 paginiCellular Reproductionapi-292966101Încă nu există evaluări
- Describing Variants: Recommendations For The Description of DNA ChangesDocument38 paginiDescribing Variants: Recommendations For The Description of DNA ChangesVasanthakumar rathinamÎncă nu există evaluări
- DNA, The Genetic Material Quiz AnswersDocument2 paginiDNA, The Genetic Material Quiz AnswersLyndon De CastroÎncă nu există evaluări
- Lec 05 Biomolecules SHS SY 2019 2020Document59 paginiLec 05 Biomolecules SHS SY 2019 2020Loise GalapateÎncă nu există evaluări
- Antibacterial Resistance: DR Ance Roslina.,M.Kes Bagian Mikrobiologi Fk-Umsu 2019Document45 paginiAntibacterial Resistance: DR Ance Roslina.,M.Kes Bagian Mikrobiologi Fk-Umsu 2019RahmiÎncă nu există evaluări
- Acjc H2 Bio P1Document19 paginiAcjc H2 Bio P1DD97Încă nu există evaluări
- Acetylcholine and Cholinergic Receptors: David A. BrownDocument10 paginiAcetylcholine and Cholinergic Receptors: David A. BrownfahmiÎncă nu există evaluări
- Pt. Pathlab Indonesia: (Laboratorium Klinik)Document1 paginăPt. Pathlab Indonesia: (Laboratorium Klinik)Aghiest Utungga Al BirruÎncă nu există evaluări
- Sequence AlignmentDocument17 paginiSequence Alignmentjohn949Încă nu există evaluări
- 11 CholesterolDocument48 pagini11 CholesterolClaudiaNathaliaÎncă nu există evaluări
- Engineering Metabolism Through Dynamic Control: SciencedirectDocument11 paginiEngineering Metabolism Through Dynamic Control: SciencedirectShampa SenÎncă nu există evaluări
- Four Major Drug TargetsDocument34 paginiFour Major Drug TargetsFlowerÎncă nu există evaluări
- Cloning Vectors For EukaryotesDocument14 paginiCloning Vectors For Eukaryotesavbhujle2Încă nu există evaluări
- Unit 3 Module 2 Science Grade 10 SummaryDocument6 paginiUnit 3 Module 2 Science Grade 10 SummaryDan Felix100% (4)
- DNA Replication Escape Room DocumentsDocument42 paginiDNA Replication Escape Room DocumentsVictoria WarrenÎncă nu există evaluări
- Earth & Life Science Mrs. Anya ManceraDocument7 paginiEarth & Life Science Mrs. Anya ManceraMarc ChanÎncă nu există evaluări
- Plant Genome ProjectDocument5 paginiPlant Genome Projectsamina iqbal100% (1)
- Biosintesis LipidDocument27 paginiBiosintesis LipidNovia EkaÎncă nu există evaluări
- Northern & Southern BlotsDocument17 paginiNorthern & Southern BlotsShawon RahmanÎncă nu există evaluări